Abstract
Purpose
The optimal monitoring during sleep with noninvasive positive pressure ventilation (NPPV) has not been validated in children. The aim of the study was to describe on polygraphic (PG) recordings the respiratory events and associated autonomic arousals (AA) and/or 3 % desaturations (DS3%) during nocturnal NPPV.
Methods
This was a prospective descriptive study performed in the pulmonology unit of a pediatric university hospital. Consecutive patients admitted for routine follow-up of long-term NPPV were enrolled. Nocturnal PG during sleep with NPPV was performed. A second PG was performed after adjustment of the ventilatory settings when a respiratory event occurred more than 50 times/h.
Results
The PG tracings of 39 patients (age range 1–18 years) were analyzed. Underlying diagnoses included neuromuscular disease (n = 13), obstructive sleep apnea (n = 15), and lung disease (n = 11). Unintentional leaks, patient–ventilator asynchronies, decrease in ventilatory drive, upper airway obstruction with or without reduction of ventilatory drive, and mixed events were observed in 27, 33, 10, 11, 12, and 3 % of the patients, respectively. A predominant respiratory event was observed in all patients. The mean duration spent with respiratory events was 32 ± 30 % (range 3–96 %) of total recording time. Unintentional leaks were the most frequently associated with AA, whereas patient–ventilator asynchronies were rarely associated with AA or DS3%. In eight re-evaluated patients, a decrease in the main event was observed (p = 0.005).
Conclusion
Respiratory events during sleep with NPPV are common in children treated with long-term NPPV. Consequences of respiratory events vary according to the type of event with unintentional leaks being associated preferentially with AA.
Similar content being viewed by others
Introduction
Although long-term noninvasive positive pressure ventilation (NPPV) is increasingly used in children with chronic alveolar hypoventilation, the optimal monitoring has not been validated. The main aim of NPPV is to correct nocturnal gas exchange but restricting monitoring to this therapeutic goal may not be sufficient as normal gas exchange during sleep does not exclude poor sleep quality or abnormal sleep structure [1]. Moreover, NPPV is usually started and set during the day when the patient is awake. Sleep-related changes in ventilatory drive, respiratory, and upper airway mechanics as well as respiratory muscles activity are likely to influence the efficacy and tolerance of NPPV [2]. In addition, NPPV itself may induce undesirable events and residual respiratory events may persist even under optimal NPPV settings. These events were recently described by Gonzalez-Bermejo et al. [3] and comprise unintentional leaks, patient–ventilator asynchronies, decrease in ventilatory drive, upper airway obstruction with or without reduction of ventilatory drive, and mixed events. A polygraphic (PG) or polysomnographic (PSG) recording is mandatory for the detection of these events. Although this strategy has been recommended [4], it may not be feasible on a routine basis in every pediatric NPPV center. Also, before recommending a systematic PG or PSG recording in children treated with long-term NPPV, a description of respiratory events during NPPV is necessary. Moreover, the clinical consequences of a specific event have to be taken in account. Indeed, it is possible that the potential deleterious effects may vary according to the type of NPPV-associated events and underlying disease.
Within this context, we performed a systematic sleep PG recording with NPPV in a consecutive cohort of children treated with long-term NPPV at home. The aims of the study were first to describe the respiratory events according to the SomnoNIV group definitions [3] and second to analyze each event’s association with an autonomic micro-arousal (AA) and/or a 3 % oxyhemoglobin desaturation (DS3%) [5, 6].
Materials and methods
Patients
All consecutive patients treated with home nocturnal NPPV were prospectively enrolled during their routine follow-up between October 2011 and April 2012 if (1) they were in a stable condition, (2) they were using NPPV for at least 1 month, and if (3) they had no clinical symptoms of nocturnal hypoventilation such as frequent arousals, nocturnal agitation, or daytime fatigue and sleepiness. Patients were divided into a neuromuscular (NM) disease group, a lung disease group, and an obstructive sleep apnea syndrome (OSAS) group. The study was conducted in agreement with the French regulations and had received appropriate legal and ethical approval from the Institutional Review Board of the French Learned Society for Respiratory Medicine (Société de Pneumologie de Langue Française).
The home NPPV program
Criteria for long-term NPPV were a maximal nocturnal transcutaneous carbon dioxide tension (PtcCO2) greater than 50 mmHg and/or a minimal pulse oximetry (SpO2) less than 90 % for at least 2 % of night-time [7, 8]. All the patients were ventilated with a pressure-targeted mode, with [“pressure support–volume targeted ventilation (PS-VT)”] or without (“bilevel ventilation”) a target volume, with a back-up respiratory rate depending on the age of the patient. The interface was a nasal or face mask, with or without a manufacturer leak, according to the ventilatory mode and settings and patient’s tolerance [9]. Objective compliance was assessed by the ventilator’s software. None of the patients included in the study required supplemental oxygen therapy during NPPV.
Study design and procedure
Overnight recordings of nocturnal PG, gas exchange, and actigraphy were performed during a single night hospitalization while respecting the child’s habitual sleep. Synchronization was ensured between the devices.
Nocturnal ventilatory PG
An overnight PG assessment was performed using the Somnoscreen device (SOMNOscreen TM plus PSG+, SOMNOmedics GmbH, Germany) which is an adaptable PG device that can be connected to a PtcCO2 monitor (SenTec Digital Monitor, SenTec Inc, Therwil, Switzerland). The recorded data included airflow using a pneumotachograph, airway pressure included in the NPPV line, body position, thoracic and abdominal movements assessed with piezoelectric belts (SleepSense, Multiple use Inductive Plethysmography Band, S.L.P. Ltd., Israel), SpO2 and photoplethysmographic pulse wave amplitude (PWA). The pneumotachograph was inserted between the interface and the expiratory valve in case of a non-leak ventilation or between the interface (with an integrated manufacturer leak) and the circuit in case of a leak ventilation.
AA were identified as a reduction in PWA greater than 30 % of the baseline amplitude [10, 11]. Mean and minimal nocturnal SpO2 as well as the percentage of night-time with mean SpO2 less than 90 % were determined. Desaturation index was defined as the number of SpO2 desaturations above 3 % from baseline value per hour of PG recording (DS3%) and AA related to a DS3% (AA + DS3%) were quantified [5, 6, 10].
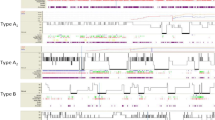
Respiratory events (see online supplement)
A respiratory event is characterized by the occurrence of a modification, discontinuity, or instability of ventilation. Respiratory events were defined and scored using the SomnoNIV Group definitions and quantified as the number of events per PG recording time [3]. A single respiratory event was defined as an event lasting at least two respiratory cycles, which varied according to the age and respiratory rate of the patient [12]. When a combination of several events occurred, we checked first for non-intentional leaks. Continuous leaks were systematically scored as the predominant event. In the absence of continuous leaks, the predominant event was assumed to be the most frequent event encountered solely. As an example, if a PG recording displayed numerous upper airway obstructions with increase in respiratory drive, associated from time to time with asynchrony, the predominant event was assumed to be the obstruction and asynchrony the consequent event. Respiratory events associated with a DS3%, an AA, or an AA + DS3% were also quantified by means of the pulse wave amplitude (PWA) [5, 6].
Six types of respiratory events were identified (see online supplement for complete description): unintentional leaks, patient–ventilator asynchrony, decrease in ventilatory drive, upper airway obstruction with or without reduction of ventilatory drive, and mixed events.
Nocturnal carbon dioxide monitoring
An overnight PtcCO2 recording was performed by the combined SpO2/PtcCO2 monitor (SenTec Digital Monitor, SenTec Inc, Therwil, Switzerland) [13]. The mean, maximal PtcCO2, and the percentage of night-time with a PtcCO2 greater than 50 mmHg data were gathered.
Overnight actigraphy
An overnight actigraphy recording was performed simultaneously (Actiwatch, Cambridge Neurotechnology Ltd., Cambridge, UK) [14, 15]. Sleep efficiency was defined as the percentage of time spent asleep whilst in bed. The movement and fragmentation index (MFI) was defined as the sum of the minutes of movement expressed as a percentage of the total recording time (movement index) and the number of times that sleep was terminated after 1 min expressed as a percentage of the total estimated sleep time (fragmentation index) [16–18]. The sum was expressed as a percentage.
Capillary arterialized blood gases
Capillary arterialized blood gases were performed in the morning following the sleep study [19].
Re-evaluation PG study after setting adjustments
When the predominant respiratory event exceeded 50 events/h on the baseline PG recording, an adjustment of the NPPV setting was performed and a second nocturnal PG was performed after 2–4 weeks.
Statistical analysis
Data are presented as mean ± SD. Baseline characteristics and rate of respiratory events associated or not with DS3% or AA were compared among the three groups using a one way analysis of variance or a Kruskal–Wallis one-way analysis of variance on ranks if necessary. A Chi-square analysis was performed to compare the distribution of the different respiratory events associated or not with a DS3% or an AA. Comparisons of data before and after resetting of NPPV were done using a paired t test or Wilcoxon signed rank test when appropriate. The correlation of the respiratory event indexes with nocturnal carbon dioxide monitoring, actigraphy, and capillary arterialized blood gases was performed using the Pearson correlation test or Spearman correlation test when appropriate. A p value of less than 0.05 was considered statistically significant.
Results
Thirty-nine patients with NM disease (n = 13), lung disease (n = 11), or OSAS (n = 15) were included in the study (Table 1). The mean age of the total population was 10.0 ± 5.8 years with no difference between the three groups. All the patients with an NM or a lung disease were ventilated with a PS-VT mode and all the patients but one with OSAS were ventilated with a bilevel ventilation mode. All the patients used a single circuit coupled either with an expiratory valve in eight patients or an intentional leak valve in the remaining patients. For the total population, the mean inspiratory positive airway pressure was 14.3 ± 2.2 cmH2O. Eight patients had no expiratory positive airway pressure set; the mean expiratory positive airway pressure was 4.2 ± 2.8 cmH2O for the other 31 patients. The patients with lung disease had a significantly lower mean SpO2 and higher maximal PtcCO2 during NPPV as compared to the two other groups with no difference in daytime blood gases (Table 1). Sleep efficiency was within the normal limits among the three groups with a fragmentation index slightly above normal values [20–22]. Compliance with NPPV was good and not different between the three groups (Table 1).
Table 2 shows the scoring of the PG tracings for the total population and among the three groups. Respiratory events were extremely common and all the patients had at least one. Patient–ventilator asynchrony and unintentional leaks were the most common events in the patients with NM or lung disease, whereas unintentional leaks, decrease in ventilatory drive, and upper airway obstruction with or without reduction of ventilatory drive were almost equally observed in the OSAS group.
For a given patient, there was a predominant respiratory event representing 75 ± 19 % of total time spent in respiratory events. The mean duration spent in a respiratory event was 32 ± 30 % (range 3–96 %) of total recording time.
Figure 1 shows the association between the different respiratory events and a DS3%, an AA, or an AA + DS3% for the total population, the NM disease group, the lung disease group, and the OSAS group. Unintentional leaks were very frequent. The majority of these unintentional leaks were associated with AA in the total population, the NM and OSAS groups but not in the lung disease group. Patient–ventilator asynchronies were also very common but conversely, the vast majority of these asynchronies were not associated with an AA or a DS3%, except for the OSAS group. Upper airway obstruction with decrease in ventilatory drive, although not common, was frequently associated with an AA + DS3% in the total population and particularly in patients with lung disease or OSAS. Decrease in ventilatory drive was frequently associated with a DS3% in NM patients.
Incidence of respiratory events and their association with autonomic arousals (AA), 3 % desaturation (DS3%) or autonomic arousals with 3 % desaturation (AA + DS3%), in the total population, the patients with neuromuscular disease, lung disease, and obstructive sleep apnea syndrome (OSAS). UL unintentional leaks, AS patient–ventilator asynchronies, UAO + DV upper airway obstruction with decrease in ventilatory drive, UAO upper airway obstruction, DV decrease in ventilatory drive, Mixed mixed events. *p < 0.05 between “No clinical consequences” and “3%DS”, “AA”, and “AA + DS3%”, # p < 0.05 between “No clinical consequences” and “3%DS”, and “AA” § p < 0.05 between “No clinical consequences” and “3%DS”
No correlation was observed between the frequency of any respiratory event and the actigraphic sleep efficiency and fragmentation index, daytime arterial blood gases, mean and maximal nocturnal PtcCO2, or the objective compliance with NPPV.
In eight patients, a second PG during NPPV was performed after the adjustment of the ventilatory settings aimed at reducing the predominant respiratory event (patient–ventilator asynchrony in 5 patients, unintentional leaks in 2 patients, and decrease in ventilatory drive in 1 patient) (Fig. 2). A significant reduction in the main respiratory event was observed on the second PG tracing (p = 0.005).
Discussion
This study is the first to analyze the PG recordings during sleep in children treated with long-term NPPV and to analyze the associated DS3% and AA. The main observations of the study are that respiratory events are extremely common with each patient harboring a predominant event; unintentional leaks were most frequently associated with an AA, whereas patient–ventilator asynchronies were rarely associated with an AA or a DS3%; and finally, the PG recording was helpful to adapt the ventilatory settings in order to reduce the predominant respiratory event.
The optimal monitoring of patients treated with long-term NPPV is still a matter of debate. There is agreement on SpO2 recording as a minimal requisite but two studies from our group have shown that overnight PtcCO2 is also mandatory as patients may remain hypercapnic despite normal SpO2 levels [13, 23]. Some authors recommend a systematic PSG as this investigation represents the gold standard for the qualitative and quantitative diagnosis of sleep abnormalities [4]. But PSG is time-consuming, expensive, technically demanding, and not feasible on a routine basis. Moreover, the analysis of the respiratory events during sleep with NPPV on a PSG requires an extensive knowledge not only of the patient’s sleep but also of the setting and functioning of the ventilator and the patient–ventilator interaction [2, 3]. Within this context, a descriptive analysis of all respiratory events observed during long-term NPPV in stable children is a preliminary and essential step.
The main observations of the present study are that respiratory events are common, even in compliant children who do not complain of any residual sleep-disordered symptom, with unintentional leaks and asynchrony representing the most common events. Reports in the literature have mainly dealt with these two types of respiratory events. In a recent systematic study in adults, Rabec et al. [24] also found that unintentional leaks represent the most frequent event occurring under NPPV. Unintentional leaks in adult patients with NM disease receiving noninvasive or invasive ventilation have been shown to be associated with persistent daytime hypercapnia [25]. In the present study, no association was observed between the occurrence of unintentional leaks and daytime PaCO2 levels. This may be explained by the fact that daytime hypercapnia, defined as a PaCO2 greater than 45 mmHg, was observed only in five patients and that the volume of leaks was not calculated. In a step by step strategy monitoring during sleep, the occurrence and the severity of leaks can be addressed firstly by the use of the devices’ built-in software [1, 26]. When leaks are detected some simple technical adjustments can be proposed before a PG or PSG [24, 27]. The incidence and consequences of one type of patient–ventilator asynchrony, i.e., ineffective inspiratory efforts (IIE), have been analyzed in 48 adults enrolled in a long-term home NPPV program [28]. A positive correlation was observed between the IIE index and the time spent with an SpO2 less than 90 %, with the patients with an IIE index above 100 having a poor nocturnal gas exchange. Conversely, patient–ventilator asynchrony was not found to be associated with a significant SpO2 desaturation or an increase in PtcCO2 in another study with adult patients with obesity hypoventilation [29]. Again, a comparison with our results is difficult because of the small number of patients with an SpO2 less than 90 % (n = 2) under NPPV.
The clinical consequences of respiratory events occurring during sleep with NPPV represent the major issue. If the respiratory event is not associated with any deterioration in gas exchange or sleep architecture and quality and does not influence the patient’s compliance with NPPV, its correction and thus detection are not mandatory. It is thus essential to detect and quantify the most deleterious respiratory events, i.e., those associated with poor subjective or objective sleep quality, gas exchange, and/or compliance. We chose to analyze the AA and SpO2 for the following reasons. PSG with electroencephalographic (EEG) recording was not performed in the present study, precluding the identification of EEG defined micro-arousals. As arousals are recognized as a major cause of poor sleep quality, we took the option to use the PWA and actigraphic data to estimate the AA and the awakenings as these two tools have already been validated [5, 10, 11, 15, 30]. Moreover, in children, the arousal threshold is higher than in adults and we have previously demonstrated that autonomic activation is more sensitive than EEG to identify sleep disruption associated with abnormal respiratory events [31]. We observed that the majority of the unintentional leaks were associated with AA in the patients with NM disease and OSAS (Fig. 1). Conversely, patient–ventilator asynchronies, although being very common, were rarely associated with an AA or a DS3%. The consequences of the respiratory events on the PtcCO2 changes were not analyzed in the present study because time-course changes in PtcCO2 are more progressive and less variable than SpO2 changes [27] and because the large majority (n = 34) of the patients had a maximal PtcCO2 less than 50 mmHg during sleep.
Our study has several limitations. The PG recording during NPPV was informative and allowed one to decrease the predominant residual respiratory event in the subgroup of patients reassessed after setting changes. As there is no validated cutoff for respiratory events during NPPV in children, we took arbitrarily a cutoff of 50 events/h because this number emerged as being very high. However, clinically relevant cutoff values for each respiratory event in the different pediatric populations need to be validated in future studies. We used the PWA to identify AA associated or not with a desaturation. We acknowledge that the use of PWA as a marker of AA has not been validated against changes in cortical electroencephalographic activity during NPPV, but a recent study from our group showed the usefulness of this marker to assess the efficacy of NPPV during sleep in children [6]. As underlined by the SomnoNIV Group, the flow and pressure signals during NPPV vary according to the ventilatory modes, the type of circuit, and the ventilatory settings [3, 32]. Seventy percent of the patients in the present study used a PS-VT mode, during which the ventilator is estimated to increase the inspiratory pressure in order to maintain a minimal tidal volume. The use of this new mode may add an additional “ventilator” factor that has not been described for the more “classical” volume or pressure targeted modes by the SomnoNIV Group [3, 32].
In conclusion, this study reports the high frequency of respiratory events during sleep with NPPV in stable children treated with long-term NPPV at home. Unintentional leaks and patient–ventilator asynchronies were the most common events but the latter were well tolerated with regard to AA and DS3%. As PG recording of sleep during NPPV requires a high level of expertise, future studies should assess which step by step strategy should be proposed for the monitoring of NPPV [26] with the final goal to reduce the number of hospitalizations for NPPV in children.
References
Adler D, Perrig S, Takahashi H, Espa F, Rodenstein D, Pépin JL, Janssens JP (2012) Polysomnography in stable COPD under non-invasive ventilation to reduce patient-ventilator asynchrony and morning breathlessness. Sleep Breath 16:1081–1090
Pépin JL, Borel JC, Janssens JP, Tamisier R, Levy P (2008) Sleep and NIV: monitoring of the patient under home ventilation. Eur Respir Monogr 41:350–366
Gonzalez-Bermejo J, Perrin C, Janssens JP, Pepin JL, Mroue G, Léger P, Langevin B, Rouault S, Rabec C, Rodenstein D, SomnoNIV Group (2012) Proposal for a systematic analysis of polygraphy or polysomnography for identifying and scoring abnormal events occurring during non-invasive ventilation. Thorax 67:546–552
Finder JD et al (2004) Respiratory care of the patient with Duchenne muscular dystrophy: ATS consensus statement. Am J Respir Crit Care Med 170:456–465
Delessert A, Espa F, Rossetti A, Lavigne G, Tafti M, Heinzer R (2010) Pulse wave amplitude drops during sleep are reliable surrogate markers of changes in cortical activity. Sleep 33:1687–1692
Ramirez A, Khirani S, Delord V, Aubertin G, Pépin JL, Fauroux B (2012) Assessment of sleep quality by pulse wave amplitude and actigraphy in children with sleep-disordered breathing: evaluation at diagnosis and under non-invasive ventilation. Sleep Breath. doi:10.1007/s11325-012-0771-6
Hukins CA, Hillman DR (2000) Daytime predictors of sleep hypoventilation in Duchenne muscular dystrophy. Am J Respir Crit Care Med 161:166–170
Ward S, Chatwin M, Heather S, Simonds AK (2005) Randomised controlled trial of non-invasive ventilation (NIV) for nocturnal hypoventilation in neuromuscular and chest wall disease patients with daytime normocapnia. Thorax 60:1019–1024
Ramirez A, Delord V, Khirani S, Leroux K, Cassier S, Kadlub N, Aubertin A, Picard A, Fauroux B (2012) Interfaces for long-term noninvasive positive pressure ventilation in children. Intensive Care Med 38:655–662
Levy P, Pepin JL (2003) Sleep fragmentation: clinical usefulness of autonomic markers. Sleep Med 4:489–491
Haba-Rubio J, Darbellay G, Hermann FR, Frey JG, Fernandes A, Vesin JM, Thiran JP, Tschopp JM (2005) Obstructive sleep apnea syndrome: effect of respiratory events and arousal on pulse wave amplitude measured by photoplethysmography in NREM sleep. Sleep Breath 9:73–81
Iber C, Ancoli-Israel S, Chesson AL, Quan SF, AAoS (2007) The AASM manual for the scoring of sleep and associated events. American Academy of Sleep Medicine, Westchester
Paiva R, Krivec U, Aubertin G, Cohen E, Clément A, Fauroux B (2009) Carbon dioxide monitoring during long-term noninvasive respiratory support in children. Intensive Care Med 35:1068–1074
Van Someren EJW, Lazeron RHC, Vonk BFM, Mirmiran M, Swaab DF (1996) Gravitational artefact in frequency spectra of movement acceleration: implications for actigraphy in young and elderly subjects. J Neurosci Methods 65:55–62
Sadeh A, Acebo C (2002) The role of actigraphy in sleep medicine. Sleep Med Rev 6:113–124
Kushida CA, Chang A, Gadkary C, Guilleminault C, Carrillo O, Dement WC (2001) Comparison of actigraphic, polysomnographic, and subjective assessment of sleep parameters in sleep-disordered patients. Sleep Med Rev 2:389–396
Sadeh A, Acebo C (2002) The role of actigraphy in sleep medicine. Sleep Med Rev 6:113–124
Morgenthaler TI, Lee-Chiong T, Alessi C, Friedman L, Aurora RN, Boehlecke B, Brown T, Chesson ALJ, Kapur V, Maganti R, Owens J, Pancer J, Swick TJ, Zak R (2007) Practice parameters for the clinical evaluation and treatment of circadian rhythm sleep disorders. An American Academy of Sleep Medicine report. Sleep 30:1445–1459
Gaultier C, Boulé M, Allaire Y, Clément A, Burry A, Girard F (1978) Determination of capillary oxygen tension in infants and children: assessment of methodology and normal values during growth. Bull Eur Physiopath Resp 14:287–294
Lehnkering H, Strauss A, Wegner B, Siegmund R (2006) Actigraphic investigations on the activity-rest behavior of right- and left-handed students. Chronobiol Int 23:593–605
Chae KY, Kripke DF, Poceta JS, Shadan F, Jamil SM, Cronin JW, Kline LE (2009) Evaluation of immobility time for sleep latency in actigraphy. Sleep Med Rev 10:621–625
Weiss AR, Johnson NL, Berger NA, Redline S (2010) Validity of activity-based devices to estimate sleep. J Clin Sleep Med 6:336–342
Nardi J, Prigent H, Adala A, Bohic M, Lebargy F, Quera-Salva MA, Orlikowski D, Lofaso F (2012) Nocturnal oximetry and transcutaneous carbon dioxide in home-ventilated neuromuscular patients. Respir Care ;57:1425–1430
Rabec C, Georges M, Kabeya NK, Baudouin N, Massin F, Reybet-Degat O, Camus P (2009) Evaluating noninvasive ventilation using a monitoring system coupled to a ventilator: a bench-to-bedside study. Eur Respir J 34:902–913
Gonzalez J, Sharshar T, Hart N, Chadda K, Raphael JC, Lofaso F (2003) Air leaks during mechanical ventilation as a cause of persistent hypercapnia in neuromuscular disorders. Intensive Care Med 29:596–602
Contal O, Vignaux L, Combescure C, Pepin JL, Jolliet P, Janssens JP (2012) Monitoring of noninvasive ventilation by built-in software of home bilevel ventilators: a bench study. Chest 141:469–476
Janssens JP, Borel JC, Pépin JL, SomnoNIV Group (2011) Nocturnal monitoring of home non-invasive ventilation: the contribution of simple tools such as pulse oximetry, capnography, built-in ventilator software and autonomic markers of sleep fragmentation. Thorax 66:438–445
Fanfulla F, Taurino AE, Lupo ND, Trentin R, D’Ambrosio C, Nava S (2007) Effect of sleep on patient/ventilator asynchrony in patients undergoing chronic non-invasive mechanical ventilation. Respir Med 101:1702–1707
Guo YF, Sforza E, Janssens JP (2007) Respiratory patterns during sleep in obesity-hypoventilation patients treated with nocturnal pressure support: a preliminary report. Chest 131:1090–1099
Catcheside PG, Chiong SC, Mercer J, Saunders NA, McEvoy RD (2002) Noninvasive cardiovascular markers of acoustically induced arousal from non-rapid-eye-movement sleep. Sleep 25:797–804
Pépin JL, Delavie N, Pin I, Deschaux C, Argod J, Bost M, Levy P (2005) Pulse transit time improves detection of sleep respiratory events and microarousals in children. Chest 127:722–730
Rabec C, Rodenstein D, Leger P, Rouault S, Perrin C, Gonzalez-Bermejo J, SomnoNIV Group (2012) Ventilator modes and settings during non-invasive ventilation: effects on respiratory events and implications for their identification. Thorax 66:170–178
Acknowledgments
The authors gratefully thank the “Scientific Council Agiradom” for their support. The research of Brigitte Fauroux is supported by the Association Française contre les Myopathies (AFM), Vaincre la Mucoviscidose (VLM), Assistance Publique-Hôpitaux de Paris, Inserm, ADEP Assistance, and Université Pierre et Marie Curie-Paris 6. V. Caldarelli is a recipient of a fellowship Fondazione Fornasini di Poggio Renatico.
Conflicts of interest
None of the authors has any conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.-L. Pépin is a member of the SomnoNIV group.
V. Caldarelli recipient of a fellowship Fondazione Fornasini di Poggio Renatico.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Caldarelli, V., Borel, J.C., Khirani, S. et al. Polygraphic respiratory events during sleep with noninvasive ventilation in children: description, prevalence, and clinical consequences. Intensive Care Med 39, 739–746 (2013). https://doi.org/10.1007/s00134-012-2806-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-012-2806-7