Abstract
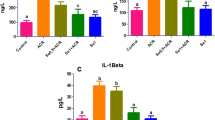
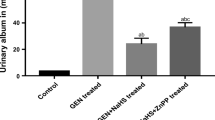
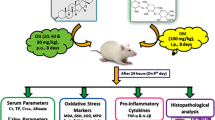
A single injection of adriamycin (ADR) induces marked and persistent proteinuria in rats that progress to glomerular and tubulointerstitial lesions. It has been shown that ADR-induced nephrotoxicity is mediated, at least in part, by oxidative stress that lead to inflammation. Endogenous hydrogen sulfide (H2S) is synthesized from l-cysteine and is an important signaling molecule in inflammation. This study evaluates the effect of DL-propargylglycine (PAG), an inhibitor of endogenous H2S formation, on the evolution of renal damage induced by ADR. The rats were injected i.p. with 0.15 M NaCl or PAG (50 mg/kg) 2 h after ADR injection (3.5 mg/kg). Control rats were injected with 0.15 M NaCl or PAG only. Twenty hours urine samples were collected for albuminuria and creatinine measurements on days 1 and 14 after saline or ADR injections and on days 2 and 15 blood samples were collected to measure plasma creatinine, then the rats were killed. The kidneys were removed for H2S formation evaluation, renal lipid peroxidation and glutathione levels, and histological and immunohistochemical analysis. On day 2 after ADR injection the rats presented increase in oxidative stress associated with neutrophils and macrophages influx in renal tissue. On day 15 the rats also presented increased desmin expression at glomerular edge and vimentin in cortical tubulointerstitium, as well as albuminuria. All these alterations were reduced by PAG injection. The protective effect of PAG on ADR nephrotoxicity was associated to decreased H2S formation and to restriction of oxidative stress and inflammation in the renal cortex.








Similar content being viewed by others
References
Arnhold J, Osipov AN, Spalteholz H, Panasenko OM, Schiller J (2001) Effects of hypochlorous acid on unsaturated phosphatidylcholines. Free Rad Biol Med 31:1111–1119
Baroni EA, Costa RS, Volpini AR, Coimbra TM (1999) Sodium bicarbonate treatment reduces renal injury, renal production of transforming growth factor-β, and urinary transforming growth factor-β excretion in rats with doxorubicin-induced nephropathy. Am J Kidney Dis 34:1–11
Bertani T, Poggi A, Pozzoni R, Delaini F, Sacchi G, Thoua Y, Mecca G, Remuzzi G, Donati MB (1982) Adriamycin-induced nephritic syndrome in rats. Lab Invest 46:16–23
Bertani T, Rocchi G, Sacchi G, Mecca G, Remuzzi G (1986) Adriamycin-induced glomerulosclerosis in the rat. Am J Kidney Dis 7:12–19
Bhatia M, Sidhapuriwala J, Moochhala SM, Moore PK (2005) Hydrogen sulphide is a mediator of carrageenan-induced hindpaw oedema in the rat. Brit J Pharmacol 145:141–144
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Charo IF, Taubman MB (2004) Chemokines in the pathogenesis of vascular disease. Circ Res 95:858–866
Chilvers ER, Cadwallader KA, Reed BJ, White JP, Condliffe AM (2000) The function and fate of neutrophils at inflamed site: prospects for therapeutic intervention. J R Coll Phys Lond 34:68–74
Coimbra TM, Janssen U, Gröne HJ, Ostendorf T, Kunter U, Schmidt H, Brabant G, Floege J (2000) Early events leading to renal injury in obese zucker (fatty) rats with type II diabetes. Kidney Int 57:167–182
Cunha TM, Dal-Secco D, Verri WA Jr, Guerrero AT, Souza GR, Vieira SM, Lotufo CM, Neto AF, Ferreira SH, Cunha FQ (2008) Dual role of hydrogen sulfide in mechanical inflammatory hypernociception. Eur J Pharmacol 590:127–135
Dal-Secco D, Cunha TM, Freitas A, Alves-Filho JC, Souto FO, Fukada SY, Grespan R, Alencar NM, Neto AF, Rossi MA, Ferreira SH, Hothersall JS, Cunha FQ (2008) Hydrogen sulfide augments neutrophil migration through enhancement of adhesion molecule expression and prevention of CXCR2 internalization: role of ATP-sensitive potassium channels. J Immunol 181:4287–4298
Dijkstra CD, Dopp EA, Joling P, Kraal G (1985) The heterogeneity of mononuclear phagocytes in lymphoid organs: distinct macrophage subpopulations in the rat recognized by antibodies ED1, 2 and 3. Immunology 54:589–599
Eardley KS, Zehnder D, Quinkler M, Lepenies J, Bates RL, Savage CO, Howie AJ, Adu D, Cockwell P (2006) The relationship between albuminuria, MCP-1/CCL2, and interstitial macrophages in chronic kidney disease. Kidney Int 69:1189–1197
Francescato HDC, Cunha FQ, Costa RS, Barbosa F Jr, Boim MA, Arnoni CP, da Silva CGA, Coimbra TM (2010) Inhibition of hydrogen sulfide formation reduces cisplatin-induced renal damage. Nephrol Dial Transplant. [Epub ahead of print]
Galli LG, Volpini RA, Costa RS, da Silva CGA, Coimbra TM (2001) Tubular cell lesion, albuminuria, and renal albumin handling in rats treated with adriamycin. Ren Fail 23:693–703
Geleilete TJ, Melo GC, Costa RS, Volpini RA, Soares TJ, Coimbra TM (2002) Role of myofibroblasts, transforming growth factor-β, endothelin, angiotensin II and fibronectin in the progression of tubulointerstitial nephritis induced by gentamicin. J Nephrol 15:633–642
Grone H-J, Weber K, Grone E, Helmcken U, Osborn M (1987) Coexpression of keratin and vimentin in damaged and regenerating tubular epithelia of kidney. Am J Pathol 129:1–8
Hartree EF (1972) Determination of protein: a modification of the Lowry method that gives a linear photometric response. Anal Biochem 48:422–427
Haugen HN (1953) The determination of endogenous creatinine in plasma and urine. Scand J Clin Lab Invest 5:48–57
Klahr S, Schreiner G, Ichikawa I (1988) The progression of renal disease. N Engl J Med 318:1657–1666
Kodama H, Mikasa H, Sasaki K, Awata S, Nakayama K (1984) Unusual metabolism of sulfur-containing amino acids in rats treated with d, l-propargylglycine. Arch Biochem Biophys 225:25–32
Laurell CB (1972) Electroimmunoassay. Scand J Clin Lab Invest 124(Suppl 1):21–37
Lauriat S, Linas SL (1998) The role of neutrophis in acute renal failure. Semin Nephrol 18:498–504
Li L, Bhatia M, Zhu YZ, Zhu YC, Ramnath RD, Wang ZJ, Anuar FBM, Whiteman M, Salto-Tellez M, Moore RK (2005) Hydrogen sulfide is a novel mediator of lipopolysaccharide-induced inflammation in the mouse. FASEB J 19:1196–1198
Martindale JL, Holbrook NJ (2002) Cellular response to oxidative stress: signaling for suicide and survival. J Cell Physiol 192:1–15
Mohan M, Kamble S, Gadhi P, Kasture S (2010) Protective effect of S. torvum on doxorubicin-induced nephrotoxicity in rats. Food Chem Toxicol 48:436–440
Moore PK, Bhatia M, Moochhala S (2003) Hydrogen sulfide: from the smell of the past to the mediator of the future? Trends Pharmacol Sci 24:609–611
Olson RD, Mushlin PS (1990) Doxorubicin cardiotoxicity analysis of prevailing hypotheses. FASEB J 4:3076–3086
Otaki Y, Miyauchi N, Higa M, Takada A, Kuroda T, Gejyo F, Shimizu F, Kawachi H (2008) Dissociation of NEPH1 from nephrin is involved in development of a rat model of focal segmental glomerulosclerosis. Am J Physiol Renal Physiol 295:F1376–F1387
Oz E, Ilhan MN (2006) Effects of melatonin in reducing the toxic effects of doxorubicin. Mol Cell Biochem 286:11–15
Rangan GK, Wang Y, Tay Y-C, Harris DCH (1999) Inhibition of nuclear factor-κB activation reduces cortical tubulointerstitial injury in proteinuric rats. Kidney Int 56:118–134
Reckless J, Tatalick LM, Grainger DL (2001) The pan-chemokine inhibitor NR58–3.14.3 abolishes tumor necrosis factor-α accumulation and leucocyte recruitment induced by lipopolysaccharide in vivo. Immunology 103:244–254
Reed DJ (1995) Cystathionine. Methods Enzimol 252:92–102
Rodriguez-Iturbe B, Pons H, Herrera-Acosta J, Johnson RJ (2001) Role of immunocompetent cells in nonimmune renal diseases. Kidney Int 59:1626–1640
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Shimizu H, Maruyama S, Yuzawa Y, Kato T, Miki Y, Suzuki S, Sato W, Morita Y, Maruyama H, Egashira K, Matsuo S (2003) Anti-monocyte chemoattractant protein-1 gene therapy attenuates renal injury induced by protein-overload proteinuria. J Am Soc Nephrol 14:1496–1505
Stipanuk MH, Beck PW (1982) Characterization of the enzyme capacity for cysteine desulphhydration in liver and kidney of the rat. Biochem J 206:267–277
Szabó C (2007) Hydrogen sulphide and its therapeutic potential. Nat Rev Drug Discov 6:917–935
Tang WW, Qi M, Warren JS, Van GY (1997) Chemokine expression in experimental tubulointerstitial nephritis. J Immunol 159:870–876
Uren JR, Ragin R, Chaykovsky M (1978) Modulation of cysteine metabolism in mice–effects of propargylglycine and l-cyst(e)ine-degrading enzymes. Biochem Pharmacol 27:2807–2814
Van Goor H, Ding H, Kees-Folts D, Grond J, Schreiner GF, Diamond JR (1994) Macrophages and renal disease. Lab Invest 71:456–464
Wada T, Furuichi K, Sakai N, Iwata Y, Kitagawa K, Ishida Y, Kondo T, Hashimoto H, Ishiwata Y, Mukaida N, Tomosugi N, Matsushima K, Egashira K, Yokoyama H (2004) Gene therapy via blockade of monocyte chemoattractant protein-1 for renal fibrosis. J Am Soc Nephrol 15:940–948
Wallace JL (2007) Hydrogen sulfide-releasing anti-inflammatory drugs. Trends Pharmacol Sci 28:501–505
Yang L, Zheng S, Epstein PN (2009) Metallothionein over-expression in podocytes reduces adriamycin nephrotoxicity. Free Rad Res 43:174–182
Yu T-W, Anderson D (1997) Reactive oxygen species-induced DNA damage and its modification: a chemical investigation. Mutat Res 379:201–210
Zhang J, Machida Y, Sugahara K, Kodama H (1994) Determination of d, l-propargylglycine and N-acetylpropargylglycine in urine and several tissues of d, l-propargylglycine-treated rats using liquid chromatography-mass spectrometry. J Chromatogr B Biomed Appl 14:375–379
Acknowledgments
The authors are grateful to Rubens Fernando de Melo, Giuliana Bertozi Francisco, and Flávio Henrique Leite, for their technical assistance. Dr. Heloísa D. C. Francescato, Dr. Roberto Silva Costa and Dr. Terezila Machado Coimbra are recipients of Conselho Nacional de Desenvolvimento Científico e Tecnológico, DF, Brazil, fellowship. This study was presented in part at the 42nd Annual Renal Week Meeting held in San Diego, California, in November of 2009.
Conflict of interest
The authors declare that they have no Conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Francescato, H.D.C., Marin, E.C.S., de Queiroz Cunha, F. et al. Role of endogenous hydrogen sulfide on renal damage induced by adriamycin injection. Arch Toxicol 85, 1597–1606 (2011). https://doi.org/10.1007/s00204-011-0717-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-011-0717-y