Abstract
Purpose
The purpose of this study was to evaluate the biomechanical behavior of an all-suture glenoid anchor in comparison with a more conventional screw-in glenoid anchor, with regard to maximum load to failure and tensile displacement.
Methods
All mechanical testing was performed using an Instron ElectroPuls E1000 mechanical machine, with a 10 N pre-load and displacement rate of 10 mm/min. Force–displacement curves were generated, with calculation of maximum load, maximum displacement, displacement at 50 N and stiffness. Pretesting of handset Y-Knots in bone analog models revealed low force displacement below 60 N of force. Subsequently, three groups of anchors were tested for pull out strength in bovine bone and cadaver glenoid bone: a bioabsorbable screw-in anchor (Bio Mini-Revo, ConMed Linvatec), a handset all-suture anchor (Y-Knot, ConMed Linvatec) and a 60 N pre-tensioned all-suture anchor (Y-Knot). A total of 8 anchors from each group was tested in proximal tibia of bovine bone and human glenoids (age range 50–90).
Results
In bovine bone, the Bio Mini-Revo displayed greater maximum load to failure (206 ± 77 N) than both the handset (140 ± 51 N; P = 0.01) and the pre-tensioned Y-Knot (135 ± 46 N; P = 0.001); no significant difference was seen between the three anchor groups in glenoid bone. Compared to the screw-in anchors, the handset all-suture anchor displayed inferior fixation, early displacement and greater laxity in the bovine bone and cadaveric bone (P < 0.05). Pre-tensioning the all-suture anchor to 60 N eliminated this behavior in all bone models.
Conclusions
Handset Y-Knots display low force anchor displacement, which is likely due to slippage in the pilot hole. Pre-tensioning the Y-Knot to 60 N eliminates this behavior.
Level of evidence
I.
Similar content being viewed by others
Introduction
Many shoulder surgeons are interested in the development of all-suture anchors, designed for use in glenoid labrum repair. The main proposed advantage is the preservation of host bone through the use of smaller pilot holes compared with conventional anchors. Furthermore, the smaller diameter pilot hole can allow increased numbers of anchors to be placed in the glenoid. Additionally, in the event of anchor pullout, the absence of hard loose bodies in the joint that can cause chondral injury is appealing.
A recent biomechanical analysis by Mazzocca et al. [15] compared the use of an all-suture anchor (JuggerKnot 1.4 mm anchor, Biomet, Inc., Warsaw, IN, USA) to a classic solid anchor (SutureTak, Arthrex, Inc., Naples, FL, USA) in glenoid labral repair. No statistical difference was detected in maximum load to failure or displacement at failure. However, the force required to cause 2 mm of labral displacement (thought to be indicative of clinical failure [6, 18]) was significantly lower with the all-suture anchor than the conventional anchor. The authors hypothesized that this may have been caused by displacement of the all-suture anchor in the pilot hole.
The Y-Knot (ConMed Linvatec, Largo, FL, USA) is a 1.3-mm all-suture anchor similar to the JuggerKnot. When deployed, the suture anchor contracts vertically and expands laterally, forming a 360° fixation within the pilot hole; this suture is intended to lock the anchor suture against the cortical bone. However, as the use of all-suture anchors increases, it will be important to understand their biomechanical behavior in order to maximize their clinical efficacy.
The purpose of this study was to evaluate the biomechanical behavior of an all-suture glenoid anchor in comparison with a more conventional screw-in glenoid anchor. The hypothesis was that an all-suture anchor would have similar maximum load to failure compared to a screw-in anchor in both bovine bone and human cadaveric glenoid bone. Additionally, based on the observations by Mazzoca et al. [15], it was hypothesized that an all-suture anchor would demonstrate early displacement in a tensile displacement curve and that pre-tensioning the all-suture anchor would eliminate this behavior.
Materials and methods
Suture anchors
Two types of anchors were selected for testing: an all-suture anchor (Y-Knot, ConMed Linvatec) and a bioabsorbable, screw-in type anchor (Bio Mini-Revo, ConMed Linvatec) (Fig. 1). The Y-Knot is made entirely of high strength #2 Hi-Fi suture (ConMed Linvatec). The Bio Mini-Revo is made from self-reinforced 96L/4D PLA, is 3.1 mm in diameter and 11 mm long and has a single strand #2 Hi-Fi suture. The Bio Mini-Revo was used as for comparison, as it has previously been demonstrated that fully threaded, screw-in anchor designs have consistently higher failure strengths than smaller, press-fit anchors [4, 5]. Furthermore, because the Bio Mini-Revo is manufactured by the same company, the suture material is identical, eliminated this as a source of variation in testing.
A single orthopedic surgeon inserted all anchors as per the manufacturer’s instructions. A 1.3-mm pilot drill hole for the Y-Knot was made through a drill guide, following which the Y-Knot was inserted, and handset by pulling on both suture limbs until the anchor was felt to deploy. Using the same drill guide, a 2.1-mm pilot hole was made for the Bio Mini-Revo, a 2.4-mm self-drilling bone tap inserted, followed by the 3.1-mm screw-in anchor. The surgeon then tied all knots with a total of 6 non-sliding, alternating half-hitch knots. The suture loop was tied over a steel cross pin attached to the load cell, set to create a suture length of 5 cm. Before each mechanical pullout test was performed, the length of the suture loop was measured using digital calipers.
Mechanical testing protocol
All mechanical testing was performed using an Instron ElectroPuls E1000 mechanical machine (Instron, Canton, MA) with a 2-kN Dynacell load cell; this testing machine is equipped with both a linear variable displacement transformer (LVDT) and digital micrometer. The Instron ElectroPuls E1000 has both measurement accuracy and precision (test–retest reliability) for force measurements of 0.25 N and for displacement of 10 microns. The bone analog blocks, bovine tibias and human glenoids were gripped securely in the testing machine base clamp. The loading vector of the actuator was placed in alignment with the suture anchor insertion; all tensile forces were applied parallel to the axis of insertion of the suture anchor [22]. A 10 N pre-load was applied in order to remove possible slack from the system and to achieve a repeatable initial test condition [21]. An actuator displacement rate of 10 mm/min was used in each test—cyclic testing was not used, in line with other studies studying anchor pullout strength [4, 5, 12]. During each test, the testing machine recorded elapsed time, tensile force and tensile displacement at 100 points per second. Marks were made on the suture using permanent marker just above the bone surface before running each test. These marks were tracked in order to confirm that any displacement measured by the testing machine was indeed due to anchor displacement within the pilot hole.
From the test data, force–displacement curves were generated, and four parameters were determined or calculated: maximum load reached during the test before or at failure, displacement occurring at maximum load, displacement at 50 N and average stiffness over the 0–50 N range. This stiffness was calculated by fitting a line to the force–displacement curve in the 0–50 N range and taking the slope of the line as the stiffness (change in force/change in extension). Failure was defined as loss of tension in the system [19, 22]. The failure mode was recorded as anchor pullout, suture breakage or eyelet failure.
In order to exclude displacement secondary to suture stretch, further testing was conducted to measure the stiffness of the suture material itself without the influence of the anchor. Loops of suture were tied in the same manner as the anchor tests and the lengths measured using vernier calipers—the loops were then mounted into the testing machine using two steel cross pins. One pin was attached to the load cell as before, and the other mounted on the machine base. The same testing protocol was used, and the test run until loss of tension was detected due to loop failure.
Pilot testing in bone analog models
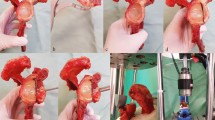
Pilot testing of the anchors was performed in a synthetic bone analog material, with a foam interior and a dense cortical bonelike shell (Fig. 2a). This material represented an idealized but highly controlled and repeatable material for initial observations regarding the mechanical behavior of the suture bone assembly.
All mechanical testing was performed using an Instron ElectroPuls E1000 mechanical machine (Instron, Canton, MA) with a 2-kN Dynacell load cell. Three groups of anchors (Bio Mini-Revo, handset Y-Knot and pre-tensioned Y-Knot) were tested each in a Bone analog model. b Bovine tibia. c Cadaveric glenoid
A total of eight Y-Knot and Bio Mini-Revo was placed in the bone analog models. Force–displacement curves for the handset Y-Knot consistently demonstrated a plateau in the curve between 30 and 50 N, with an average displacement of 4 mm (Fig. 3); this displacement corresponded to visible movement of the suture mark suggesting motion within the drill hole. Using this data, a second group of Y-Knot anchors was pre-tensioned to 60 N before testing, a force above the plateau on the force–displacement curve. Comparison of the 60 N pre-tensioned Y-Knot and the handset Y-Knot demonstrated that pre-tensioning eliminated low force displacement (Fig. 3). Using these preliminary findings, three groups of anchors were designated for testing in bovine tibial metaphysis and cadaveric glenoids: Bio Mini-Revo, handset Y-Knot and 60 N pre-tensioned Y-Knot.
Representative force displacement curves for the Bio Mini-Revo, handset Y-Knot and 60 N pre-tensioned Y-Knot in bone analog foam blocks. The handset Y-Knot curves demonstrate a significant plateau, indicating low force displacement. Pre-tensioning the Y-Knot to 60 N eliminates the low force displacement, resulting in much stiffer curves
Bone testing materials
The first stage of testing used eight bovine metaphyseal bones from steer tibia, obtained from a local abattoir; the cattle were all aged between 1 and 2 years at the time of slaughter. One of each anchor group was placed in each bovine tibia—for a total of eight of each anchor type. The proximal metaphyseal cortex was stripped of soft tissue, and each anchor type was inserted directly perpendicular to the metaphyseal surface, separated by at least 1 cm (Fig. 2b) [4].
In the second stage of testing, a total of eight fresh-frozen human male and female cadaveric glenoids was used (4 matched pairs in order to limit variations in bone density); the age of donors ranged between 50 and 90 years old. Bone density testing was not performed. After thawing to room temperature, the labrum was removed, and one of each anchor group placed in the posterior aspect of each glenoid, again separated by 1 cm (Fig. 2c). Because of the advanced age of the cadaveric glenoids in comparison with the normal population undergoing labral repair, anchors were placed in the posterior glenoid, as it is known that the bone mineral density of the posterior glenoid is increased compared to the anterior glenoid; the position of each type of anchor on the glenoid was alternated [11, 13].
Ethical considerations
Approval for the study was obtained from the Mt Sinai Research Ethics Board.
Statistical analysis
Statistical analysis was conducted using SigmaStat (v.3.1, Systat Software Inc, San Jose, CA, USA). Group sizes were based on previous studies [15] and pretesting in bone analog models. Means and standard deviations were calculated for each group. Kolmogorov–Smirnov tests were used to check for normal distributions. The Levene statistic was used to test for equality of variances. The groups were compared statistically using two-way analysis of variance with bone type and anchor type as factors. The interaction between bone type and anchor type was also examined. Multiple comparisons were conducted using Holm–Sidak post hoc testing.
Results
Eight anchors from each group were tested in both bovine bone and human glenoid, with representative force–displacement curves displayed in Fig. 4 and representative force–displacement curves in the 0–60 N range displayed in Fig. 5. In both bovine bone and human glenoid, the handset Y-Knots displayed similar behavior to that seen in the bone analog foam block; compared to the screw-in anchor, the handset Y-Knots displayed inferior fixation, early displacement and greater amounts of laxity measured before failure by pullout. Visible movement of the mark applied to the suture limbs confirmed movement of the suture anchor within the drill hole. Pre-tensioning to 60 N completely eliminated this behavior, resulting in stiffness comparable to the screw-in anchor and minimal low force displacement.
The maximum load to failure during pullout for the three anchor groups in both types of bone is seen in Table 1. In bovine bone, the Bio Mini-Revo displayed greater maximum tensile load than both the handset and the pre-tensioned Y-Knot, with no significant difference in ultimate pullout strength detected between the handset Y-Knot and the pre-tensioned Y-Knot. In glenoid bone, no significant difference in maximum pullout strength was seen between the three anchor groups.
Tensile displacement at 50 N data is presented in Table 2. In bovine and glenoid bone, the handset Y-Knot demonstrates greater tensile displacement than both the Bio Mini-Revo and the pre-tensioned Y-Knot. In bovine bone, the 60 N pre-tensioned Y-Knot displayed reduced tensile displacements compared to the Bio Mini-Revo; in cadaveric glenoid, there was no significant difference.
Mean stiffness measurements in the 0–50 N range are presented in Table 3. No significant difference was seen in stiffness of any of the anchors depending on whether they were inserted in bone analog, bovine bone or glenoid bone. However, in both bovine and glenoid bone, the Bio Mini-Revo was stiffer than the handset Y-Knot, with the pre-tensioned Y-Knot stiffer again than the Bio Mini-Revo.
The primary mode of failure in all-suture anchors was anchor pullout. No suture breakage was seen, and eyelet failure (2/8) was observed only with Bio Mini-Revo anchors inserted into the extremely dense bovine bone. Some anchor pullout was seen below the 60 N set as the pre-tensioning force. During pre-tensioning in bovine bone, five Y-Knots pulled out at forces lower than 60 N, while during pre-tensioning in the human glenoid, two Y-Knots pulled out below 60 N; three handset Y-Knots in glenoid pulled out at forces lower than 60 N. Each case of early pullout was excluded from analysis, and a new Y-Knot was reinserted via a different pilot hole and testing recommenced. This phenomenon was not seen with the Bio Mini-Revo.
In all-suture tests, the suture material itself broke by snapping close to the knot. After accounting for slight variations in loop length between tests, the tensile behavior of the loop material was highly repeatable. Normalized stiffness of the suture material (accounting for the effect of loop length) was measured to be 825 N/(mm/mm) (± 3.3 % standard error of mean) over the 0–50 N range. Therefore, it seems that pre-tensioning the Y-Knot can push the low force stiffness of the Y-Knot toward values close to the stiffness of the suture loop itself, which represents an upper bound of the possible stiffness range (a perfectly bound suture).
Discussion
The most important finding of this biomechanical study of the pullout behavior of the all-suture Y-Knot anchor was that a handset all-suture can display laxity and displacement prior to reaching forces of 50–60 N. This behavior was observed in bone analog foam block, bovine tibia and cadaveric glenoids. Importantly, pre-tensioning the Y-Knot to 60 N ensures that the anchor is well fixed, consistently eliminating laxity and displacement in both high-density bovine and lower-density cadaveric bone.
These findings are consistent with the study by Mazzocca et al. [15] who hypothesized that the JuggerKnot, another all-suture anchor, demonstrated slippage of the soft anchor through the cancellous bone at the site of fixation. It seems plausible that pre-tensioning secures the all-suture anchors either in dense cancellous bone (similar to the bovine bone) or under cortical bone in specimens where the cancellous bone may be less dense (human glenoid bone). It is unlikely that these findings can be explained by stretching of the suture material, as a visible movement of a mark on the suture limbs was observed in the handset Y-Knots was associated with early displacement. Furthermore, this behavior was not seen in pre-tensioned Y-Knots nor in the Bio Mini-Revo which uses the same #2 Hi-Fi suture.
All-suture anchors have been biomechanically tested in porcine femur, demonstrating that the JuggerKnot 1.4 had median force to failure of 239.1 N (range 215–263) [4], and the Y-Knot had a median force to failure of 151.8 N (range 135–174) [3]. In our study, the median force to failure of the Y-Knot in bovine tibial metaphysis was comparable at 140 N (range 114–166). The difference in maximum force before failure (lower mean and larger range) between the JuggerKnot and the Y-Knot may be caused by the different design of the anchors (the JuggerKnot is composed of a single No. 1 MaxBraid suture passed through a sleeve of polyester suture in a “V” configuration). The study by Barber et al. focused on maximum load to failure, using a displacement rate of 12.5 mm/s that is much greater than the 10 mm/min used in this study; this may mean that a plateau or early displacement would not be detected or observable.
The minimal fixation strength required to tolerate an early passive range-of-motion rehabilitation program after stabilization is uncertain and could be below the 40 N plateau seen on the force–displacement curves with the all-suture anchor [15]. An analysis of forces on the glenohumeral capsule demonstrated that with anterior loading, the anterior band of the inferior glenohumeral ligament carried 30 ± 21 N of force at 90° [8]. Thus, it is uncertain whether early displacement of an all-suture anchor would be encountered in the clinical setting, during the rehabilitation stage after labral surgery. However, other cadaveric research has identified that displacement of repaired tissue (including labrum) is possible after repair with suture anchors [9, 14].
A number of Y-Knots pulled out below a 60 N threshold in bovine and glenoid bone, which is concerning. Pre-tensioning seemed to have the added benefit of removing poorly functioning anchors. The exact mechanism of this early failure is unknown, but may be a function of bone density. A higher rate of early pullout was seen in the high-density bovine bone—it may be that the cancellous bone was too dense to allow the Y-Knot to expand laterally into a ball, a crucial component of obtaining fixation under the cortical bone. Conversely, low-density bone in elderly glenoids may be insufficient to hold the all-suture anchor. These biomechanical behaviors require further attention, with testing of all-sutures anchors in both young and elderly glenoids.
Limited published literature is available on the use of synthetic bone analog material in biomechanical studies of shoulder implants; synthetic humeral heads have been found to display different torsional behavior to cadaveric bone [20]. However, bone analog models of the femur, skull and vertebra have been found to have similar biomechanical behavior to bone [7, 16, 17]. Another limitation of this study is the use of cadaveric glenoids with an age range between 50 and 90, without testing of bone mineral density. While bone density testing was not performed, it could be safely assumed that the bone density of the glenoids was lower than both the bovine bone used in this study and possible glenoids in the standard population undergoing labral repair. However, all anchors were tested together in each specimen, with the handset Y-Knot consistently displaying increased displacement in all bone types. Pre-tensioning the Y-Knot eliminated this behavior, regardless of bone quality. It has previously been shown that suture anchor biomechanical performance varies with the age of cadavers and assumed that this is a function of reduced bone density [2]. In their biomechanical analysis, Mazzoca et al. [15] used cadaveric scapulas from both males and females, ranging in age between 53 and 73—testing in fact revealed minimal variation in bone mineral density. Bovine tibia is known to have a similar bone mineral density (449 mg/cm2, range 342–536) to porcine bone used in other studies, but is significantly more than human bone (178 mg/cm2, range 98–270) [1, 10].
A significant number of early pullouts were seen with the handset and pre-tensioned Y-Knots in both bovine bone and glenoid bone; these were excluded from analysis and the anchors reinserted. Including these in the analysis would have significantly decreased the maximum load to failure in both Y-Knot groups. In this study, the Y-Knot was inserted as per the manufacturer’s instructions, with an upward force applied to deploy the all-suture anchor, which is easily felt by the surgeon. It was impossible to know how much force was applied to the handset anchors or how reproducible this force was throughout testing. It could be argued that application of a greater upwards forces or that the act of knot tying itself (sliding or none sliding) may pre-tension the all-suture anchor safely above any plateau in the force–displacement curve. It is possible that some displacement could be attributed to suture stretch—however, the type of suture was a constant in all anchors used. Furthermore, the parallel vector of pull applied to the anchor is a worst-case scenario and may not replicate the more horizontal forces likely to be encountered in vivo [23].
The authors believe that this biomechanical information will be important to clinicians using all-suture anchors in labral repair. At this time, a tensiometer device for use with all-suture anchors does not exist and would require testing to ensure its suitability in clinical use. While it is also unknown if this behavior is displayed by other designs of all-suture anchor, the authors suggest when using the Y-Knot that an attempt is made to ensure adequate deployment and seating of the Y-Knot by way of a strong upward force (equivalent to lifting a 6 kg weight), before proceeding to the labral repair.
Conclusion
The Y-Knot all-suture anchor has a similar maximum load to failure compared with a screw-in anchor in human cadaveric glenoid bone. However, handset Y-Knots display some low force anchor displacement within the anchor hole, which is eliminated by pre-tensioning the all-suture anchor to 60 N.
References
Aerssens J, Boonen S, Lowet G, Dequeker J (1998) Interspecies differences in bone composition, density, and quality: potential implications for in vivo bone research. Endocrinology 139:663–670
Barber FA, Coons DA, Ruiz-Suarez M (2007) Cyclic load testing of biodegradable suture anchors containing 2 high-strength sutures. Arthroscopy 23:355–360
Barber FA, Herbert MA (2013) Cyclic loading biomechanical analysis of the pullout strengths of rotator cuff and glenoid anchors: 2013 update. Arthroscopy 29:832–844
Barber FA, Herbert MA, Hapa O, Rapley JH, Barber CA, Bynum JA, Hrnack SA (2011) Biomechanical analysis of pullout strengths of rotator cuff and glenoid anchors: 2011 update. Arthroscopy 27:895–905
Barber FA, McGarry JE, Herbert MA, Anderson RB (2008) A biomechanical study of achilles tendon repair augmentation using graft jacket matrix. Foot Ankle Int 29:329–333
Burkhart SS, Wirth MA, Simonich M, Salem D, Lanctot D, Athanasiou K (2000) Knot security in simple sliding knots and its relationship to rotator cuff repair: how secure must the knot be? Arthroscopy 16:202–207
Chong AC, Friis EA, Ballard GP, Czuwala PJ, Cooke FW (2007) Fatigue performance of composite analogue femur constructs under high activity loading. Ann Biomed Eng 35:1196–1205
Debski RE, Wong EK, Woo SL, Sakane M, Fu FH, Warner JJ (1999) In situ force distribution in the glenohumeral joint capsule during anterior-posterior loading. J Orthop Res 17:769–776
Diop A, Maurel N, Chang VK, Kany J, Duranthon LD, Grimberg J (2011) Tendon fixation in arthroscopic latissimus dorsi transfer for irreparable posterosuperior cuff tears: an in vitro biomechanical comparison of interference screw and suture anchors. Clin Biomech (Bristol, Avon) 26:904–909
Frich LH, Odgaard A, Dalstra M (1998) Glenoid bone architecture. J Shoulder Elbow Surg 7:356–361
Kalouche I, Crepin J, Abdelmoumen S, Mitton D, Guillot G, Gagey O (2010) Mechanical properties of glenoid cancellous bone. Clin Biomech (Bristol, Avon) 25:292–298
Leedle BP, Miller MD (2005) Pullout strength of knotless suture anchors. Arthroscopy 21:81–85
Lehtinen JT, Tingart MJ, Apreleva M, Warner JJ (2004) Total, trabecular, and cortical bone mineral density in different regions of the glenoid. J Shoulder Elbow Surg 13:344–348
Martetschlager F, Michalski MP, Jansson KS, Wijdicks CA, Millett PJ (2013) Biomechanical evaluation of knotless anterior and posterior Bankart repairs. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-013-2602-0
Mazzocca AD, Chowaniec D, Cote MP, Fierra J, Apostolakos J, Nowak M, Arciero RA, Beitzel K (2012) Biomechanical evaluation of classic solid and novel all-soft suture anchors for glenoid labral repair. Arthroscopy 5:642–648
McLain RF, McKinley TO, Yerby SA, Smith TS, Sarigul-Klijn N (1997) The effect of bone quality on pedicle screw loading in axial instability: a synthetic model. Spine (Phila Pa 1976) 22:1454–1460
Mullett H, O’Donnell T, Felle P, O’Rourke K, FitzPatrick D (2002) Development of a model for occipital fixation–validation of an analogue bone material. Proc Inst Mech Eng H 216:37–42
Provencher MT, Verma N, Obopilwe E, Rincon LM, Tracy J, Romeo AA, Mazzocca A (2008) A biomechanical analysis of capsular plication versus anchor repair of the shoulder: can the labrum be used as a suture anchor? Arthroscopy 24:210–216
Roth CA, Bartolozzi AR, Ciccotti MG, Wetzler MJ, Gillespie MJ, Snyder-Mackler L, Santare MH (1998) Failure properties of suture anchors in the glenoid and the effects of cortical thickness. Arthroscopy 14:186–191
Siffri PC, Peindl RD, Coley ER, Norton J, Connor PM, Kellam JF (2006) Biomechanical analysis of blade plate versus locking plate fixation for a proximal humerus fracture: comparison using cadaveric and synthetic humeri. J Orthop Trauma 20:547–554
Sileo MJ, Lee SJ, Kremenic IJ, Orishimo K, Ben-Avi S, McHugh M, Nicholas SJ (2009) Biomechanical comparison of a knotless suture anchor with standard suture anchor in the repair of type II SLAP tears. Arthroscopy 25:348–354
Sparks BS, Nyland J, Nawab A, Blackburn E, Krupp R, Burden R (2008) Cyclic loading comparison of bio-suturetak-#2 fiberwire and Bio Mini-Revo-#2 Hi-Fi suture anchor-sutures in cadaveric scapulae. Knee Surg Sports Traumatol Arthrosc 16:317–325
Wetzler MJ, Bartolozzi AR, Gillespie MJ, Roth CA, Ciccotti MG, Snyder-Mackler L, Santare MH (1996) Fatigue properties of suture anchors in anterior shoulder reconstructions: mitek GII. Arthroscopy 12:687–693
Author information
Authors and Affiliations
Corresponding author
Additional information
T. Dwyer and T. L. Willett have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dwyer, T., Willett, T.L., Dold, A.P. et al. Maximum load to failure and tensile displacement of an all-suture glenoid anchor compared with a screw-in glenoid anchor. Knee Surg Sports Traumatol Arthrosc 24, 357–364 (2016). https://doi.org/10.1007/s00167-013-2760-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-013-2760-0