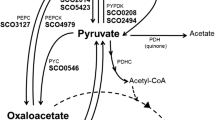
Abstract
The chromosome of Streptomyces coelicolor A3(2), a model organism for the genus Streptomyces, contains a cryptic type I polyketide synthase (PKS) gene cluster which was revealed when the genome was sequenced. The ca. 54-kb cluster contains three large genes, cpkA, cpkB and cpkC, encoding the PKS subunits. In silico analysis showed that the synthase consists of a loading module, five extension modules and a unique reductase as a terminal domain instead of a typical thioesterase. All acyltransferase domains are specific for a malonyl extender, and have a B-type ketoreductase. Tailoring and regulatory genes were also identified within the gene cluster. Surprisingly, some genes show high similarity to primary metabolite genes not commonly identified in any antibiotic biosynthesis cluster. Using western blot analysis with a PKS subunit (CpkC) antibody, CpkC was shown to be expressed in S. coelicolor at transition phase. Disruption of cpkC gave no obvious phenotype.



Similar content being viewed by others
Abbreviations
- AT:
-
Acyltransferase
- ACP:
-
Acyl carrier protein
- DH:
-
Dehydratase
- DEBS:
-
6-Deoxyerythronolide synthase
- ER:
-
Enoyl reductase
- KR:
-
Ketoreductase
- KS:
-
β-Ketoacylsynthase
- MS:
-
Mannitol soya flour medium
- NRPS:
-
Non-ribosomal peptide synthetase
- PKS:
-
Polyketide synthase
- SMM:
-
Supplemented minimal medium
- TD:
-
Terminal reductase
References
Aparicio JF, Molnar I, Schwecke T, Konig A, Haydock SF, Khaw L, Staunton J, Leadlay PF (1996) Organization of the biosynthetic gene cluster for rapamycin in Streptomyces hygroscopicus: analysis of the enzymatic domains in the modular polyketide synthase. Gene 169:9–16
Bate N, Butler AR, Gandecha AR, Cundliffe E (1999) Multiple regulatory genes in the tylosin biosynthetic cluster of Streptomyces fradiae. Chem Biol 6:617–624
Bentley SD, Chater KF, Cerdeno-Tarraga AM, Challis GL, Thomson NR, James KD, Harris DE, Quail MA, Kieser H, Harper D, Bateman A, Brown S, Chandra G, Chen CW, Collins M, Cronin A, Fraser A, Goble A, Hidalgo J, Hornsby T, Howarth S, Huang CH, Kieser T, Larke L, Murphy L, Oliver K, O’Neil S, Rabbinowitsch E, Rajandream MA, Rutherford K, Rutter S, Seeger K, Saunders D, Sharp S, Squares R, Squares S, Taylor K, Warren T, Wietzorrek A, Woodward J, Barrell BG, Parkhill J, Hopwood DA (2002) Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 417:141–147
Black T, Wolk C (1994) Analysis of a Het- mutation in Anabaena sp. strain PCC 7120 implicates a secondary metabolite in the regulation of heterocyst spacing. J Bacteriol 176:2282–2292
Brautaset T, Sekurova ON, Sletta H, Ellingsen TE, StrLm AR, Valla S, Zotchev SB (2000) Biosynthesis of the polyene antifungal antibiotic nystatin in Streptomyces noursei ATCC 11455: analysis of the gene cluster and deduction of the biosynthetic pathway. Chem Biol 7:395–403
Broadhurst RW, Nietlispach D, Wheatcroft MP, Leadlay PF, Weissman KJ (2003) The structure of docking domains in modular polyketide synthases. Chem Biol 10:723–731
Caffrey P (2003) Conserved Amino Acid Residues Correlating With Ketoreductase Stereospecificity in Modular Polyketide Synthases. ChemBioChem 4:649–662
Caffrey P(2005) The stereochemistry of ketoreduction. Chem Biol 12:1060–1062
Casqueiro J, Gutierrez S, Banuelos O, Hijarrubia MJ, Martin JF(1999) Gene targeting in Penicillium chrysogenum: disruption of the lys2 gene leads to penicillin overproduction. J Bacteriol 181:1181–1188
Cerdeno AM, Bibb MJ, Challis GL (2001) Analysis of the prodiginine biosynthesis gene cluster of Streptomyces coelicolor A3(2): new mechanisms for chain initiation and termination in modular multienzymes. Chem Biol 8:817–829
Challis GL, Hopwood DA (2003) Synergy and contingency as driving forces for the evolution of multiple secondary metabolite production by Streptomyces species. Proc Natl Acad Sci USA 100(Suppl 2):14555–14561
Cho YH, Kim EJ, Chung HJ, Choi JH, Chater KF, Ahn BE, Shin JH, Roe JH (2003) The pqrAB operon is responsible for paraquat resistance in Streptomyces coelicolor. J Bacteriol 185:6756–6763
Donadio S, Katz L (1992) Organization of the enzymatic domains in the multifunctional polyketide synthase involved in erythromycin formation in Saccharopolyspora erythraea. Gene 111: 51–60
Faust B, Hoffmeister D, Weitnauer G, Westrich L, Haag S, Schneider P, Decker H, Kunzel E, Rohr J, Bechthold A (2000) Two new tailoring enzymes, a glycosyltransferase and an oxygenase, involved in biosynthesis of the angucycline antibiotic urdamycin A in Streptomyces fradiae Tu2717. Microbiology 146:147–154
Feitelson JS, Malpartida F, Hopwood DA (1985) Genetic and biochemical characterization of the red gene cluster of Streptomyces coelicolor A3(2). J Gen Microbiol 131: 2431–2441
Ginolhac A, Jarrin C, Robe P, Perriere G, Vogel TM, Simonet P, Nalin R. (2005) Type I polyketide synthases may have evolved through horizontal gene transfer. J Mol Evol 60:716–725
Gokhale RS, Tsuji SY, Cane DE, Khosla C (1999) Dissecting and exploiting intermodular communication in polyketide synthases. Science 284:482–485
Han L, Yang K, Kulowski K, Wendt-Pienkowski E, Hutchinson CR, Vining LC (2000) An acyl-coenzyme A carboxylase encoding gene associated with jadomycin biosynthesis in Streptomyces venezuelae ISP5230. Microbiology 146: 903–910
Haydock SF, Aparicio JF, Molnar I, Schwecke T, Khaw LE, Konig A, Marsden AF, Galloway IS, Staunton J, Leadlay PF (1995) Divergent sequence motifs correlated with the substrate specificity of (methyl)malonyl-CoA:acyl carrier protein transacylase domains in modular polyketide synthases. FEBS Lett 374: 246–248
Hesketh AR, Chandra G, Shaw AD, Rowland JJ, Kell DB, Bibb MJ, Chater K.F (2002). Primary and secondary metabolism, and post-translational protein modifications, as portrayed by proteomic analysis of Streptomyces coelicolor. Mol Microbiol 46: 917–932
Huang J, Shi J, Molle V, Sohlberg B, Weaver D, Bibb MJ, Karoonuthaisiri N, Lih CJ, Kao C, Buttner MJ, Cohen SN. (2005) Cross-regulation among disparate antibiotic biosynthetic pathways of Streptomyces coelicolor. Mol Microbiol 58:1276–1287
Ikeda H, Ishikawa J, Hanamoto A, Shinose M, Kikuchi H, Shiba T, Sakaki Y, Hattori M, Omura S (2003) Complete genome sequence and comparative analysis of the industrial microorganism Streptomyces avermitilis. Nat Biotechnol 21: 526–531
Ikeno S, Aoki D, Hamada M, Hori M, Tsuchiya KS. (2006) DNA sequencing and transcriptional analysis of the kasugamycin biosynthetic gene cluster from Streptomyces kasugaensis M338-M1. J Antibiot 59: 18–28
Ishikawa J, Hotta K (1999) FramePlot: a new implementation of the frame analysis for predicting protein-coding regions in bacterial DNA with a high G + C content. FEMS Microbiol Lett 174: 251–253
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical streptomyces genetics. The John Innes Foundation, Norwich, UK
Konz D, Marahiel MA (1999) How do peptide synthetases generate structural diversity? Chem Biol 6:R39–R48
Kotowska M, Pawlik K, Butler AR, Cundliffe E, Takano E, Kuczek K (2002) Type II thioesterase from Streptomyces coelicolor A3(2). Microbiology 148:1777–1783
Kuczek K, Pawlik K, Kotowska M, Mordarski M. (1997) Streptomyces coelicolor DNA homologous with acyltransferase domains of type I polyketide synthase gene complex. FEMS Microbiol Lett 157: 195–200
Lau J, Fu H, Cane DE, Khosla C (1999) Dissecting the role of acyltransferase domains of modular polyketide synthases in the choice and stereochemical fate of extender units. Biochemistry 38:1643–1651
Li A, Piel J (2002) A gene cluster from a marine Streptomyces encoding the biosynthesis of the aromatic spiroketal polyketide griseorhodin A. Chem Biol 9:1017–1026
Liu W, Shen B (2000) Genes for production of the enediyne antitumor antibiotic C-1027 in Streptomyces globisporus are clustered with the cagA gene that encodes the C-1027 apoprotein. Antimicrob Agents Chemother 44:382–392
Liu W, Christenson SD, Standage S, Shen B (2002) Biosynthesis of the enediyne antitumor antibiotic C-1027. Science 297:1170–1173
Liu W, Nonaka K, Nie L, Zhang J, Christenson SD, Bae J, Van Lanen SG, Zazopoulos E, Farnet CM, Yang CF, Shen B (2005) The neocarzinostatin biosynthetic gene cluster from S. carzinostaticus ATCC 15944 involving two iterative type I polyketide synthases. Chem Biol 12:293–302
Lombo F, Brana AF, Salas JA, Mendez C (2004) Genetic organization of the biosynthetic gene cluster for the antitumor angucycline oviedomycin in Streptomyces antibioticus ATCC 11891. ChemBioChem 5:1181–1187
Matsuno K, Yamada Y, Lee CK, Nihira T (2004) Identification by gene deletion analysis of barB as a negative regulator controlling an early process of virginiamycin biosynthesis in Streptomyces virginiae. Arch Microbiol 181:52–59
Otsuka M, Ichinose K, Fujii I, Ebizuka Y (2004) Cloning, sequencing, and functional analysis of an iterative type I polyketide synthase gene cluster for biosynthesis of the antitumor chlorinated polyenone neocarzilin in “Streptomyces carzinostaticus”. Antimicrob Agents Chemother 48:3468–3476
Reid R, Piagentini M, Rodriguez E, Ashley G, Viswanathan N, Carney J, Santi DV, Hutchinson CR, McDaniel R. (2003) A model of structure and catalysis for ketoreductase domains in modular polyketide synthases. Biochemistry 42:72–79
Rodriguez E, Gramajo H (1999) Genetic and biochemical characterization of the alpha and beta components of a propionyl–CoA carboxylase complex of Streptomyces coelicolor A3(2). Microbiology 145:3109–3119
Rodriguez E, Banchio C, Diacovich L, Bibb MJ, Gramajo H (2001) Role of an essential acyl coenzyme A carboxylase in the primary and secondary metabolism of Streptomyces coelicolor A3(2). Appl Environ Microbiol 67:4166–4176
Rudd BA, Hopwood DA (1979) Genetics of actinoorhodin biosynthesis by Streptomyces coelicolor A3(2). J Gen Microbiol 114:35–43
Sambrook J, Fritsch EF, Maniatis T (1989) Moleculer cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Shen B (2003) Polyketide biosynthesis beyond the type I, II and III polyketide synthase paradigms. Curr Opin Chem Biol 7:285–295
Silakowski B, Nordsiek G, Kunze B, Blocker H, Muller R (2001) Novel features in a combined polyketide synthase/non-ribosomal peptide synthetase: the myxalamid biosynthetic gene cluster of the myxobacterium Stigmatella aurantiaca Sga15. Chem Biol 8:59–69
Siskos AP, Baerga-Ortiz A, Bali S, Stein V, Mamdani H, Spiteller D, Popovic B, Spencer JB, Staunton J, Weissman KJ, Leadlay PF. (2005) Molecular basis of Celmer’s rules: stereochemistry of catalysis by isolated ketoreductase domains from modular polyketide synthases. Chem Biol 12:1145–1153
Staunton J, Weissman KJ (2001) Polyketide biosynthesis:a millennium review. Nat Prod Rep 18:380–416
Takano E (2006) gamma-Butyrolactones: Streptomyces signalling molecules regulating antibiotic production and differentiation. Curr Opin Microbiol 9:1–8
Takano E, Chakraburtty R, Nihira T, Yamada Y, Bibb MJ (2001) A complex role for the gamma-butyrolactone SCB1 in regulating antibiotic production in Streptomyces coelicolor A3(2). Mol Microbiol 41:1015–1028
Takano E, Kinoshita H, Mersinias V, Bucca G, Hotchkiss G, Nihira T, Smith CP, Bibb M, Wohlleben W, Chater K (2005) A bacterial hormone (the SCB1) directly controls the expression of a pathway-specific regulatory gene in the cryptic type I polyketide biosynthetic gene cluster of Streptomyces coelicolor. Mol Microbiol 56:465–479
Wang L, Vining LC (2003) Control of growth, secondary metabolism and sporulation in Streptomyces venezuelae ISP5230 by jadW(1), a member of the afsA family of gamma-butyrolactone regulatory genes. Microbiology 149:1991–2004
Weber T, Welzel K, Pelzer S, Vente A, Wohlleben W (2003) Exploiting the genetic potential of polyketide producing streptomycetes. J Biotechnol 106:221–232
Wendt-Pienkowski E, Huang Y, Zhang J, Li B, Jiang H, Kwon H, Hutchinson CR, Shen B (2005) Cloning, sequencing, analysis, and heterologous expression of the fredericamycin biosynthetic gene cluster from Streptomyces griseus. J Am Chem Soc 127:16442–16452
Wietzorrek A, Bibb M (1997) A novel family of proteins that regulates antibiotic production in streptomycetes appears to contain an OmpR-like DNA-binding fold. Mol Microbiol 25:1181–1184
Yadav G, Gokhale RS, Mohanty D (2003) Computational approach for prediction of domain organization and substrate specificity of modular polyketide synthases. J Mol Biol 328:335–363
Yonaha K, Nishie M, Aibara S. (1992) The primary structure of omega-amino acid: pyruvate aminotransferase. J Biol Chem 267:12506–12510
Yu TW, Hopwood DA (1995) Ectopic expression of the S. coelicolor whiE genes for polyketide spore pigment synthesis and their interaction with the act genes for actinorhodin biosynthesis. Microbiology 141:2779–2791
Yu TW, Bai L, Clade D, Hoffmann D, Toelzer S, Trinh K, Xu J, Moss S, Leistner E, Floss HG. (2002) The biosynthetic gene cluster of the maytansinoid antitumor agent ansamitocin from Actinosynnema pretiosum. Proc Natl Acad Sci USA 99:7968–7973
Zhu G, LaGier MJ, Stejskal F, Millership JJ, Cai X, Keithly JS (2002) Cryptosporidium parvum: the first protist known to encode a putative polyketide synthase. Gene 298:79–89
Acknowledgments
This manuscript is dedicated to the late Katarzyna Kuczek. We thank David Hopwood, Wolfgang Wohlleben, Tillman Weber, Jolanta Zakrzewska-Czerwińska and Kira Weissman for critical reading of the manuscript. K. Pawlik, M. Kotowska and K. Kuczek were supported by Polish Ministry of Scientific Research and Information Technology, grant number 3 P04B 004 25 and by the Institute of Immunology and Experimental Therapy. E. Takano was supported from a grant from Human Frontier Science Program RG0330/1998. K. Pawlik was also supported by the John Innes Foundation. Antibodies were raised in the Institute of Immunology and Experimental Therapy in Wroclaw.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pawlik, K., Kotowska, M., Chater, K.F. et al. A cryptic type I polyketide synthase (cpk) gene cluster in Streptomyces coelicolor A3(2). Arch Microbiol 187, 87–99 (2007). https://doi.org/10.1007/s00203-006-0176-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-006-0176-7