Abstract
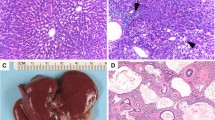
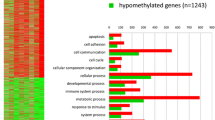
Epigenetic modifications, such as DNA methylation, play an important role in carcinogenesis. In a recent NTP study, chronic exposure of B6C3F1/N mice to Ginkgo biloba extract (GBE) resulted in a high incidence of hepatocellular carcinomas (HCC). Genome-wide promoter methylation profiling on GBE-exposed HCC (2000 mg/kg group), spontaneous HCC (vehicle-control group), and age-matched vehicle control liver was performed to identify differentially methylated genes in GBE-exposed HCC and spontaneous HCC. DNA methylation alterations were correlated to the corresponding global gene expression changes. Compared to control liver, 1296 gene promoters (719 hypermethylated, 577 hypomethylated) in GBE-exposed HCC and 738 (427 hypermethylated, 311 hypomethylated) gene promoters in spontaneous HCC were significantly differentially methylated, suggesting an impact of methylation on GBE-exposed HCC. Differential methylation of promoter regions in relevant cancer genes (cMyc, Spry2, Dusp5) and their corresponding differential gene expression was validated by quantitative pyrosequencing and qRT-PCR, respectively. In conclusion, we have identified differentially methylated promoter regions of relevant cancer genes altered in GBE-exposed HCC compared to spontaneous HCC. Further study of unique sets of differentially methylated genes in chemical-exposed mouse HCC could potentially be used to differentiate treatment-related tumors from spontaneous-tumors in cancer bioassays and provide additional understanding of the underlying epigenetic mechanisms of chemical carcinogenesis.







Similar content being viewed by others
Abbreviations
- GBE:
-
Ginkgo biloba extract
- HCC:
-
Hepatocellular carcinoma
- SPNT:
-
Spontaneous
- CNTL:
-
Control
- Myc:
-
Myelocytomatosis
- Spry2:
-
Sprouty RTK signaling antagonist 2
- Dusp5:
-
Dual specificity phosphatase 5
- DEGs:
-
Differentially expressed genes
- DMR:
-
Differentially methylated regions
- DMGs:
-
Differentially methylated genes
- H&E:
-
Hematoxylin and eosin
- FFPE:
-
Formalin-fixed paraffin embedded
- RMA:
-
Robust multiarray normalization
- ANOVA:
-
Analysis of variance
References
Ally A, Balasundaram M, Carlsen R, Chuah E (2017) Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell 169(7):1327 e23–1341 e23. https://doi.org/10.1016/j.cell.2017.05.046
Aravalli RN, Steer CJ, Cressman EN (2008) Molecular mechanisms of hepatocellular carcinoma. Hepatology 48(6):2047–2063. https://doi.org/10.1002/hep.22580
Bolstad BM, Irizarry RA, Astrand M, Speed TP (2003) A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19(2):185–193. https://doi.org/10.1093/bioinformatics/19.2.185
Calin GA, Croce CM (2006) MicroRNA signatures in human cancers. Nat Rev Cancer 6(11):857–866. https://doi.org/10.1038/nrc1997
Calvisi DF, Ladu S, Gorden A et al (2006) Ubiquitous activation of Ras and Jak/Stat pathways in human HCC. Gastroenterology 130(4):1117–1128. https://doi.org/10.1053/j.gastro.2006.01.006
Calvisi DF, Pinna F, Meloni F et al (2008) Dual-specificity phosphatase 1 ubiquitination in extracellular signal-regulated kinase-mediated control of growth in human hepatocellular carcinoma. Cancer Res 68(11):4192–4200. https://doi.org/10.1158/0008-5472.CAN-07-6157
Casper J, Zweig AS, Villarreal C et al (2018) The UCSC Genome Browser database: 2018 update. Nucleic Acids Res 46(D1):D762–D769. https://doi.org/10.1093/nar/gkx1020
Chaganty NR (1997) An alternative approach to the analysis of longitudinal data via generalized estimating equations. J Stat Plan Inference 63(1):39–54. https://doi.org/10.1016/S0378-3758(96)00203-0
Chan KL, Guan XY, Ng IO (2004) High-throughput tissue microarray analysis of c-myc activation in chronic liver diseases and hepatocellular carcinoma. Hum Pathol 35(11):1324–1331. https://doi.org/10.1016/j.humpath.2004.06.012
Chan PC, Xia Q, Fu PP (2007) Ginkgo biloba leave extract: biological, medicinal, and toxicological effects. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 25(3):211–244. https://doi.org/10.1080/10590500701569414
Cheng J, Wei D, Ji Y et al (2018) Integrative analysis of DNA methylation and gene expression reveals hepatocellular carcinoma-specific diagnostic biomarkers. Genome Med 10(1):42. https://doi.org/10.1186/s13073-018-0548-z
Cleveland WS, Devlin SJ (1988) Locally weighted regression: an approach to regression analysis by local fitting. J Am Stat Assoc 83(403):596–610. https://doi.org/10.1080/01621459.1988.10478639
Coleman WB (2003) Mechanisms of human hepatocarcinogenesis. Curr Mol Med 3(6):573–588
Costello JF, Fruhwald MC, Smiraglia DJ et al (2000) Aberrant CpG-island methylation has non-random and tumour-type-specific patterns. Nat Genet 24(2):132–138. https://doi.org/10.1038/72785
Cui M, Sun J, Hou J et al (2016) The suppressor of cytokine signaling 2 (SOCS2) inhibits tumor metastasis in hepatocellular carcinoma. Tumour Biol 37(10):13521–13531. https://doi.org/10.1007/s13277-016-5215-7
Dang CV, O’Donnell KA, Zeller KI, Nguyen T, Osthus RC, Li F (2006) The c-Myc target gene network. Semin Cancer Biol 16(4):253–264. https://doi.org/10.1016/j.semcancer.2006.07.014
DeFeudis FV, Papadopoulos V, Drieu K (2003) Ginkgo biloba extracts and cancer: a research area in its infancy. Fundam Clin Pharmacol 17(4):405–417
Ding X, Cheng X, Gong M, Chen X, Yin F, Lai K (2016) Hypermethylation and expression silencing of PDCD4 gene in hepatocellular carcinoma: a consort study. Medicine (Baltimore) 95(6):e2729. https://doi.org/10.1097/MD.0000000000002729
Doerfler W (1983) DNA methylation and gene activity. Annu Rev Biochem 52:93–124. https://doi.org/10.1146/annurev.bi.52.070183.000521
Dragani TA, Manenti G, Sacchi MR, Colombo BM, Della Porta G (1989) Major urinary protein as a negative tumor marker in mouse hepatocarcinogenesis. Mol Carcinog 2(6):355–360
Fang CH, Zhang GQ, Zhu XY, Gong JQ (2004) Distribution of oval cells and c-myc mRNA expression in mouse hepatocarcinogenesis. Hepatobiliary Pancreat Dis Int 3(3):433–439
Feinberg AP, Vogelstein B (1983) Hypomethylation distinguishes genes of some human cancers from their normal counterparts. Nature 301(5895):89–92
Fu Q, McKnight RA, Yu X, Callaway CW, Lane RH (2006) Growth retardation alters the epigenetic characteristics of hepatic dual specificity phosphatase 5. FASEB J 20(12):2127–2129. https://doi.org/10.1096/fj.06-6179fje
Hastie T, Tibshirani R (1990) Generalized additive models, 1st edn. Chapman and Hall, London
Hernandez-Vargas H, Lambert MP, Le Calvez-Kelm F et al (2010) Hepatocellular carcinoma displays distinct DNA methylation signatures with potential as clinical predictors. PLoS One 5(3):e9749. https://doi.org/10.1371/journal.pone.0009749
Hoenerhoff MJ, Pandiri AR, Snyder SA et al (2013) Hepatocellular carcinomas in B6C3F1 mice treated with Ginkgo biloba extract for two years differ from spontaneous liver tumors in cancer gene mutations and genomic pathways. Toxicol Pathol 41(6):826–841. https://doi.org/10.1177/0192623312467520
Hou C, Feng W, Wei S et al (2018) Bioinformatics analysis of key differentially expressed genes in nonalcoholic fatty liver disease mice models. Gene Expr 19(1):25–35. https://doi.org/10.3727/105221618X15341831737687
Hu TH, Huang CC, Lin PR et al (2003) Expression and prognostic role of tumor suppressor gene PTEN/MMAC1/TEP1 in hepatocellular carcinoma. Cancer 97(8):1929–1940. https://doi.org/10.1002/cncr.11266
Hui AM, Sakamoto M, Kanai Y et al (1996) Inactivation of p16INK4 in hepatocellular carcinoma. Hepatology 24(3):575–579. https://doi.org/10.1002/hep.510240319
Hwang YH, Choi JY, Kim S et al (2004) Over-expression of c-raf-1 proto-oncogene in liver cirrhosis and hepatocellular carcinoma. Hepatol Res 29(2):113–121. https://doi.org/10.1016/j.hepres.2004.02.009
Imbeaud S, Ladeiro Y, Zucman-Rossi J (2010) Identification of novel oncogenes and tumor suppressors in hepatocellular carcinoma. Semin Liver Dis 30(1):75–86. https://doi.org/10.1055/s-0030-1247134
Jones PA, Baylin SB (2007) The epigenomics of cancer. Cell 128(4):683–692. https://doi.org/10.1016/j.cell.2007.01.029
Jones PA, Martienssen R (2005) A blueprint for a human epigenome project: the AACR human epigenome workshop. Cancer Res 65(24):11241–11246. https://doi.org/10.1158/0008-5472.CAN-05-3865
Kang YH, Ji NY, Lee CI et al (2011) ESM-1 silencing decreased cell survival, migration, and invasion and modulated cell cycle progression in hepatocellular carcinoma. Amino Acids 40(3):1003–1013. https://doi.org/10.1007/s00726-010-0729-6
Katayama A, Nakatsuka A, Eguchi J et al (2015) Beneficial impact of Gpnmb and its significance as a biomarker in nonalcoholic steatohepatitis. Sci Rep 5:16920. https://doi.org/10.1038/srep16920
Kim Y, Sills RC, Houle CD (2005) Overview of the molecular biology of hepatocellular neoplasms and hepatoblastomas of the mouse liver. Toxicol Pathol 33(1):175–180. https://doi.org/10.1080/01926230590522130
Ladd-Acosta C (2015) Epigenetic signatures as biomarkers of exposure. Curr Environ Health Rep 2(2):117–125. https://doi.org/10.1007/s40572-015-0051-2
Lee SA, Ladu S, Evert M et al (2010) Synergistic role of Sprouty2 inactivation and c-Met up-regulation in mouse and human hepatocarcinogenesis. Hepatology 52(2):506–517. https://doi.org/10.1002/hep.23681
Lo TL, Fong CW, Yusoff P et al (2006) Sprouty and cancer: the first terms report. Cancer Lett 242(2):141–150. https://doi.org/10.1016/j.canlet.2005.12.032
Maronpot RR, Haseman JK, Boorman GA, Eustis SE, Rao GN, Huff JE (1987) Liver lesions in B6C3F1 mice: the National Toxicology Program, experience and position. Arch Toxicol Suppl (Archiv fur Toxikologie Supplement) 10:10–26
Moon YJ, Wang X, Morris ME (2006) Dietary flavonoids: effects on xenobiotic and carcinogen metabolism. Toxicol In Vitro 20(2):187–210. https://doi.org/10.1016/j.tiv.2005.06.048
Naik SR, Panda VS (2007) Antioxidant and hepatoprotective effects of Ginkgo biloba phytosomes in carbon tetrachloride-induced liver injury in rodents. Liver Int 27(3):393–399. https://doi.org/10.1111/j.1478-3231.2007.01463.x
Nambu S, Inoue K, Saski H (1987) Site-specific hypomethylation of the c-myc oncogene in human hepatocellular carcinoma. Jpn J Cancer Res 78(7):695–704
Niu ZS, Niu XJ, Wang WH (2016) Genetic alterations in hepatocellular carcinoma: an update. World J Gastroenterol 22(41):9069–9095. https://doi.org/10.3748/wjg.v22.i41.9069
NTP (2013) Toxicology and carcinogenesis studies of Ginkgo biloba extract (CAS No 90045-36-6) in F344/N rats and B6C3F1/N mice (Gavage studies). Natl Toxicol Program Tech Rep, Ser 578(0888-8051 (Print)):1–183
Plass C (2002) Cancer epigenomics. Hum Mol Genet 11(20):2479–2488
Pulukuri SM, Estes N, Patel J, Rao JS (2007) Demethylation-linked activation of urokinase plasminogen activator is involved in progression of prostate cancer. Cancer Res 67(3):930–939. https://doi.org/10.1158/0008-5472.CAN-06-2892
Rashid A, Wang JS, Qian GS, Lu BX, Hamilton SR, Groopman JD (1999) Genetic alterations in hepatocellular carcinomas: association between loss of chromosome 4q and p53 gene mutations. Br J Cancer 80(1–2):59–66. https://doi.org/10.1038/sj.bjc.6690321
Roche NimbleGen (2010) Nimblescan 64-bit. Release 2.6. Roche NimbleGen, Madison, WI
Rovida E, Di Maira G, Tusa I et al (2015) The mitogen-activated protein kinase ERK5 regulates the development and growth of hepatocellular carcinoma. Gut 64(9):1454–1465. https://doi.org/10.1136/gutjnl-2014-306761
Rozen S, Skaletsky HJ (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–386
Schubbert S, Shannon K, Bollag G (2007) Hyperactive Ras in developmental disorders and cancer. Nat Rev Cancer 7(4):295–308. https://doi.org/10.1038/nrc2109
Son KS, Kang HS, Kim SJ et al (2010) Hypomethylation of the interleukin-10 gene in breast cancer tissues. Breast 19(6):484–488. https://doi.org/10.1016/j.breast.2010.05.011
Terranova R, Vitobello A, Del Rio Espinola A et al (2017) Progress in identifying epigenetic mechanisms of xenobiotic-induced non-genotoxic carcinogenesis. Curr Opin Toxicol 3:62–70. https://doi.org/10.1016/j.cotox.2017.06.005
Tian F, Liu C, Wu Q et al (2013) Upregulation of glycoprotein nonmetastatic B by colony-stimulating factor-1 and epithelial cell adhesion molecule in hepatocellular carcinoma cells. Oncol Res 20(8):341–350. https://doi.org/10.3727/096504013X13657689382851
Tost J, El Abdalaoui H, Gut IG (2006) Serial pyrosequencing for quantitative DNA methylation analysis. Biotechniques 40(6):721–722. https://doi.org/10.2144/000112190 (724, 726)
Wahid B, Ali A, Rafique S, Idrees M (2017) New insights into the epigenetics of hepatocellular carcinoma. Biomed Res Int 2017:1609575. https://doi.org/10.1155/2017/1609575
Wand MP (1997) Data-based choice of histogram bin width. Am Stat 51(1):59–64. https://doi.org/10.2307/2684697
Wang Q, Sun Z, Yang HS (2008) Downregulation of tumor suppressor Pdcd4 promotes invasion and activates both beta-catenin/Tcf and AP-1-dependent transcription in colon carcinoma cells. Oncogene 27(11):1527–1535. https://doi.org/10.1038/sj.onc.1210793
Wang Q, Zhang Y, Yang HS (2012) Pdcd4 knockdown up-regulates MAP4K1 expression and activation of AP-1 dependent transcription through c-Myc. Biochim Biophys Acta 1823 10:1807–1814. https://doi.org/10.1016/j.bbamcr.2012.07.004
Wang XQ, Ng RK, Ming X et al (2013) Epigenetic regulation of pluripotent genes mediates stem cell features in human hepatocellular carcinoma and cancer cell lines. PLoS One 8(9):e72435. https://doi.org/10.1371/journal.pone.0072435
Woo HG, Choi JH, Yoon S et al (2017) Integrative analysis of genomic and epigenomic regulation of the transcriptome in liver cancer. Nat Commun 8(1):839. https://doi.org/10.1038/s41467-017-00991-w
Yang B, Guo M, Herman JG, Clark DP (2003) Aberrant promoter methylation profiles of tumor suppressor genes in hepatocellular carcinoma. Am J Pathol 163(3):1101–1107. https://doi.org/10.1016/S0002-9440(10)63469-4
Yeo W, Wong N, Wong WL, Lai PB, Zhong S, Johnson PJ (2005) High frequency of promoter hypermethylation of RASSF1A in tumor and plasma of patients with hepatocellular carcinoma. Liver Int 25(2):266–272. https://doi.org/10.1111/j.1478-3231.2005.01084.x
Zardo G, Tiirikainen MI, Hong C et al (2002) Integrated genomic and epigenomic analyses pinpoint biallelic gene inactivation in tumors. Nat Genet 32(3):453–458. https://doi.org/10.1038/ng1007
Zeller E, Hammer K, Kirschnick M, Braeuning A (2013) Mechanisms of RAS/beta-catenin interactions. Arch Toxicol 87(4):611–632. https://doi.org/10.1007/s00204-013-1035-3
Zhang YJ, Chen Y, Ahsan H et al (2005) Silencing of glutathione S-transferase P1 by promoter hypermethylation and its relationship to environmental chemical carcinogens in hepatocellular carcinoma. Cancer Lett 221(2):135–143. https://doi.org/10.1016/j.canlet.2004.08.028
Zhou Q, Lui VW, Yeo W (2011) Targeting the PI3 K/Akt/mTOR pathway in hepatocellular carcinoma. Future Oncol 7(10):1149–1167. https://doi.org/10.2217/fon.11.95
Acknowledgements
We would like to thank Florida State University Genomics Core and the NIEHS Microarray Core for running the Nimblegen methylation Arrays and Affymetrix Arrays, respectively. We would also like to acknowledge the staff in the NIEHS Pathology Support Core Laboratories and the NTP tissue archives for their expertise. We appreciate Drs. Jian-Liang Li and Alex Merrick for critically reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kovi, R.C., Bhusari, S., Mav, D. et al. Genome-wide promoter DNA methylation profiling of hepatocellular carcinomas arising either spontaneously or due to chronic exposure to Ginkgo biloba extract (GBE) in B6C3F1/N mice. Arch Toxicol 93, 2219–2235 (2019). https://doi.org/10.1007/s00204-019-02505-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-019-02505-7