Abstract
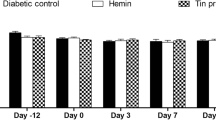
Oxidative injury is markedly responsible for wound complications in diabetes mellitus. The biological actions of bilirubin may be relevant to prevent oxidant-mediated cell death, as bilirubin application at a low concentration scavenges reactive oxygen species. Hence, we hypothesized that topical bilirubin application might improve wound healing in diabetic rats. Diabetes was induced in adult male Wistar rats, which were divided into two groups, i.e., diabetic control and diabetic treated. Non-diabetic healthy rats were also taken as healthy control group. Wound area was measured on days 3, 7, 14, and 19 post-wounding. The levels of malondialdehyde (MDA) and reduced glutathione (GSH) and the activities of glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) were estimated in the granulation tissue. There was a significant increase in percent wound closure in healthy control and diabetic treated rats on days 7, 14, and 19, as compared to diabetic control rats on days 7, 14, and 19. There was significant decrease in MDA levels on days 7, 14, and 19 in diabetic treated rats, as compared to diabetic control rats. Levels of GSH were significantly increased on days 3, 7, 14, and 19 in diabetic treated rats, as compared to diabetic control rats. GPx, SOD, and CAT activities were significantly higher on days 3, 7, and 14 in diabetic treated rats, as compared to diabetic control rats. The findings indicate that bilirubin is effective in reducing the oxidant status in wounds of diabetic rats which might have accelerated wound healing in these rats.



Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Aydyn A, Orhan H, Sayal A, Ozata M, Pahin G, Ibymer A (2001) Oxidative stress and nitric oxide related parameters in type II diabetes mellitus: effects of glycemic control. Clin Biochem 34:65–70
Barua CC, Talukdar A, Begum SA, Buragohain B, Roy JD, Pathak DC, Sarma DK, Gupta AK, Bora RS (2012) Effect of Alternanthera brasiliana (L) Kuntze on healing of dermal burn wound. Indian J Exp Biol 50:56–60
Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414:813–820
Carmody RJ, McGowan AJ, Cotter TG (1999) Reactive oxygen species as mediators of photoreceptor apoptosis in vitro. Exp Cell Res 248(2):520–530
Clark RAF (2008) Oxidative stress and “senescent” fibroblasts in non-healing wounds as potential therapeutic targets. J Invest Dermatol 128(10):2361–2364
Davi G, Falco A, Patrono C (2005) Lipid peroxidation in diabetes mellitus. Antioxid Redox Signal 7:256–268
Dissemond J, Goos M, Wagner SN (2002) The role of oxidative stress in the pathogenesis and therapy of chronic wounds. Hautarzt 53:718–723
Gumieniczek A, Hopkala H, Wojtowicz Z, Nikolajuk J (2002) Changes in antioxidant status of heart muscle tissue in experimental diabetes in rabbits. Acta Biochem Pol 49(2):529–535
Gupta R, Mathur M, Vijay KB, Katariya P, Yadav S, Kamal R, Gupta RS (2012) Evaluation of antidiabetic and antioxidant activity of Moringa oleifera in experimental diabetes. J Diabetes 4:164–171
Heck DE, Laskin DL, Gardner CR, Laskin JD (1992) Epidermal growth factor suppresses nitric oxide and hydrogen peroxide production by keratinocytes. Potential role for nitric oxide in the regulation of wound healing. J Biol Chem 267:21277–21280
Hunt JV, Smith CC, Wolff SP (1990) Autoxidative glycosylation and possible involvement of peroxides and free radicals in LDL modification by glucose. Diabetes 39:1420–1424
Ihara Y, Toyokuni S, Uchida K, Odaka H, Tanaka T, Ikeda H, Hiai H, Seino Y, Yamada Y (1999) Hyperglycemia causes oxidative stress in pancreatic beta-cells of GK rats, a model of type 2 diabetes. Diabetes 48:927–932
James FC, Maryanne D, Thomas GC (2002) Regulation and measurement of oxidative stress in apoptosis. J Immunol Methods 265:49–72
Kakkar R, Mantha SV, Radhi J, Prasad K, Kalra J (1998) Increased oxidative stress in rat liver and pancreas during progression of streptozotocin-induced diabetes. Clin Sci 94:623–632
King GL, Loeken MR (2004) Hyperglycemia-induced oxidative stress in diabetic complications. Histochem Cell Biol 122:333–338
Kowluru RA, Chan PS (2007) Oxidative stress and diabetic retinopathy. Exp Diabetes Res 2007:43603
Kushida T, LiVolti G, Goodman AI, Abraham NG (2003) TNF-α-mediated cell death is attenuated by retrovirus delivery of human heme oxygenase-1 gene into human microvessel endothelial cells. Transplant Proc 34:2973–2978
Lan Cheng-Che E, Ching-Shuang W, Huang Shu-Mei W, I-Hui G-SC (2013) High-glucose environment enhanced oxidative stress and increased interleukin-8 secretion from keratinocytes. New insights into impaired diabetic wound healing. Diabetes 62:2530–2538
Lledias F, Rangel P, Hansberg W (1998) Oxidation of catalase by singlet oxygen. J Biol Chem 273(17):10630–10637
Luo JD, Wang YY, Fu WL, Wu J, Chen AF (2004) Gene therapy of endothelial nitric oxide synthase and manganese superoxide dismutase restores delayed wound healing in type 1 diabetic mouse. Circulation 110(16):2484–2493
Madesh M, Balasubramanian KA (1998) Microtiter plate assay for superoxide dismutase using MTT reduction by superoxide. Indian J Biochem Biophys 35(3):184–188
Morse D, Choi AM (2005) Heme oxygenase-1: from bench to bedside. Am J Respir Crit Care Med 172:660–670
Oda T, Sadakata N, Komatsu N, Muramatsu T (1999) Specific efflux of glutathione from the basolateral membrane domain in polarized MDCK cells during ricin-induced apoptosis. J Biochem 126(4):715–721
Opara EC (2002) Oxidative stress, micronutrients, diabetes mellitus and its complications. J R Soc Promot Health 122:28–34
Osawa T, Kato Y (2005) Protective role of antioxidative food factors in oxidative stress caused by hyperglycemia. Ann NY Acad Sci 1043:440–445
Pompella A, Visvikis A, Paolicchi A, De Tata V, Casini AF (2003) The changing faces of glutathione, a cellular protagonist. Biochem Pharmacol 66:1499–1503
Ponrasu T, Kannappan MS, Ganeshkumar M, Suguna L (2013) Role of Annona squamosa on antioxidants during wound healing in streptozotocin nicotinamide induced diabetic rats. J Pharmacog Phytochem 2(4):77–84
Rao MC, Sudheendra AT, Nayak PG, Paul P, Kutty GN, Shenoy RR (2011) Effect of dehydrozingerone, a half analog of curcumin on dexamethasone-delayed wound healing in albino rats. Mol Cell Biochem 355:249–256
Rasik AM, Shukla A (2000) Antioxidant status in delayed healing type of wounds. Int J Exp Path 81:257–263
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179(73):588–590
Sabu MC, Kuttan R (2004) Antidiabetic activity of Aegle marmelos and its relationship with its antioxidant properties. Indian J Physiol Pharmacol 48:81–88
Sedlak J, Lindsay RHC (1968) Estimation of total, protein bound and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Segui J, Gironella M, Sans M, Granell S, Gil F, Gimeno M, Coronel P, Pique JM, Panes J (2004) Superoxide dismutase ameliorates TNBS-induced colitis by reducing oxidative stress, adhesion molecule expression and leukocyte recruitment into the inflamed intestine. J Leukoc Biol 76:537–544
Sen CK (2003) The general case for redox control in wound repair. Wound Repair Regen 11:431–438
Shafiq-Ur-Rehman (1984) Lead-induced regional lipid peroxidation in brain. Toxicol Lett 21(3):333–337
Shirwaikar A, Somashekar AP, Udupa AL, Udupa SL, Somashekar S (2003) Wound healing studies of the Aristolochia bracteolate Lam with supportive action of antioxidant enzymes. Phytomedicine 10:558–562
Singer AJ, Clark RAF (1999) Cutaneous wound healing. New Engl J Med 341(10):738–746
Stocker R, Yamamoto Y, McDonagh AF, Glazer AN, Ames BN (1987) Bilirubin is an antioxidant of possible physiological importance. Science 235:1043–1046
Thomas W, Sedlaka B, Masoumeh S, Daniel SH, Bindu DP, Krishna RJ (2009) Bilirubin and glutathione have complementary antioxidant and cytoprotective roles and Solomon H. Snyder. Proc Natl Acad Sci U S A 106(13):5171–5176
Venukumar MR, Latha MS (2002) Antioxidant activity of Curculigo orchioides in carbon tetra chloride-induced hepatopathy in rats. Indian J Clin Biochem 17:80–87
Wolff SP, Jiang ZY, Hunt JV (1991) Protein glycation and oxidative stress in diabetes mellitus and ageing. Free Radic Biol Med 10:339–352
Xu K, Thornalley PJ (2001) Involvement of glutathione metabolism in the cytotoxicity of the phenethyl isothiocyanate and its cysteine conjugate to human leukaemia cells in vitro. Biochem Pharmacol 61(2):165–177
Acknowledgments
The authors are thankful to the Director of Indian Veterinary Research Institute for providing funds and necessary facilities to conduct the study.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ram, M., Singh, V., Kumar, D. et al. Antioxidant potential of bilirubin-accelerated wound healing in streptozotocin-induced diabetic rats. Naunyn-Schmiedeberg's Arch Pharmacol 387, 955–961 (2014). https://doi.org/10.1007/s00210-014-1011-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-014-1011-3