Abstract
Purpose
Biological augmentation to heal a torn anterior cruciate ligament (ACL) has gained significant interest. This study examined the potential advantages of using extracellular matrix (ECM) bioscaffolds from galactosyl-α(1,3)galactose deficient pigs to heal the transected ACL.
Methods
In 16 skeletally mature goats, the ACL in the right hindlimb was transected and repaired. In 9 of these animals, an ECM sheet was wrapped around the injury site and with an ECM hydrogel injected into the transected site. The remaining 7 animals were treated with suture repair only. The left hindlimb served as a sham-operated control.
Results
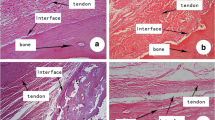
After 12 weeks, the healing ACL in the ECM-treated group showed an abundance of continuous neo-tissue formation, while only limited tissue growth was found after suture repair only. The cross-sectional area of the ACL from the ECM-treated group was similar to sham-operated controls (n.s.) and was 4.5 times those of the suture repair group (P < 0.05). The stiffness of the femur-ACL-tibia complexes from the ECM-treated group was 2.4 times those of the suture repair group (P < 0.05). Furthermore, these values reached 48% of the sham-operated controls (53 ± 19 N/mm and 112 ± 21 N/mm, respectively, P < 0.05).
Conclusions
The application of an ECM bioscaffold and hydrogel was found to accelerate the healing of a transected ACL following suture repair in the goat model with limited tissue hypertrophy and improvement in some of its biomechanical properties. Although more work is necessary to fully restore the function of the normal ACL, these early results offer a potential new approach to aid ACL healing.






Similar content being viewed by others
References
Abramowitch SD, Papageorgiou CD, Withrow JD, Gilbert TW, Woo SL-Y (2003) The effect of initial graft tension on the biomechanical properties of a healing ACL replacement graft: a study in goats. J Orthop Res 21(4):708–715
Agung M, Ochi M, Yanada S et al (2006) Mobilization of bone marrow-derived mesenchymal stem cells into the injured tissues after intraarticular injection and their contribution to tissue regeneration. Knee Surg Sports Traumatol Arthrosc 14(12):1307–1314
Anderson AF, Snyder RB, Lipscomb AB Jr (2001) Anterior cruciate ligament reconstruction. A prospective randomized study of three surgical methods. Am J Sports Med 29(3):272–279
Anitua E, Sanchez M, Orive G, Andia I (2007) The potential impact of the preparation rich in growth factors (PRGF) in different medical fields. Biomaterials 28(31):4551–4560
Badylak SF (2007) The extracellular matrix as a biologic scaffold material. Biomaterials 28(25):3587–3593
Chen G, Qian H, Starzl T et al (2005) Acute rejection is associated with antibodies to non-Gal antigens in baboons using Gal-knockout pig kidneys. Nat Med 11(12):1295–1298
Cummings JF, Grood ES (2002) The progression of anterior translation after anterior cruciate ligament reconstruction in a caprine model. J Orthop Res 20(5):1003–1008
Dai Y, Vaught TD, Boone J et al (2002) Targeted disruption of the alpha1,3-galactosyltransferase gene in cloned pigs. Nat Biotechnol 20(3):251–255
Drogset JO, Grontvedt T, Robak OR et al (2006) A 16-year follow-up of three operative techniques for the treatment of acute ruptures of the anterior cruciate ligament. J Bone Jt Surg Am 88(5):944–952
Feagin JA Jr, Curl WW (1976) Isolated tear of the anterior cruciate ligament: 5-year follow-up study. Am J Sports Med 4(3):95–100
Fisher MB, Jung HJ, McMahon PJ, Woo SL-Y (2010) Evaluation of bone tunnel placement for suture augmentation of an injured anterior cruciate ligament: effects on joint stability in a goat model. J Orthop Res 28(10):1373–1379
Freytes DO, Martin J, Velankar SS, Lee AS, Badylak SF (2008) Preparation and rheological characterization of a gel form of the porcine urinary bladder matrix. Biomaterials 29(11):1630–1637
Gilbert TW, Stolz DB, Biancaniello F, Simmons-Byrd A, Badylak SF (2005) Production and characterization of ECM powder: implications for tissue engineering applications. Biomaterials 26(12):1431–1435
Gobbi A, Bathan L, Boldrini L (2009) Primary repair combined with bone marrow stimulation in acute anterior cruciate ligament lesions: results in a group of athletes. Am J Sports Med 37(3):571–578
Iannotti JP, Codsi MJ, Kwon YW et al (2006) Porcine small intestine submucosa augmentation of surgical repair of chronic two-tendon rotator cuff tears. A randomized, controlled trial. J Bone Jt Surg Am 88(6):1238–1244
Jomha NM, Borton DC, Clingeleffer AJ, Pinczewski LA (1999) Long-term osteoarthritic changes in anterior cruciate ligament reconstructed knees. Clin Orthop Relat Res 358:188–193
Joshi SM, Mastrangelo AN, Magarian EM, Fleming BC, Murray MM (2009) Collagen-platelet composite enhances biomechanical and histologic healing of the porcine anterior cruciate ligament. Am J Sports Med 37(12):2401–2410
Kaplan N, Wickiewicz TL, Warren RF (1990) Primary surgical treatment of anterior cruciate ligament ruptures. A long-term follow-up study. Am J Sports Med 18(4):354–358
Karaoglu S, Fisher MB, Woo SL-Y et al (2008) Use of a bioscaffold to improve healing of a patellar tendon defect after graft harvest for ACL reconstruction: a study in rabbits. J Orthop Res 26(2):255–263
Kuwaki K, Tseng YL, Dor FJ et al (2005) Heart transplantation in baboons using alpha1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med 11(1):29–31
Lee TQ, Woo SL-Y (1988) A new method for determining cross-sectional shape and area of soft tissues. J Biomech Eng 110(2):110–114
Li G, Van de Velde SK, Bingham JT (2008) Validation of a non-invasive fluoroscopic imaging technique for the measurement of dynamic knee joint motion. J Biomech 41(7):1616–1622
Liang R, Fisher M, Yang G, Hall C, Woo SL-Y (2011) Alpha1,3-galactosyltransferase knockout does not alter the properties of porcine extracellular matrix bioscaffolds. Acta Biomater 7(4):1719–1727
Liang R, Woo SL-Y, Takakura Y et al (2006) Long-term effects of porcine small intestine submucosa on the healing of medial collateral ligament: a functional tissue engineering study. J Orthop Res 24(4):811–819
Livesay GA, Fujie H, Kashiwaguchi S et al (1995) Determination of the in situ forces and force distribution within the human anterior cruciate ligament. Ann Biomed Eng 23(4):467–474
Ma CB, Papageogiou CD, Debski RE, Woo SL-Y (2000) Interaction between the ACL graft and MCL in a combined ACL + MCL knee injury using a goat model. Acta Orthop Scand 71(4):387–393
Malcarney HL, Bonar F, Murrell GA (2005) Early inflammatory reaction after rotator cuff repair with a porcine small intestine submucosal implant: a report of 4 cases. Am J Sports Med 33(6):907–911
Ng GY, Oakes BW, Deacon OW, McLean ID, Lampard D (1995) Biomechanics of patellar tendon autograft for reconstruction of the anterior cruciate ligament in the goat: 3-year study. J Orthop Res 13(4):602–608
Papageorgiou CD, Gil JE, Kanamori A et al (2001) The biomechanical interdependence between the anterior cruciate ligament replacement graft and the medial meniscus. Am J Sports Med 29(2):226–231
Phelps CJ, Koike C, Vaught TD et al (2003) Production of alpha 1,3-galactosyltransferase-deficient pigs. Science 299(5605):411–414
Reing JE, Zhang L, Myers-Irvin J et al (2009) Degradation products of extracellular matrix affect cell migration and proliferation. Tiss Eng Part A 15(3):605–614. doi:10.1089/ten.tea.2007.0425
Sakane M, Livesay GA, Fox RJ et al (1999) Relative contribution of the ACL, MCL, and bony contact to the anterior stability of the knee. Knee Surg Sports Traumatol Arthrosc 7(2):93–97
Scherping SC Jr, Schmidt CC, Georgescu HI et al (1997) Effect of growth factors on the proliferation of ligament fibroblasts from skeletally mature rabbits. Connect Tiss Res 36(1):1–8
Spindler KP, Kuhn JE, Freedman KB et al (2004) Anterior cruciate ligament reconstruction autograft choice: bone-tendon-bone versus hamstring: does it really matter? A systematic review. Am J Sports Med 32(8):1986–1995
Steadman JR, Cameron-Donaldson ML, Briggs KK, Rodkey WG (2006) A minimally invasive technique (“healing response”) to treat proximal ACL injuries in skeletally immature athletes. J Knee Surg 19(1):8–13
Torry MR, Shelburne KB, Peterson DS et al (2011) Knee kinematic profiles during drop landings: a biplane fluoroscopy study. Med Sci Sports Exerc 43(3):533–541
Vorotnikova E, McIntosh D, Dewilde A et al (2010) Extracellular matrix-derived products modulate endothelial and progenitor cell migration and proliferation in vitro and stimulate regenerative healing in vivo. Matrix Biol 29(8):690–700
Warren RF (1983) Primary repair of the anterior cruciate ligament. Clin Orthop Relat Res 172:65–70
Wiig ME, Amiel D, VandeBerg J et al (1990) The early effect of high molecular weight hyaluronan (hyaluronic acid) on anterior cruciate ligament healing: an experimental study in rabbits. J Orthop Res 8(3):425–434
Woo SL-Y, Abramowitch SD, Kilger R, Liang R (2006) Biomechanics of knee ligaments: injury, healing, and repair. J Biomech 39(1):1–20
Woo SL-Y, Danto MI, Ohland KJ, Lee TQ, Newton PO (1990) The use of a laser micrometer system to determine the cross-sectional shape and area of ligaments: a comparative study with two existing methods. J Biomech Eng 112(4):426–431
Woo SL-Y, Hollis JM, Adams DJ, Lyon RM, Takai S (1991) Tensile properties of the human femur-anterior cruciate ligament-tibia complex: the effects of specimen age and orientation. Am J Sports Med 19(3):217–225
Woo SL-Y, Peterson RH, Ohland KJ, Sites TJ, Danto MI (1990) The effects of strain rate on the properties of the medial collateral ligament in skeletally immature and mature rabbits: a biomechanical and histological study. J Orthop Res 8(5):712–721
Woo SL-Y, Takakura Y, Liang R, Jia F, Moon DK (2006) Treatment with bioscaffold enhances the fibril morphology and the collagen composition of healing medial collateral ligament in rabbits. Tiss Eng 12(1):159–166
Yamada K, Yazawa K, Shimizu A et al (2005) Marked prolongation of porcine renal xenograft survival in baboons through the use of alpha1,3-galactosyltransferase gene-knockout donors and the cotransplantation of vascularized thymic tissue. Nat Med 11(1):32–34
Acknowledgments
Financial support provided by the McGowan Institute for Regenerative Medicine, Commonwealth of Pennsylvania, the National Institutes of Health (T32 EB0003392), and the National Science Foundation Engineering Research Center Grant (#0812348). The authors thank Dr. John Bianchi of Revivicor, Inc. for providing the small intestines from the GalSafe™ pigs.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fisher, M.B., Liang, R., Jung, HJ. et al. Potential of healing a transected anterior cruciate ligament with genetically modified extracellular matrix bioscaffolds in a goat model. Knee Surg Sports Traumatol Arthrosc 20, 1357–1365 (2012). https://doi.org/10.1007/s00167-011-1800-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-011-1800-x