Abstract
Purpose
This large population-based cohort study evaluated the association between certain critical illnesses and the incidence of newly diagnosed type 2 diabetes mellitus (T2DM) in Taiwan.
Methods
Data were obtained from the Taiwan National Health Insurance Research Database. According to age, sex, and propensity score-matching, a cohort comprising 9528 patients with critical illness, including septicemia, septic shock, acute myocardial infarction (AMI), and stroke, and a control cohort of 9528 patients with no critical illness were identified. Cox proportional-hazard regression and competing-risk regression models were employed to evaluate the risk of developing T2DM.
Findings
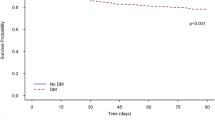
With the median follow-up periods (interquartile range) of 3.86 (1.64–6.93) and 5.12 (2.51–8.13) years for the patients in the critical illness and control cohorts, respectively, the risk of developing T2DM in the critical illness cohort was significantly higher than in the control cohort (adjusted hazard ratio, aHR = 1.32; 95 % confidence interval, CI 1.16–1.50). In the multivariate competing-risk regression models, the aHR of T2DM was 1.58 (95 % CI 1.45–1.72) in the critical illness cohort. Moreover, among the patients with these critical illnesses, those with septicemia or septic shock exhibited the highest risk of developing T2DM (aHR = 1.51, 95 % CI 1.37–1.67), followed by AMI compared with the control cohort.
Conclusion
Our results suggest that patients with certain critical illnesses are associated with a high risk of developing T2DM. Clinicians should be aware of this association and intensively screen for T2DM in patients following diagnosis of critical illness.

Similar content being viewed by others
References
Preiser JC, Ichai C, Orban JC, Groeneveld AB (2014) Metabolic response to the stress of critical illness. Br J Anaesth 113:945–954
Tamita K, Katayama M, Takagi T, Akasaka T, Yamamuro A, Kaji S, Morioka S, Kihara Y (2007) Impact of newly diagnosed abnormal glucose tolerance on long-term prognosis in patients with acute myocardial infarction. Circ J 71:834–841
Gornik I, Vujaklija A, Lukic E, Madzarac G, Gasparovic V (2010) Hyperglycemia in sepsis is a risk factor for development of type II diabetes. J Crit Care 25:263–269
Benfield T, Jensen JS, Nordestgaard BG (2007) Influence of diabetes and hyperglycaemia on infectious disease hospitalisation and outcome. Diabetologia 50:549–554
Christiansen C, Toft P, Jorgensen HS, Andersen SK, Tonnesen E (2004) Hyperglycaemia and mortality in critically ill patients. A prospective study. Intensive Care Med 30:1685–1688
Antonelli M, Bonten M, Chastre J, Citerio G, Conti G, Curtis JR, De Backer D, Hedenstierna G, Joannidis M, Macrae D, Mancebo J, Maggiore SM, Mebazaa A, Preiser JC, Rocco P, Timsit JF, Wernerman J, Zhang H (2012) Year in review in Intensive Care Medicine 2011. II. Cardiovascular, infections, pneumonia and sepsis, critical care organization and outcome, education, ultrasonography, metabolism and coagulation. Intensive Care Med 38:345–358
Eslami S, Taherzadeh Z, Schultz MJ, Abu-Hanna A (2011) Glucose variability measures and their effect on mortality: a systematic review. Intensive Care Med 37:583–593
Gornik I, Vujaklija A, Lukic E, Madzarac G, Gasparovic V (2010) Hyperglycaemia in critical illness is a risk factor for later development of type II diabetes mellitus. Acta Diabetol 47(Suppl 1):29–33
MacIntyre EJ, Majumdar SR, Gamble JM, Minhas-Sandhu JK, Marrie TJ, Eurich DT (2012) Stress hyperglycemia and newly diagnosed diabetes in 2124 patients hospitalized with pneumonia. Am J Med 125(1036):e1017–e1023
Okosieme OE, Peter R, Usman M, Bolusani H, Suruliram P, George L, Evans LM (2008) Can admission and fasting glucose reliably identify undiagnosed diabetes in patients with acute coronary syndrome? Diabetes Care 31:1955–1959
Parsons LS (2001) Performing a 1: N case-control match on propensity score. SUGI 29:11
Fine JP, Gray RJ (1999) A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 94:496–509
Harris MI, Klein R, Welborn TA, Knuiman MW (1992) Onset of NIDDM occurs at least 4–7 yr before clinical diagnosis. Diabetes Care 15:815–819
Ginde AA, Cagliero E, Nathan DM, Camargo CA Jr (2008) Point-of-care glucose and hemoglobin A1c in emergency department patients without known diabetes: implications for opportunistic screening. Acad Emerg Med 15:1241–1247
Bartnik M, Malmberg K, Norhammar A, Tenerz A, Ohrvik J, Ryden L (2004) Newly detected abnormal glucose tolerance: an important predictor of long-term outcome after myocardial infarction. Eur Heart J 25:1990–1997
McAllister DA, Hughes KA, Lone N, Mills NL, Sattar N, McKnight J, Wild SH (2014) Stress hyperglycaemia in hospitalised patients and their 3-year risk of diabetes: a Scottish retrospective cohort study. PLoS Med 11:e1001708
Boonen E, Van den Berghe G (2014) Endocrine responses to critical illness: novel insights and therapeutic implications. J Clin Endocrinol Metab 99:1569–1582
Van den Berghe G (2002) Neuroendocrine pathobiology of chronic critical illness. Crit Care Clin 18:509–528
Langouche L, Van den Berghe G (2006) The dynamic neuroendocrine response to critical illness. Endocrinol Metab Clin N Am 35:777–791
Mehta VK, Hao W, Brooks-Worrell BM, Palmer JP (1994) Low-dose interleukin 1 and tumor necrosis factor individually stimulate insulin release but in combination cause suppression. Eur J Endocrinol 130:208–214
Grimble RF (2002) Inflammatory status and insulin resistance. Curr Opin Clin Nutr Metab Care 5:551–559
Marik PE, Raghavan M (2004) Stress-hyperglycemia, insulin and immunomodulation in sepsis. Intensive Care Med 30:748–756
Montane J, Cadavez L, Novials A (2014) Stress and the inflammatory process: a major cause of pancreatic cell death in type 2 diabetes. Diabetes Metab Syndr Obes 7:25–34
Acknowledgments
Hsu CW was supported by Taipei Medical University (TMU 102-AE1-B07) and Wan Fang Hospital, Taipei Medical University (104-wf-eva-13). Lin CS was supported by grants from Tri-Service General Hospital (TSGH-C104-026) and the Taiwan Ministry of Science and Technology (MOST 103-2628-B-016-002-MY3). Kao CH was supported by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW104-TDU-B-212-113002); China Medical University Hospital, Academia Sinica Taiwan Biobank, Stroke Biosignature Project (BM104010092); NRPB Stroke Clinical Trial Consortium (MOST 103-2325-B-039 -006); Tseng-Lien Lin Foundation, Taichung, Taiwan; Taiwan Brain Disease Foundation, Taipei, Taiwan; Katsuzo and Kiyo Aoshima Memorial Funds, Japan; and CMU under the Aim for Top University Plan of the Ministry of Education, Taiwan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No additional external funding received for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors report no conflict of interest.
Additional information
C.-S. Lin and C.-H. Kao are equal contributors.
Take-home message: Our results suggest that patients with critical illness are associated with a high risk of T2DM. Clinicians should be aware of this association and screen carefully for T2DM in patients following diagnosis of critical illness.
Rights and permissions
About this article
Cite this article
Hsu, CW., Lin, CS., Chen, SJ. et al. Risk of type 2 diabetes mellitus in patients with acute critical illness: a population-based cohort study. Intensive Care Med 42, 38–45 (2016). https://doi.org/10.1007/s00134-015-4044-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-015-4044-2