Abstract
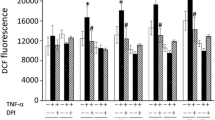
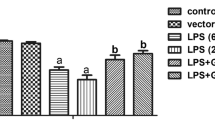
Endotoxin (LPS)-induced cardiac failure is associated with an up-regulation of RGS16 protein expression and repression of phospholipase C activity in vivo. Since the release of pro-inflammatory cytokines plays an important role in mediating LPS-induced myocardial dysfunction, we examined the effect of recombinant cytokines on the expression of RGS16 protein in neonatal cardiac myocytes. Myocytes in culture were treated with 50 ng/ml recombinant tumor necrosis factor α (TNFα), 2 ng/ml interleukin 1β (IL-1β), interleukin 6 (IL-6), interferon γ (IFNγ) or diluent (NaCl) for 24 h. Before stimulation with LPS (4 µg/ml for 24 h) cells were treated with 200 ng/ml interleukin 1-receptor antagonist (IL-1ra), 500 ng/ml soluble TNF receptor (sTNFr), or NaCl for 1 h. Isolated membrane proteins were used for Western blot analysis. Cell-associated and secreted IL-1β and TNFα protein content were determined in myocyte protein homogenates and cell culture supernatants by ELISA immunoblotting 3, 6, 24, 48 and 72 h after treatment with LPS. IL-1β (1.75-fold) and TNFα (1.62-fold) but not IL-6 and IFNγ induced RGS16 protein expression. LPS stimulated intracellular IL-1β expression within 6 h (847.1±172.9 pg/3×106 cells) followed by an increase in extracellular secretion up to 70.8±8.1 pg/3×106 cells after 48 h. In contrast, intracellular protein concentrations of TNFα were almost not detectable (0.03±0.01 pg/3×106 cells), but extracellular secretion was induced by LPS with a maximum at 6 h (653.9±36.3 pg/3×106 cells). The LPS-induced increase in RGS16 (1.6-fold) was blunted by IL-1ra but not by TNFα scavenging. Interestingly, both, the IL-1β- and TNFα-effect could be blocked by IL-1ra, indicating that also the TNFα-induced RGS16 expression is mediated by IL-1. We therefore conclude that LPS induces RGS16 protein expression by activation of the cytokine IL-1β in cardiac myocytes. Our data substantiate the role of IL-1β as an important mediator in LPS-induced cardiac failure.





Similar content being viewed by others
References
Baumgarten G, Knuefermann P, Nozaki N, Sivasubramanian N, Mann D-L, Vallejo J-G (2001) In vivo expression of proinflammatory mediators in the adult heart after endotoxin administration: the role of toll-like receptor-4. J Infect Dis 183:1617–1624
Böhm M, Kirchmayr R, Gierschik P, Erdmann E (1995) Increase of myocardial inhibitory G-proteins in catecholamine-refractory septic shock or in septic multiorgan failure. Am J Med 98:183–186
Cannon JG, Tompkins RG, Gelfand JA, Michie HR, Stanford GG, van de Meer JWM, Endres S, Lonnemann G, Corsetti J, Chernow B, Wilmore DW, Wolff SM, Burke JF, Dinarello CA (1990) Circulating interleukin-1 and tumor necrosis factor in septic shock and experimental endotoxin fever. J Infect Dis 161:79–84
Comstock K, Krown K, Page M, Martin D, Ho P, Pedraza M, Castro EN, Nakajima N, Glembotski CC, Quintana PJ, Sabbadini RA (1998) LPS-induced TNF-α release from and apoptosis in rat cardiomyocytes: obligatory role for CD14 in mediating the LPS response. J Mol Cell Cardiol 30:2761–2775
Danis VA, Franic GM, Rathjen DA, Brooks PM (1991) Effects of granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-2, interferon-γ (IFN-γ), tumor necrosis factor-α (TNF-α) and IL-6 on the production of immunoreactive IL-1 and TNF-α by human monocytes. Clin Exp Immunol 85:143–150
Detmer K, Wang Z, Warejcka D, Leeper-Woodford SK, Newman WH (2001) Endotoxin stimulated cytokine production in rat vascular smooth muscle cells. Am J Physiol 281:H661–H668
Dinarello CA (1984) Interleukin-1. Rev Infect Dis 6:51–95
Feldman AM, Cates AE, Veazey WB, Hershberger RE, Bristow MR, Baughman KL, Baumgartner WA, VanDop C (1988) Increase of the 40,000-mol wt pertussis toxin substrate (G-protein) in the failing human heart. J Clin Invest 82:189–197
Feldman MD, Copelas L, Gwathmey JK, Phillips P, Warren SE, Schoen FJ, Grossman W, Morgan JP (1987) Deficient production of cyclic AMP: pharmacologic evidence of an important cause of contractile dysfunction in patients with end-stage heart failure. Circulation 75:331–339
Finkel MS, Oddis CV, Jacob TD, Watkins SC, Hattler BG, Simmons RL (1992) Negative inotropic effects of cytokines on the heart mediated by nitric oxide. Science 257:387–389
Frantz S, Kobzik L, Kim YD, Fukazawa R, Medzhitov R, Lee RT, Kelly RA (1999) Toll like receptor 4 (TLR4) expression in cardiac myocytes in normal and failing myocardium. J Clin Invest 104:271–280
Girardin E, Brau GE, Dayer JM, Roux-Lombard P, Group TJS, Lambert P (1988) Tumor necrosis factor and interleukin-1 in the serum of children with severe infectious purpura. N Engl J Med 319:397–400
Kinugawa K, Takahashi T, Kohmoto O, Yao A, Aoyagi T, Momomura S, Hirata Y, Serizawa T (1994) Nitric oxide-mediated effects of interleukin-6 on [Ca2+] and cell contraction in cultured chick ventricular myocytes. Circ Res 75:285–295
Kraut E, Chen S, Hubbard N, Erickson K, Wisner D (1999) Tumor necrosis factor depresses myocardial contractility in endotoxemic swine. J Trauma 46:900–906
Kumar A, Thota V, Dee L, Olson J, Uretz E, Parrillo JE (1996) Tumor necrosis factor α and interleukin 1β are responsible for in vitro myocardial cell depression induced by human septic shock serum. J Exp Med 183:949–958
Kwon J, Chung I-Y, Benveniste EN (1993) Cloning and sequence analysis of the rat tumor necrosis factor-encoding genes. Gene 132:227–236
Libby P, Ordovas JM, Auger KR, Robbins AH, Birinyi LK, Dinarello CA (1986) Endotoxin and tumor necrosis factor induce interleukin-1 gene expression in adult human vascular endothelial cells. Am J Pathol 124:179–185
Mittmann C, Schüler C, Chung CH, Höppner G, Nose M, Kehrl JH, Weiland T (2001) Evidence for a short form of RGS3 preferentially expressed in the human heart. Naunyn-Schmiedebergs Arch Pharmacol 363:456–463
Mittmann C, Chung C-H, Höppner G, Michalek C, Nose M, Schüler C, Schuh A, Eschenhagen T, Weil J, Pieske B, Hirt S, Wieland T (2002) Expression of ten RGS proteins in human myocardium: functional characterization of an upregulation of RGS4 in heart failure. Cardiovasc Res 55:778–786
Müller-Werdan U, Schumann H, Loppnow H, Fuchs R, Darmer D, Stadler J, Holtz J, Werdan K (1998) Endotoxin and tumor necrosis factor α exert a similar proinflammatory effect in neonatal rat cardiomyocytes, but have different cardiodepressant profiles. J Mol Cell Cardiol 30:1027–1036
Parker JL, Adams RH (1985) Development of myocardial dysfunction in endotoxin shock. Am J Physiol 248:H818–H826
Patten M, Krämer E, Bünemann J, Wenck C, Thoenes M, Wieland T, Long C (2001) Endotoxin and cytokines alter contractile protein expression in cardiac myocytes in vivo. Pflügers Arch Eur J Physiol 442:920–927
Patten M, Bünemann J, Thoma B, Krämer E, Thoenes M, Mittmann C, Wieland T (2002) Endotoxin induces desensitization of cardiac endothelin-1 receptor signaling by increased expression of RGS4 and RGS16. Cardiovasc Res 53:159–164
Reithmann C, Werdan K (1994) Tumor necrosis factor α decreases inositol phosphate formation and phosphatidylinositol-bisphosphate (PIP2) synthesis in rat cardiomyocytes. Naunyn-Schmiedebergs Arch Pharmacol 349:175–182
Reithmann C, Gierschik P, Werdan P, Jakobs KH (1991) Tumor necrosis factor α up-regulates Giα and Gβ proteins and adenylyl cyclase responsiveness in rat cardiomyocytes. Eur J Pharmacol 206:53–60
Stein B, Frank P, Schmitz W, Scholz H, Thoenes M (1996) Endotoxin and cytokines induces direct cardiodepressive effects in mammalian cardiomyocytes via induction of nitric oxide synthase. J Mol Cell Cardiol 28:1631–1639
Sun X, Delbridge MD, Dusting GJ (1998) Cardiodepressant effects of interferon-γ and endotoxin reversed by inhibition of NO synthase in rat myocardium. J Mol Cell Cardiol 30:989–997
Tamirisa P, Blumer KJ, Muslin AJ (1999) RGS4 inhibits G-protein signaling in cardiomyocytes. Circulation 99:441–447
Ulich TR, Guo KZ, Irwin B, Remick DG, Davatelis GN (1990) Endotoxin-induced cytokine gene expression in vivo. Regulation of tumor necrosis factor and interleukin 1 α/β expression and suppression. Am J Pathol 137:1173–1185
Wakabayashi G, Gelfand JA, Burke JF, Thompson RC, Dinarello CA (1991) A specific receptor antagonist for interleukin 1 prevents Escherichia coli-induced shock in rabbits. FASEB J 5:338–343
Wieland T, Chen CK (1999) Regulators of G-protein signalling: a novel protein family involved in timely deactivation and desensitization of signalling via heterotrimeric G proteins. Naunyn-Schmiedebergs Arch Pharmacol 360:14–26
Wieland T, Mittmann C (2003) Regulators of G protein signalling: multifunctional proteins with impact on signalling in the cardiovascular system. Pharmacol Ther 97:95–115
Zeuke S, Ulmer AJ, Kusumoto S, Katus HA, Heine H (2002) TLR4-mediated inflammatory activation of human coronary artery endothelial cells by LPS. Cardiovasc Res 56:124–136
Zimmermann WH, Fink C, Kralisch D, Remmers U, Weil J, Eschenhagen T (2000) Three-dimensional engineered heart tissue from neonatal rat cardiac myocytes. Biotechnol Bioeng 68:106–114
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patten, M., Stübe, S., Thoma, B. et al. Interleukin-1β mediates endotoxin- and tumor necrosis factor α-induced RGS16 protein expression in cultured cardiac myocytes. Naunyn-Schmiedeberg's Arch Pharmacol 368, 360–365 (2003). https://doi.org/10.1007/s00210-003-0798-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-003-0798-0