Abstract
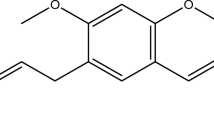
Accumulating evidence shows that iron overload is a new risk factor for diabetes mellitus. l-type Ca2+ channel (LTCC) has been identified as an important mediator for ferrous iron uptake into cardiomyocytes. In this study, we aimed to examine the effects of verapamil, the LTCC blocker, on myocardial iron metabolism in diabetic rats. Diabetes was induced by intraperitoneal injection of streptozocin after intragastric administration of fat emulsion, and then treated by verapamil (5 mg · kg−1 · day−1) for 1 week. The results showed that verapamil did not alter the blood glucose level of diabetic rats. However, elevated levels of superoxide dismutase, malonaldehyde, and serum ferritin in diabetic rats were decreased significantly by verapamil treatment. Moreover, serum, myocardial, and urine iron were elevated remarkably along with a decrease of hepatic iron in diabetic rats. After verapamil administration, serum and myocardial iron in diabetic rats were reduced significantly but urine and hepatic iron were increased. Furthermore, confocal microscopy demonstrated that intracellular-free iron concentration was elevated dramatically in cardiomyocytes of diabetic rats, which was markedly attenuated after verapamil treatment. In summary, our data demonstrated that verapamil prevented myocardial iron overload by inhibiting intracellular iron accumulation in diabetic cardiomyocytes.




Similar content being viewed by others
References
Castoldi M, Vujic Spasic M, Altamura S, Elmén J, Lindow M, Kiss J, Stolte J, Sparla R, D’Alessandro LA, Klingmüller U, Fleming RE, Longerich T, Gröne HJ, Benes V, Kauppinen S, Hentze MW, Muckenthaler MU (2011) The liver-specific microRNA miR-122 controls systemic iron homeostasis in mice. J Clin Invest 121:1386–1396
Cekic O, Bardak Y (1998) Lenticular calcium, magnesium, and iron levels in diabetic rats and verapamil effect. Ophthalmic Res 30:107–112
Cooksey RC, Jones D, Gabrielsen S, Huang J, Simcox JA, Luo B, Soesanto Y, Rienhoff H, Abel ED, McClain DA (2010) Dietary iron restriction or iron chelation protects from diabetes and loss of beta-cell function in the obese (ob/ob lep−/−) mouse. Am J Physiol Endocrinol Metab 298:E1236–E1243
Das SK, Oudit GY (2012) Voltage-gated Ca2+ channels as key mediators of iron-transport and iron-overload cardiomyopathy: l-type vs. T-type Ca2+ channels. Eur J Haematol 88:476–477
Day SM, Duquaine D, Mundada LV, Menon RG, Khan BV, Rajagopalan S, Fay WP (2003) Chronic iron administration increases vascular oxidative stress and accelerates arterial thrombosis. Circulation 107:2601–2606
Huang J, Jones D, Luo B, Sanderson M, Soto J, Abel ED, Cooksey RC, McClain DA (2011) Iron overload and diabetes risk: a shift from glucose to fatty acid oxidation and increased hepatic glucose production in a mouse model of hereditary hemochromatosis. Diabetes 60:80–87
Kell DB (2009) Iron behaving badly: inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases. BMC Med Genomics 2:2
Kohgo Y, Ikuta K, Ohtake T, Torimoto Y, Kato J (2008) Body iron metabolism and pathophysiology of iron overload. Int J Hematol 88:7–15
Kumfu S, Chattipakorn S, Srichairatanakool S, Settakorn J, Fucharoen S, Chattipakorn N (2011) T-type calcium channel as a portal of iron uptake into cardiomyocytes of beta-thalassemic mice. Eur J Haematol 86:156–166
Kumfu S, Chattipakorn S, Chinda K, Fucharoen S, Chattipakorn N (2012) T-type calcium channel blockade improves survival and cardiovascular function in thalassemic mice. Eur J Haematol 88:535–548
Lima PM, Neves Rde C, dos Santos FA, Pérez CA, da Silva MO, Arruda MA, de Castro GR, Padilha PM (2010) Analytical approach to the metallomic of Nile tilapia (Oreochromis niloticus) liver tissue by SRXRF and FAAS after 2D-PAGE separation: preliminary results. Talanta 82:1052–1056
Mladenka P, Simůnek T, Hübl M, Hrdina R (2006) The role of reactive oxygen and nitrogen species in cellular iron metabolism. Free Radic Res 40:263–272
Oudit GY, Sun H, Trivieri MG, Koch SE, Dawood F, Ackerley C, Yazdanpanah M, Wilson GJ, Schwartz A, Liu PP, Backx PH (2003) l-type Ca2+ channels provide a major pathway for iron entry into cardiomyocytes in iron-overload cardiomyopathy. Nat Med 9:1187–1194
Oudit GY, Trivieri MG, Khaper N, Liu PP, Backx PH (2006) Role of l-type Ca2+ channels in iron transport and iron-overload cardiomyopathy. J Mol Med (Berl) 84:349–364
Papanikolaou G, Pantopoulos K (2005) Iron metabolism and toxicity. Toxicol Appl Pharmacol 202:199–211
Ramos E, Ruchala P, Goodnough JB, Kautz L, Preza GC, Nemeth E, Ganz T (2012) Minihepcidins prevent iron overload in a hepcidin-deficient mouse model of severe hemochromatosis. Blood 120:3829–3836
Ray PD, Huang BW, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24:981–990
Ryu DY, Lee SJ, Park DW, Choi BS, Klaassen CD, Park JD (2004) Dietary iron regulates intestinal cadmium absorption through iron transporters in rats. Toxicol Lett 152:19–25
Stadler N, Heeneman S, Vöö S, Stanley N, Giles GI, Gang BP, Croft KD, Mori TA, Vacata V, Daemen MJ, Waltenberger J, Davies MJ (2012) Reduced metal ion concentrations in atherosclerotic plaques from subjects with type 2 diabetes mellitus. Atherosclerosis 222:512–518
Sun L, Ai J, Wang N, Zhang R, Li J, Zhang T, Wu W, Hang P, Lu Y, Yang B (2010) Cerebral ischemia elicits aberration in myocardium contractile function and intracellular calcium handling. Cell Physiol Biochem 26:421–430
Tajima S, Ikeda Y, Sawada K, Yamano N, Horinouchi Y, Kihira Y, Ishizawa K, Izawa-Ishizawa Y, Kawazoe K, Tomita S, Minakuchi K, Tsuchiya K, Tamaki T (2012) Iron reduction by deferoxamine leads to amelioration of adiposity via the regulation of oxidative stress and inflammation in obese and type 2 diabetes KKAy mice. Am J Physiol Endocrinol Metab 302:E77–E86
Tsushima RG, Wickenden AD, Bouchard RA, Oudit GY, Liu PP, Backx PH (1999) Modulation of iron uptake in heart by l-type Ca2+ channel modifiers: possible implications in iron overload. Circ Res 84:1302–1309
Umbreit J (2005) Iron deficiency: a concise review. Am J Hematol 78:225–31
Veuthey T, D'Anna MC, Roque ME (2008) Role of the kidney in iron homeostasis: renal expression of prohepcidin, ferroportin, and DMT1 in anemic mice. Am J Physiol Renal Physiol 295:F1213–F1221
Wang X, Miller DS, Zheng W (2008) Intracellular localization and subsequent redistribution of metal transporters in a rat choroid plexus model following exposure to manganese or iron. Toxicol Appl Pharmacol 230:167–174
Ward DT, Hamilton K, Burnand R, Smith CP, Tomlinson DR, Riccardi D (2005) Altered expression of iron transport proteins in streptozotocin-induced diabetic rat kidney. Biochim Biophys Acta 1740:79–84
Yang L, Zhang B, Yin L, Cai B, Shan H, Zhang L, Lu Y, Bi Z (2011) Tanshinone IIA prevented brain iron dyshomeostasis in cerebral ischemic rats. Cell Physiol Biochem 27:23–30
Zhao N, Sun Z, Mao Y, Hang P, Jiang X, Sun L, Zhao J, Du Z (2010) Myocardial iron metabolism in the regulation of cardiovascular diseases in rats. Cell Physiol Biochem 25:587–594
Acknowledgments
This work was supported by the research grant (no. 1154z1008) from Education Department of Heilongjiang Province of China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hong-li Shan and Yan Wang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
PDF 91 kb
Rights and permissions
About this article
Cite this article
Shan, Hl., Wang, Y., Wu, Jw. et al. Verapamil reverses cardiac iron overload in streptozocin-induced diabetic rats. Naunyn-Schmiedeberg's Arch Pharmacol 386, 645–650 (2013). https://doi.org/10.1007/s00210-013-0863-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-013-0863-2