Abstract.
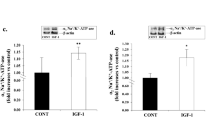
We tested the hypothesis that altered phosphorylation of Ca2+ regulatory proteins contributes to contractile anomalies in cardiac hypertrophy. Cardiac hypertrophy was induced in rats by chronic s.c. administration of isoproterenol (Iso, 2.4 mg/kg/day) via osmotic minipumps. On day 2 of Iso treatment the expression of atrial natriuretic factor was increased, time of relaxation in isolated papillary muscles shortened and protein expression of phospholamban (PLB) and sarcoplasmic reticulum Ca2+-ATPase reduced. In addition, the phosphorylation state of PLB at serine-16 and threonine-17 was decreased from (arbitrary units) 2.3±0.3 to 1.1±0.2 and from 4.1±0.6 to 2.1±0.2, respectively. This was not accompanied by altered activity of PLB-phosphorylating protein kinases (protein kinase A or Ca2+/calmodulin-dependent protein kinase II), whereas the activity of types 1 and 2A protein phosphatases (PP1 and -2A respectively) was enhanced from 1.1±0.08 to 1.71±0.13 nmol/mg/min. Iso treatment did not alter the PP1/PP2A activity ratio and 1 nmol/l okadaic acid, a concentration which completely blocks the catalytic subunit of PP2A, inhibited about 40% of total PP activity in all groups studied. These data indicate that the activity of both PP1 and PP2A were increased. All effects of Iso treatment were abolished by co-administration of propranolol (29.7 mg/kg/day). It is concluded that dephosphorylation of PLB is due to enhanced activity of PP1 and PP2A. We suggest that chronic β-adrenergic stimulation, which occurs in human cardiac hypertrophy and failure, can lead to increased activity of PPs. This may contribute to altered contractile responses in the hypertrophied heart.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Bokník, P., Fockenbrock, M., Herzig, S. et al. Protein phosphatase activity is increased in a rat model of long-term β-adrenergic stimulation. Naunyn-Schmied Arch Pharmacol 362, 222–231 (2000). https://doi.org/10.1007/s002100000283
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002100000283