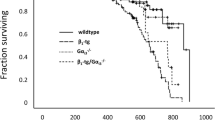
Abstract
Muscarinic receptors (MR) are main cardioinhibitory receptors. We investigated the changes in gene expression, receptor number, echocardiography, muscarinic/adrenergic agonist/antagonist changes in heart rate (HR) and HR biorhythm in M2 KO mice (mice lacking the main cardioinhibitory receptors) in the left ventricle (LV) and right ventricle (RV). We hypothesize that the disruption of M2 MR, key players in parasympathetic bradycardia, would change the number of receptors with antagonistic effects on the heart (β1- and β2-adrenoceptors, BAR), while the function of the heart would be changed only marginally. We have found changes in LV, but not in RV: decrease in M3 MR, β1- and β2-adrenoceptor gene expressions that were accompanied by a decrease in MR and BAR receptor binding. No changes were found both in LV systolic and diastolic function as assessed by echocardiography (e.g., similar LV end-systolic and end-diastolic diameter, fractional shortening, mitral flow characteristics, and maximal velocity in LV outflow tract). We have found only marginal changes in specific HR biorhythm parameters. The effects of isoprenaline and propranolol on HR were similar in WT and KO (but with lesser extent). Atropine was not able to increase HR in KO animals. Carbachol decreased the HR in WT but increased HR in KO, suggesting the presence of cardiostimulatory MR. Therefore, we can conclude that although the main cardioinhibitory receptors are not present in the heart, the function is not much affected. As possible mechanisms of almost normal cardiac function, the decreases of both β1- and β2-adrenoceptor gene expression and receptor binding should be considered.





Similar content being viewed by others
References
Arraj M, Lemmer B (2006) Circadian rhythms in heart rate, motility, and body temperature of wild-type C57 and eNOS knock out mice under light–dark, free run, and after time zone transition. Chronobiol Int 23:795–812
Benes J, Novakova M, Rotkova J, Farar V, Kvetnansky R, Riljak V, Myslivecek J (2012) Beta3 adrenoceptors substitute the role of M2 muscarinic receptor in coping with cold stress in the heart: evidence from M2KO mice. Cell Mol Neurobiol 32:589–869
Boknik P, Grote-Wessels S, Barteska G, Jiang M, Müller FU, Schmitz W, Neumann J, Birnbaumer L (2009) Genetic disruption of G proteins, Gi2α or Goα, does not abolish inotropic and chronotropic effects of stimulating muscarinic cholinoceptors in atrium. Br J Pharmacol 158:1557–1564
Brodde OE (2008) Beta-1 and beta-2 adrenoceptor polymorphisms: functional importance, impact on cardiovascular diseases and drug responses. Pharmacol Ther 117:1–29
Brodde OE, Leineweber K (2004) Autonomic receptor systems in the failing and aging human heart: similarities and differences. Eur J Pharmacol 500:167–176
Burnstock G (2009) Autonomic neurotransmission: 60 years since Sir Henry Dale. Annu Rev Pharmacol Toxicol 49:1–30
Bymaster FP, Carter PA, Zhang L, Falcone JF, Stengel PW, Cohen ML, Shannon HE, Gomeza J, Wess J, Felder CC (2001) Investigations into the physiological role of muscarinic M2 and M4 muscarinic and M4 receptor subtypes using receptor knockout mice. Life Sci 68:2473–2479
Chruscinski AJ, Rohrer DK, Schauble E, Desai KH, Bernstein D, Kobilka BK (1999) Targeted disruption of the beta 2 adrenergic receptor gene. J Biol Chem 274:16694–16700
Ecker PM, Lin CC, Powers J, Kobilka BK, Dubin AM, Bernstein D (2006) Effect of targeted deletions of beta1- and beta2-adrenergic-receptor subtypes on heart rate variability. Am J Physiol Heart Circ Physiol 290:H192–H199
Fasano C, Niel J-P (2009) The mammalian sympathetic prevertebral ganglia: models for the study of neuronal networks and basic neuronal properties. Auton Neurosci 150:8–20
Fisher JT, Vincent SG, Gomeza J, Yamada M, Wess J (2004) Loss of vagally mediated bradycardia and bronchoconstriction in mice lacking M2 or M3 muscarinic acetylcholine receptors. FASEB J 18:711–713
Garofolo MC, Seidler FJ, Auman JT, Slotkin TA (2002) Beta-adrenergic modulation of muscarinic cholinergic receptor expression and function in developing heart. Am J Physiol Regul Integr Comp Physiol 282:R1356–R1363
Giménez LED, Hernández CCQ, Mattos EC, Brandão IT, Olivieri B, Campelo RP, Araújo-Jorge T, Silva CL, Campos de Carvalho AC, Kurtenbach E (2005) DNA immunizations with M2 muscarinic and β1 adrenergic receptor coding plasmids impair cardiac function in mice. J Mol Cell Cardiol 38:703–714
Gomeza J, Shannon H, Kostenis E, Felder C, Zhang L, Brodkin J, Grinberg A, Sheng H, Wess J (1999) Pronounced pharmacologic deficits in M2 muscarinic acetylcholine receptor knockout mice. Proc Natl Acad Sci USA 96:1692–1697
Gomeza J, Zhang L, Kostenis E, Felder CC, Bymaster FP, Brodkin J, Shannon H, Xia B, Duttaroy A, Deng CX, Wess J (2001) Generation and pharmacological analysis of M2 and M4 muscarinic receptor knockout mice. Life Sci 68:2457–2466
Göttle M, Geduhn J, König B, Gille A, Höcherl K, Seifert R (2009) Characterization of mouse heart adenylyl cyclase. J Pharmacol Exp Ther 329:1156–1165
Grouzmann E, Cavadas C, Grand D, Moratel M, Aubert J-F, Brunner H, Mazzolai L (2003) Blood sampling methodology is crucial for precise measurement of plasma catecholamines concentrations in mice. Pflügers Arch Eur J Physiol 447:254–258
Hoffert JD, Chou CL, Fenton RA, Knepper MA (2005) Calmodulin is required for vasopressin-stimulated increase in cyclic AMP production in inner medullary collecting duct. J Biol Chem 280:13624–13630
Imeri L, Bianchi S, Angeli P, Mancia M (1996) Muscarinic receptor subtypes in the medial preoptic area and sleep–wake cycles. Neuroreport 7:417–420
Ito Y, Oyunzul L, Seki M, Fujino T, Matsui M, Yamada S (2009) Quantitative analysis of the loss of muscarinic receptors in various peripheral tissues in M1–M5 receptor single knockout mice. Br J Pharmacol 156:1147–1153
Kiriazis H, Wang K, Xu Q, Gao XM, Ming Z, Su Y, Moore XL, Lambert G, Gibbs ME, Dart AM, Du XJ (2008) Knockout of β1- and β2-adrenoceptors attenuates pressure overload-induced cardiac hypertrophy and fibrosis. Br J Pharmacol 153:684–692
Kitazawa T, Asakawa K, Nakamura T, Teraoka H, Unno T, S-i K, Yamada M, Wess J (2009) M3 muscarinic receptors mediate positive inotropic responses in mouse atria: a study with muscarinic receptor knockout mice. J Pharmacol Exp Ther 330:487–493
LaCroix C, Freeling J, Giles A, Wess J, Li Y-F (2008) Deficiency of M2 muscarinic acetylcholine receptors increases susceptibility of ventricular function to chronic adrenergic stress. Am J Physiol Heart Circ Physiol 294:H810–H820
Lee NH, Fraser CM (1993) Cross-talk between m1 muscarinic acetylcholine and beta 2-adrenergic receptors. cAMP and the third intracellular loop of m1 muscarinic receptors confer heterologous regulation. J Biol Chem 268:7949–7957
Lee S, Grafweg S, Schneider T, Jimenez M, Giacobino JP, Ghanem A, Tiemann K, Bloch W, Muller-Ehmsen J, Schwinger RH, Brixius K (2010) Total beta-adrenoceptor deficiency results in cardiac hypotrophy and negative inotropy. Physiological Research/Academia Scientiarum Bohemoslovaca 59:679–689
Myslivecek J, Trojan S, Tucek S (1996) Biphasic changes in the density of muscarinic and beta-adrenergic receptors in cardiac atria of rats treated with diisopropylfluorophosphate. Life Sci 58:2423–2430
Myslivecek J, Lisa V, Trojan S, Tucek S (1998) Heterologous regulation of muscarinic and beta-adrenergic receptors in rat cardiomyocytes in culture. Life Sci 63:1169–1182
Myslivecek J, Duysen EG, Lockridge O (2007) Adaptation to excess acetylcholine by downregulation of adrenoceptors and muscarinic receptors in lungs of acetylcholinesterase knockout mice. Naunyn Schmiedebergs Arch Pharmacol 376:83–92
Myslivecek J, Klein M, Novakova M, Ricny J (2008) The detection of the non-M2 muscarinic receptor subtype in the rat heart atria and ventricles. Naunyn Schmiedebergs Arch Pharmacol 378:103–116
Ondicova K, Mravec B (2010) Multilevel interactions between the sympathetic and parasympathetic nervous systems: a minireview. Endocr Regul 44:69–75
Paraschos A, Karliner JS (1994) Receptor crosstalk: effects of prolonged carbachol exposure on beta 1-adrenoceptors and adenylyl cyclase activity in neonatal rat ventricular myocytes. Naunyn Schmiedebergs Arch Pharmacol 350:267–276
Racké K, Juergens UR, Matthiesen S (2006) Control by cholinergic mechanisms. Eur J Pharmacol 533:57–68
Rhodes ME, Billings TE, Czambel RK, Rubin RT (2005) Pituitary–adrenal responses to cholinergic stimulation and acute mild stress are differentially elevated in male and female M2 muscarinic receptor knockout mice. J Neuroendocrinol 17:817–826
Rohrer DK, Desai KH, Jasper JR, Stevens ME, Regula DP Jr, Barsh GS, Bernstein D, Kobilka BK (1996) Targeted disruption of the mouse beta1-adrenergic receptor gene: developmental and cardiovascular effects. Proc Natl Acad Sci USA 93:7375–7380
Stavrakis S, Kem DC, Patterson E, Lozano P, Huang S, Szabo B, Cunningham MW, Lazzara R, Yu X (2011) Opposing cardiac effects of autoantibody activation of beta-adrenergic and M2 muscarinic receptors in cardiac-related diseases. Int J Cardiol 148:331–336
Stengel PW, Gomeza J, Wess J, Cohen ML (2000) M(2) and M(4) receptor knockout mice: muscarinic receptor function in cardiac and smooth muscle in vitro. J Pharmacol Exp Ther 292:877–885
Swoap SJ, Li C, Wess J, Parsons AD, Williams TD, Overton JM (2008) Vagal tone dominates autonomic control of mouse heart rate at thermoneutrality. Am J Physiol Heart Circ Physiol 294:H1581–H1588
Tanaka N, Dalton N, Mao L, Rockman HA, Peterson KL, Gottshall KR, Hunter JJ, Chien KR, Ross J (1996) Transthoracic echocardiography in models of cardiac disease in the mouse. Circulation 94:1109–1117
Trendelenburg A-U, Meyer A, Wess J, Starke K (2005) Distinct mixtures of muscarinic receptor subtypes mediate inhibition of noradrenaline release in different mouse peripheral tissues, as studied with receptor knockout mice. Br J Pharmacol 145:1153–1159
Wang Z, Shi H, Wang H (2004) Functional M3 muscarinic acetylcholine receptors in mammalian hearts. Br J Pharmacol 142:395–408
Wang H, Lu Y, Wang Z (2007) Function of cardiac M3 receptors. Auton Autacoid Pharmacol 27:1–11
Werry TD, Wilkinson GF, Willars GB (2003) Mechanisms of cross-talk between G-protein-coupled receptors resulting in enhanced release of intracellular Ca2+. Biochem J 374:281–296
Wess J, Duttaroy A, Zhang W, Gomeza J, Cui Y, Miyakawa T, Bymaster FP, McKinzie L, Felder CC, Lamping KG, Faraci FM, Deng C, Yamada M (2003) M1–M5 muscarinic receptor knockout mice as novel tools to study the physiological roles of the muscarinic cholinergic system. Receptors Channels 9:279–290
Willmy-Matthes P, Leineweber K, Wangemann T, Silber RE, Brodde OE (2003) Existence of functional M3-muscarinic receptors in the human heart. Naunyn Schmiedebergs Arch Pharmacol 368:316–319
Yang T, Levy M (1984) The phase-dependency of the cardiac chronotropic responses to vagal stimulation as a factor in sympathetic-vagal interactions. Circ Res 54:703–710
Zhou H, Meyer A, Starke K, Gomeza J, Wess J, Trendelenburg AU (2002) Heterogeneity of release-inhibiting muscarinic autoreceptors in heart atria and urinary bladder: a study with M(2)- and M(4)-receptor-deficient mice. Naunyn Schmiedebergs Arch Pharmacol 365:112–122
Acknowledgments
This work was supported by Grant GACR309/09/0406. We thank Dr. Jürgen Wess (NIH, Bethesda, MD, USA) for providing the muscarinic receptor mutant mice, Prof. Michal Zeman (Faculty of Natural Sciences, Comenius University, Bratislava, Slovakia) for advice with biorhythm analysis, and Prof. Ludek Cervenka (Institute for Clinical and Experimental Medicine, Prague, Czech Republic) for the great help with echocardiography study. The technical assistance of Petra Svatosova is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jan Benes and Eva Varejkova participated equally on this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Saturation binding to muscarinic (left) and -adrenergic (right) receptors in WT (above) and KO (below) animals. Abscissa: concentration of free radioligand [nmol.l-1].Ordinate: Bmax [fmol.mg prot.-1]. Insert: Scatchard plot of binding. MR Muscarinic receptors; BAR, β-adrenoceptors. (JPEG 75 kb)
Figure S2
Competition binding curves for CGP 20712A (1-AR antagonist) and ICI118.551 (2-AR antagonist) with 3H-CGP12177. Average data from 4 independent experiments. Abscissa: log of antagonist concentration.Ordinate: Bound [% of binding in the absence of antagonist]. (JPEG 49 kb)
Figure S3
The representative echocardiograms in WT and KO mice. Left: WT mice, right: KO mice. Top: Two-dimensional echocardiogram: end diastole, M-mode echocardiogram from the same mouse in the parasternal short-axis view. Middle: left ventricle outflow tract (two-dimensional echocardiogram, pulsed wave Doppler tracing). Bottom: mitral view (two-dimensional echocardiogram, pulsed wave Doppler tracing). (JPEG 129 kb)
Figure S4
The representative figure of spectral density vs. period and spectral density vs. frequency in WT and KO animals. No difference was observed between KO and WT mice. (JPEG 58 kb)
Figure S5
The gene expression of TH, DBH and PNMT in WT and KO animals. No change in gene expression was observed. (JPEG 133 kb)
Figure S6
The changes in activity in KO mice. Top: average activity. Middle: the activity in the light and dark phase (day or night). Bottom: comparison of biorhythm in WT and KO animals. ***p<0.001 different from WT. ###p<0.001 different from KO day, one-way ANOVA (p< 0.0001, F=15.83, df=93). (JPEG 90 kb)
Figure S7
The changes in temperature in KO mice. Top: average temperature. Middle: the temperature in the light and dark phase (day or night). Bottom: comparison of biorhythm in WT and KO animals. ****p<0.0001 different from WT, ***p<0.001 different from WT day, ###p<0.001 different from KO day, +++p<0.001 different from WT night, one-way ANOVA (p< 0.0001, F=36.18, df=93). (JPEG 79 kb)
Figure S8
The actograms of representative WT and KO mice showing increased values of respective parameters (black rectangles). Abscissa: time starting at 6:00 AM and showing two consecutive days in one row. Ordinate: The number of row representing two days. From top to bellow: WT activity, KO activity, WT HR, KO HR, WT temperature, KO temperature. (JPEG 176 kb)
ESM 9
(DOCX 13 kb)
ESM 10
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Benes, J., Varejkova, E., Farar, V. et al. Decrease in heart adrenoceptor gene expression and receptor number as compensatory tool for preserved heart function and biological rhythm in M2 KO animals. Naunyn-Schmiedeberg's Arch Pharmacol 385, 1161–1173 (2012). https://doi.org/10.1007/s00210-012-0800-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-012-0800-9