Abstract
Rationale
Misuse or dislike of nicotine replacement treatments (NRTs) undermines their effectiveness. Brief testing among NRTs could allow tailoring by preference to improve outcome.
Objective
To test initial reactions/preferences to NRTs in a single session crossover design with guided use.
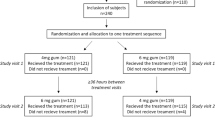
Methods
Smokers were offered two doses of three NRTs: gum (2 and 4 mg), inhaler, and nasal spray (NNS) in a 5-h test with proper use enforced. Subjects rated each NRT and ranked among NRTs on use variables and preferences.
Results
Gum was ranked over inhaler and NNS for “ease of use,” “safety” and “prefer in public.” Four-milligram gum was rated higher than 2 mg on several variables. With experience, “ease of use” and “liking” improved for gum. Both inhaler and NNS ranked low on considering “use >3 months” vs gum. Dislike of NRT was reflected in refusal of second doses. For those testing all doses (n=9), inhaler ranked last on “relief of withdrawal,” “choose under stress,” and “choice to help quit.” Craving and withdrawal were relieved over time with any NRT use.
Conclusions
Sampling of treatments can identify reactions key to initial compliance with these NRTs.

Similar content being viewed by others
References
Benowitz NL, Jacob P II, Savanapridi C (1987) Determinants of nicotine intake while chewing nicotine polacrilex gum. Clin Pharmacol Ther 41:467–473
Bohadana A, Nilsson F, Rasmussen T et al (2000) Nicotine inhaler and nicotine patch as a combination therapy for smoking cessation: a randomized, double-blind controlled trial. Arch Intern Med 160(20):3128–3134
Fagerstrom KO, Tejding R, Westin A, Lunell E (1997) Aiding reduction of smoking with nicotine replacement medications: hope for the recalcitrant smoker? Tob Control 6(4):311–316
Fiore MC, Bailey WC, Cohen SJ et al (2000) Treating tobacco use and dependence (clinical practice guideline). U.S. Department of Health and Human Services, Public Health Service, Rockville, MD
Foulds J, Burke M, Steinberg M et al (2004) Advances in pharmacotherapy for tobacco dependence. Expert Opin Emerg Drugs 9(1):39–53
Heatherton TF, Koslowski LT, Frecker RC, Fagerstrom KO (1991) The Fagerstrom Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict 86:1119–1127
Hughes JR, Shiffman S, Callas P, Zhang J (2003) A meta-analysis of the efficacy of over-the-counter nicotine replacement. Tob Control 12:21–27
Hurt RD, Offord KP, Croghan IT et al (1998) Temporal effects of nicotine nasal spray and gum on nicotine withdrawal symptoms. Psychopharmacology (Berl) 140(1):98–104
Leischow SJ, Valente SN, Hill AL et al (1997) Effects of nicotine dose and administration method on withdrawal symptoms and side effects during short-term smoking abstinence. Exp Clin Psychopharmacol 5(1):54–64
Niaura R, Sayette M, Shiffman S et al (in press) Comparative efficacy of rapid-release nicotine gum versus nicotine polacrilex gum in relieving smoking cue-provoked craving. Addiction
Sachs DPL (2005) California Thoracic Society. Position paper. Medical management for Tobacco Dependence. http://www.thoracic.org/chapters/california/publications.asp
Schneider NG (1986) Use of 2 mg and 4 mg nicotine gum in an individual treatment trial. In: Ockene JK (ed) Pharmacologic treatment of tobacco dependence: proceedings of the World Congress, 4–5 November 1985. Institute for the Study of Smoking Behavior and Policy, Cambridge, MA, pp 233–248
Schneider NG, Lunell E, Olmstead RE, Fagerstrom KO (1996) Clinical pharmacokinetics of nasal nicotine delivery: a review and comparison to other nicotine systems. Clin Pharmacokinet 31(1):65–80
Schneider NG, Olmstead RE, Franzon MA et al (2001) The nicotine inhaler: clinical pharmacokinetics and comparison with other nicotine treatments. Clin Pharmacokinet 40(9):661–684
Schneider NG, Olmstead RE, Nides M et al (2004) Comparative testing of 5 nicotine systems: initial use and preferences. Am J Health Behav 28(1):72–86
Shiffman S, Dresler CM, Hajek P et al (2002) Efficacy of a nicotine lozenge for smoking cessation. Arch Intern Med 162:1267–1276
Shiffman S, Fant RV, Gitchell JG et al (2003) Nicotine delivery systems: how far has technology come? Am J Drug Deliv 1(2):113–124
Silagy C, Lancaster T, Stead L et al (2004) Nicotine replacement therapy for smoking cessation. The cochrane database of systematic reviews 2004, Issue 3. Art. No.: CD000146.pub2. DOI: 10.1002/14651858.CD000146.pub2. Cited 19 July 2004
West R, Hajek P, Nilsson F et al (2001) Individual differences in preference for and responses to four nicotine replacement products. Psychopharmacology 153:225–230
Acknowledgements
Study funded with first author's VA Merit Review and NIDA Grant (DA 39811). Thanks to Dr. Martin Lee for additional help with statistics. This experiment complies with US laws for conducting human research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schneider, N.G., Terrace, S., Koury, M.A. et al. Comparison of three nicotine treatments: initial reactions and preferences with guided use. Psychopharmacology 182, 545–550 (2005). https://doi.org/10.1007/s00213-005-0123-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-0123-3