Abstract.
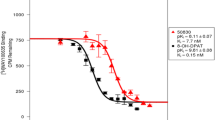
LE300 (7-methyl-6,7,8,9,14,15-hexahydro-5H-benz[d]indolo[2,3-g]azecine), a previously reported subnanomolar antagonist at rat striatal dopamine D1 receptors, and three of its azecine-N-substituted congeners combining structural elements of serotonin and dopamine were comprehensively characterised (binding and function) at recombinant human dopamine receptors. Radioligand competition experiments at D1 and D2L receptors were performed by using [3H]SCH23390 and [3H]spiperone, respectively. Functional assays included measurements of cAMP, intracellular [Ca2+], and [35S]GTPγS-binding. LE300 was the most potent compound with a 10- to 20-fold selectivity for D1 over D2L receptors as measured in equilibrium binding experiments [competition radioligand binding: K i(D1)=1.9 nM, K i(D2L)=44.7 nM; [35S]GTPγS-binding: K i(D1)=1.8 nM, K i(D2L)=21.5 nM]. In functional (non-equilibrium) experiments, LE300 did not reveal a D1 over D2L selectivity but retained nanomolar K i values at human dopamine receptors (measurement of cAMP: K i(D1)=25.9 nM, K i(D2L)=5.2 nM; measurement of intracellular [Ca2+]: K i(D1)=60.4 nM, K i(D2L)=19.0 nM). LE300 is currently under investigation for usefulness as positrone emission tomography ligand. In conclusion, LE300 is a novel type of a nanomolar dopamine receptor antagonist combining structural core elements of dopamine and serotonin, and may become useful as positrone emission tomography ligand.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Kassack, M.U., Höfgen, B., Decker, M. et al. Pharmacological characterization of the benz[d]indolo[2,3-g]azecine LE300, a novel type of a nanomolar dopamine receptor antagonist. Naunyn-Schmiedeberg's Arch Pharmacol 366, 543–550 (2002). https://doi.org/10.1007/s00210-002-0641-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00210-002-0641-z