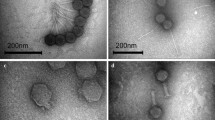
Abstract
Phages are called “good viruses” due to their ability to infect and kill pathogenic bacteria. Chlamydia are small, Gram-negative (G−) microbes that can be dangerous to human and animals. In humans, these bacteria are etiological agents of diseases such as psittacosis or respiratory tract diseases, while in animals, the infection may result in enteritis in cattle and chronic bowel diseases, as well as miscarriages in sheep. The first-known representative of chlamydiaphages was Chp1. It was discovered in Chlamydia psittaci isolates. Since then, four more species of chlamydiaphages have been identified [Chp2, Chp3, φCPG1 φCPAR39 (φCpn1) and Chp4]. All of them were shown to infect Chlamydia species. This paper described all known chlamydiaphages. They were characterised in terms of origin, host range, and their molecular structure. The review concerns the characterisation of bacteriophages that infects pathogenic and dangerous bacteria with unusual, intracellular life cycles that are pathogenic. In the era of antibiotic resistance, it is difficult to cure chlamydophilosis. Those bacteriophages can be an alternative to antibiotics, but before this happens, we need to get to know chlamydiaphages better.
Similar content being viewed by others
References
Ackermann H-W, Prangishvili D (2012) Prokaryote viruses studied by electron microscopy. Arch Virol 157:1843–1849
Bergey DH, Harrison FC, Breed RS, Hammer BW, Huntoon FM (2011) Bergey’s manual of systematic bacteriology. Volume 4. The Bacteroidetes, Spirochaetes, Tenericutes (Mollicutes), Acidobacteria, Fibrobacteres, Fusobacteria, Dictyoglomi, Gemmatimonadetes, Lentisphaerae, Verrucomicrobia, Chlamydiae and Planctomycetes. Springer, New York USA, pp 846–865
Bevan BJ, Labram J (1983) Laboratory transfer of a virus between isolates of Chlamydia psittaci. Vet Rec 112:280
Dąbrowska K, Switała-Jeleń K, Opolski A, Weber-Dąbrowska B, Górski A (2005) Bacteriophage penetration in vertebrates. J Appl Microbiol 98:7–13
Deptuła W, Pawlikowska M, Travnicek M (2002) Animal and human chlamydophilosis. Vet Med 58:337–340
Dokland T, McKenna R, Ilag LL, Bowman BR, Incardona NL, Fane BA, Rossmann MG (1997) Structure of a viral procapsid with molecular scaffolding. Nature 389:308–313
Dokland T, Bernal RA, Burch A, Pletnev S, Fane BA, Rossmann MG (1999) The role of scaffolding proteins in the assembly of the small single-stranded DNA virus ΦX174. J Mol Biol 288:595–608
Everson JS, Garner SA, Fane B, Liu BL, Lambden PR, Clarke IN (2002) Biological properties and cell tropism of Chp2, a bacteriophage of the obligate intracellular bacterium Chlamydophila abortus. J Bacteriol 184:2748–2754
Everson JS, Garner SA, Lambden PR, Fane BA, Clarke IN (2003) Host range of chlamydiaphages φCPAR39 and Chp3. J Bacteriol 185:6490–6492
Garner SA, Everson JS, Lambden PR, Fane BA, Clarke IN (2004) Isolation, molecular characterization and genome sequence of a bacteriophage (Chp3) from Chlamydophila pecorum. Virus Genes 28:207–214
Hoestgaard-Jensen K, Christiansen G, Honoré B, Birkelund S (2011) Influence of the Chlamydia pneumoniae AR39 bacteriophage φCPAR39 on chlamydial inclusion morphology. FEMS Immunol Med Microbiol 62:148–156
Hsia RC, Ohayon H, Gounon P, Dautry-Varsat A, Bavoil PM (2000a) Phage infection of the obligate intracellular bacterium, Chlamydia psittaci strain guinea pig inclusion conjunctivitis. Microbes Infect 2:761–772
Hsia RC, Ting LM, Bavoil PM (2000b) Microvirus of Chlamydia psittaci strain guinea pig inclusion conjunctivitis: isolation and molecular characterization. Microbiology 146:1651–1660
Karunakaran KP, Blanchard JF, Raudonikiene A, Shen C, Murdin AD, Brunham RC (2002) Molecular detection and seroepidemiology of the Chlamydia pneumoniae bacteriophage (φCpn1). J Clin Microbiol 40:4010–4014
Kim M, Park E, Roh SW, Bae J (2011) Diversity and abundance of single-stranded DNA viruses in human feces. Appl Environ Microbiol 77:8062–8070
King AMQ, Adams MJ, Carstens EB, Lefkowitz EJ (2012) Virus Taxonomy: classification and nomenclature of viruses. Ninth report of the International Committee on Taxonomy of Viruses. Elsevier–Academic Press, London, UK (ISBN: 978-0-12-384684-6), pp 385–393
Krupovic M, Forterre P (2011) Microviridae goes temperate: microvirus-related provirus reside in the genomes of Bacteroidetes. PLoS ONE 6:e19893
Liu BL, Everson JS, Fane B, Giannikopoulou P, Vretou E, Lambden PR, Clarke IN (2000) Molecular characterization of a bacteriophage (Chp2) from Chlamydia psittaci. J Virol 74:3464–3469
Mårdh PA, Paavonen J, Puolakkainen M (1989) Chlamydia. Plenum Medical Book Company, New York
Pawlikowska M, Deptuła W (2012) Chlamydia and chlamydophila in human and animals. University of Stettin, Stettin
Pawlikowska M, Deptuła W (2007) Human diseases caused by Chlamydiae and Chlamydophila. Adv Hyg Med Exp 61:708–717
Rank RG, Bowlin AK, Cané S, Shou H, Liu Z, Nagarajan UM, Bavoli PM (2009) Effect of chlamydiaphage phiCPG1 on the course of conjunctival infection with “Chlamydia cavie” in guinea pigs. Infect Immun 77:1216–1221
Read TD, Brunham RC, Shen C et al (2000a) Genome sequences of Chlamydia trachomatis MoPN and Chlamydia pneumoniea AR39. Nucleic Acids Res 28:1397–1406
Read TD, Fraser CM, Hsia RC, Bavoil PM (2000b) Comparative analysis of Chlamydia bacteriophages reveals variation localization to a putative receptor binding domain. Microb Comp Genomics 5:223–231
Roux S, Krupovic M, Poulet A, Debroas D, Enault F (2012) Evolution and diversity of the Microviridae family through a collection of 81 new complete genomes assembled from virome reads. PLoS ONE 7(7):e40418
Rupp J, Solbach W, Gieffers J (2007) Prevalence, genetic conservation and transmissibility of the Chlamydia pneumoniae bacteriophage (φCpn1). FEMS Microbiol Lett 273:45–49
Sait M, Livingstone M, Graham R, Inglis NF, Wheelhouse N, Longbottom D (2011) Identification, sequencing and molecular analysis of Chp4, a novel chlamydiaphage of Chlamydophila abortus belonging to the family Microviridae. J Gen Virol 92:1733–1737
Salim O, Skilton RJ, Lambden PR, Fane BA, Clarke IN (2008) Behind the chlamydial cloak: the replication cycle of chlamydiaphage Chp2, revealed. Virology 377:440–445
Skilton RJ, Cutcliffe LT, Pickett MA, Lambden PR, Fane BA, Clarke IN (2007) Intracellular parasitism of chlamydiae: specific infectivity of chlamydiophage Chp2 in Chlamydophila abortus. J Bacteriol 189:4957–4959
Storey CC, Lusher M, Richmond SJ, Bacon J (1989a) Further characterization of a bacteriophage recovered from an avian strain of Chlamydia psittaci. J Gen Virol 70:1321–1327
Storey CC, Lusher M, Richmond SJ (1989b) Analysis of the complete nucleotide sequence of Chp1, a phage which infects avian Chlamydia psittaci. J Gen Virol 70:3381–3390
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Śliwa-Dominiak, J., Suszyńska, E., Pawlikowska, M. et al. Chlamydia bacteriophages. Arch Microbiol 195, 765–771 (2013). https://doi.org/10.1007/s00203-013-0912-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-013-0912-8