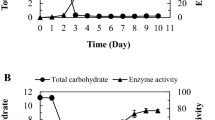
Abstract
Genes encoding dextranolytic enzymes were isolated from Paenibacillus strains Dex40-8 and Dex50-2. Single, similar but non-identical dex1 genes were isolated from each strain, and a more divergent dex2 gene was isolated from strain Dex50-2. The protein deduced from the Dex40-8 dex1 gene sequence had 716 amino acids, with a predicted Mr of 80.8 kDa. The proteins deduced from the Dex50-2 dex1 and dex2 gene sequences had 905 and 596 amino acids, with predicted Mr of 100.1 kDa and 68.3 kDa, respectively. The deduced amino acid sequences of all three dextranolytic proteins had similarity to family 66 glycosyl hydrolases and were predicted to possess cleavable N-terminal signal peptides. Homology searches suggest that the Dex40-8 and Dex50-2 Dex1 proteins have one and two copies, respectively, of a carbohydrate-binding module similar to CBM_4_9 (pfam02018.11). The Dex50-2 Dex2 deduced amino acid sequence had highest sequence similarity to thermotolerant dextranases from thermophilic Paenibacillus strains, while the Dex40-8 and Dex50-2 Dex1 deduced protein sequences formed a distinct sequence clade among the family 66 proteins. Examination of seven Paenibacillus strains, using a polymerase chain reaction-based assay, indicated that multiple family 66 genes are common within this genus. The three recombinant proteins expressed in Escherichia coli possessed dextranolytic activity and were able to convert ethanol-insoluble blue dextran into an ethanol-soluble product, indicating they are endodextranases (EC 3.2.1.11). The reaction catalysed by each enzyme had a distinct temperature and pH dependence.




Similar content being viewed by others
References
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Bruijn JM de (2000) Processing of frost-damaged beets at CSM and the use of dextranases. Zuckerindustrie 125:898–902
Coutinho PM, Henrissat B (1999) Carbohydrate-active enzymes: an integrated database approach. In: Gilbert HJ, Davies G, Henrissat B, Svensson B (eds) Recent advances in carbohydrate bioengineering. The Royal Society of Chemistry, Cambridge, pp 3–12
Davis KA, Anseth KS (2002) Controlled release from crosslinked degradable networks. Crit Rev in Ther Drug Carrier Syst 19:385–423
Finnegan PM, Brumbley SM, O’Shea MG, Nevalainen KMH, Bergquist PL (2004a) Paenibacillus isolates possess diverse dextran-degrading enzymes. J Appl Microbiol 97:477–485. DOI 10.1111/j.1365-2672.2004.02325.x
Finnegan PM, Brumbley SM, O’Shea MG, Nevalainen KMH, Bergquist PL (2004b) Isolation and characterisation of genes encoding thermoactive and thermostable dextranases from two thermotolerant soil bacteria. Curr Microbiol 49:327–333. DOI 10.1007/s00284-004-4308-5
Flores N, Valle F, Bolivar F, Merino E (1992) Recovery of DNA from agarose gels stained with methylene blue. Biotechniques 13:203–205
Igarashi T, Yamamoto A, Goto N (1995) Sequence analysis of the Streptococcus mutans Ingbritt dexA gene encoding extracellular dextranase. Microbiol Immunol 39:853–860
Igarashi T, Yamamoto A, Goto N (2001) Nucleotide sequence and molecular characterization of a dextranase gene from Streptococcus downei. Microbiol Immunol 45:341–348
Igarashi T, Morisaki H, Yamamoto Y, Goto N (2002) An essential amino acid residue for catalytic activity of the dextranase of Streptococcus mutans. Oral Microbiol Immunol 17:193–196
Igarashi T, Morisaki H, Goto N (2004) Molecular characterization of dextranse from Streptococcus rattus. Microbiol Immunol 48:155–162
Khalikova E, Susi P, Usanov N, Korpela T (2003) Purification and properties of extracellular dextranase from a Bacillus sp. J Chromatogr B 796:315–326
Lever M (1973) Colorimetric and fluorometric carbohydrate determination with p-hydroxybenzoic acid hydrazide. Biochem Med 7:274–281
Loesche WJ (1986) Role of Streptococcus mutans in human dental decay. Microbiol Rev 50:353–380
Marchler-Bauer A, Anderson JB, DeWeese-Scott C, Fedorova ND, Geer LY, He S, Hurwitz DI, Jackson JD, Jacobs AR, Lanczycki CJ, Liebert CA, Liu C, Madej T, Marchler GH, Mazumder R, Nikolskaya AN, Panchenko AR, Rao BS, Shoemaker BA, Simonyan V, Song JS, Thiessen PA, Vasudevan S, Wang Y, Yamashita RA, Yin JJ, Bryant SH (2003) CDD: a curated Entrez database of conserved domain alignments. Nucleic Acids Res 31:383–387
Morisaki H, Igarashi T, Yamamoto A, Goto N (2002) Analysis of a dextran-binding domain of the dextranase of Streptococcus mutans. Lett Appl Microbiol 35:223–227
Morris DD, Reeves RA, Gibbs MD, Saul DJ, Bergquist PL (1995) Correction of the β-mannase domain of the celC pseudogene from Caldicellulosiruptor saccharolyticus and activity of the gene product on kraft pulp. Appl Environ Microbiol 61:2262–2269
Oguma T, Kouichiro T, Kobayashi M (1994) Purification and properties of a novel enzyme from Bacillus spp T-3040, which catalyzes the conversion of dextran to cyclic isomaltooligosaccharides. FEBS Lett 345:135–138
Oguma T, Kurokawa T, Tobe K, Kitao S, Kobayashi M (1999) Cloning and sequence analysis of the gene for glucodextranase from Arthrobacter globiformis T-3044 and expression in Escherichia coli cells. Biosci Biotechnol Biochem 63:2174–2182
Ohnishi Y, Kubo S, Ono Y, Nozaki M, Gonda Y, Okano H, Matsuya T, Matsushiro A, Morita T (1995) Cloning and sequencing of the gene coding for dextranase from Streptococcus salivarius. Gene 156:93–96
Porath J (1997) From gel filtration to adsorptive size exclusion. J Prot Chem 16:463–468
Rose TM, Schultz ER, Henikoff JG, Pietrokovski S, McCallum CM, Henikoff S (1998) Consensus-degenerate hybrid oligonucleotide primers for amplification of distantly related sequences. Nucleic Acids Res 26:1628–1635
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Tilbury RH, French SM (1974) Further studies on enzymic hydrolysis of dextrans in mill juice by dextranases and amylases. In: Proceedings of the 15th congress of the international society of sugar cane technology, pp 1277–1287
Tollofsrud S, Elgjo GI, Prough DS, Williams CA, Traber DL, Kramer GC (2001) The dynamics of vascular volume and fluid shifts of lactated Ringer’s solution and hypertonic-saline-dextran solutions infused in normovolemic sheep. Anesth Analg 93:823–831
Walker GJ (1978) Dextrans. In: Manners DJ (ed) Biochemistry of carbohydrates, vol 16. University Park, Baltimore, pp 75–126
Wanda S-Y, Curtiss R III (1994) Purification and characterization of Streptococcus sobrinus dextranase produced in recombinant Escherichia coli and sequence analysis of the dextranase gene. J Bacteriol 176:3839–3850
Watanabe K, Miyake K, Suzuki Y (2001) Identification of catalytic and substrate-binding site residues in Bacillus cereus ATCC7064 oligo-1,6-glucosidase. Biosci Biotechnol Biochem 65:2058–2064
Wynter CVA, Galea CF, Cox LM, Dawson MW, Pate BK, Hamilton S, De Jersey J, Inkerman PA (1995) Thermostable dextranases: screening, detection and preliminary characterization. J Appl Bacteriol 79:203–212
Wynter CVA, Chang M, De Jersey J, Patel B, Inkerman PA, Hamilton S (1997) Isolation and characterization of a thermostable dextranase. Enzyme Microb Technol 20:242–247
Acknowledgements
This work was funded by grants from BSES Limited, Indooroopilly, Queensland, Australia, the External Collaborative Research Grant scheme of Macquarie University, Sydney, Australia and the SPIRT scheme of the Australian Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finnegan, P.M., Brumbley, S.M., O’Shea, M.G. et al. Diverse dextranase genes from Paenibacillus species. Arch Microbiol 183, 140–147 (2005). https://doi.org/10.1007/s00203-004-0756-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-004-0756-3