Abstract
Objective
We investigated the efficacy of locally applied nasal decongestant agents and corticosteroids for preventing nosocomial maxillary sinusitis in mechanically ventilated patients with multiple trauma.
Design and setting
A prospective, open-label randomized study in two intensive care units (ICUs).
Patients
79 consecutive multiple trauma patients admitted to the ICU who were expected to be mechanically ventilated for more than 3 days.
Interventions
Patients were randomly assigned to receive either a combination of a locally applied nasal decongestant agents: 2 drops twice/day of xylometazoline nasal solution 0.1% and 100 μg budesonide (NDCA group, n = 39) or placebo (control group, n = 40).
Measurements
For the diagnosis of radiological maxillary sinusitis patients underwent paranasal computed tomography within 48 h of admission and thereafter every 4–7 days. Infectious maxillary sinusitis was diagnosed by microbiological analysis of fluid aspirated after transnasal puncture of maxillary sinuses.
Results
Radiological maxillary sinusitis was detected in 54% of patients in the NDCA group (n = 21) but in 82% of controls (p < 0.01), and infectious maxillary sinusitis in 8% of the NDCA group (n = 3) but in 20% of controls (n = 8; p = 0.11). The most common pathogen micro-organisms identified from maxillary aspirates were Acinetobacter (32%) followed by anaerobes (21%).
Conclusion
Our results indicate that the combination of locally applied xylometazoline hydrochloride and budesonide reduces the incidence of radiological maxillary sinusitis and may reduce also that of nosocomial maxillary sinusitis in mechanically ventilated patients with multiple trauma.
Similar content being viewed by others
Introduction
Nosocomial maxillary sinusitis (NMS) has recently been described as an important occult source of nosocomial infection and should be investigated in patients with fever of unknown origin [1, 2, 3, 4]. The incidence of NMS in ICU patients varies widely, ranging from 0–100%, probably due to different diagnostic criteria applied and to different populations studied [5]. It is highest in neurosurgical patients and lowest in medical and surgical ICU patients [6]. However, the rate of infectious maxillary sinusitis (IMS), based on stringent diagnostic criteria (using antroscopy, histopathological, and microbiological examination of the antral mucosa), may be only less than 10% among critically ill patients with radiological maxillary sinusitis (RMS) [2, 7]. Furthermore, no consensus on therapeutic and preventive interventions has been established. Recommendations for prevention include removal of long-term nasopharyngeal and nasogastric tubes, semirecubent positioning, and strict adherence to well-known measures of hospital hygiene (hand disinfection, use of aseptic solutions for mouth care, etc.) [8, 9]. The use of pharmacological agents such as topical decongestants or α-adrenergic agonists such as phenylephrine and antihistamines to reduce inflammation may have a beneficial effect in preventing NMS but have not yet been tested.
This study examined the efficacy of a locally applied combination of a nasal decongestant agent, xylometazoline hydrochloride, and a corticosteroid, budesonide, for reducing the incidence of NMS in mechanically ventilated patients with multiple traumas.
Patients and methods
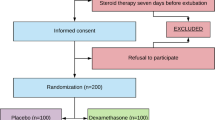
This open-label randomized study was carried out between January 2003 and April 2005 in the seven-bed intensive care unit (ICU) of the General District Hospital of Arta and the ten-bed ICU of the University Hospital of Alexandroupolis and included all 93 patients admitted with multiple trauma and expected duration of mechanical ventilation longer than 3 days. Exclusion criteria included facial trauma associated with hemosinus on admission and coagulation disorders contraindicating transnasal puncture; 14 patients were excluded (6 early extubation, 6 facial trauma with hemosinus, and 2 coagulation disorder). The remaining 79 patients were randomly allocated to the study group receiving nasal decongestant agents (NDCAs, n = 39; 33 men, 6 women; age 42 ± 18 years) or the control group (n = 40; 31 men, 9 women; age 39.8 ± 17 years). A computer program was used to provide randomization between the two compared groups with blocks of four patients (Fig. 1). No marked imbalance was observed between the two groups (Table 1).
Soon after admission patients allocated to study group started treatment by local NDCA consisting of 2 drops of xylometazoline hydrochloride (Otrivin nas sol 0.1% Novartis, Basel, Switzerland) administered to each nostril twice daily. One week after starting the therapy with xylometazoline we added 50 μg budesonide twice daily (Pulmicort m.d. nasal spray, Astra, Sweden) to prevent rhinitis medicamentosa (also known as rebound rhinitis). The administration of xylometazoline was continued for a total of 14 days or was stopped if patients already were weaned from the ventilator and nasogastric tube was removed. Budesonide continued to be given for 1 additional week after xylometazoline withdrawal. Before treatment began, a careful irrigation of both nostrils using instillation of isotonic saline 0.9% followed by aspiration of nasal secretions was performed in each patient in treated group.
For the diagnosis of RMS all patients underwent paranasal computed tomography within 48 h of admission and thereafter every 4–7 days; 129 scans were carried out in the treatment group (3.3 per patient) and 136 in the control group (3.4 per patient). RMS defined as either total opacification of one or both maxillary sinuses or as the presence of an air fluid level in one or both maxillary sinuses. For confirmation of IMS transnasal puncture was performed by an otorhinolaryngologist using a standardized protocol: A large disinfection of the nasal mucosa using povidone-iodine solution (Betadine ORL, Mundipharma, Switzerland) was performed. Then general anesthesia was induced using a combination of fentanyl and midazolam. Transnasal puncture was then performed using an Albertini trocar placed below the inferior turbinate and sinus content was directly aspirated and immediately transported for bacteriological examination. A drain was inserted in all punctured patients. Cultures of sinus aspirate were performed on the following plates: blood agar for aerobic and anaerobic cultures, chocolate agar for culture in CO2 (5%) incubator, and Sabouraud's agar. Infectious maxillary sinusitis defined as a positive culture of sinus aspirate with more than 103 cfu/ml. This definition of IMS has been used in previous studies [1, 2]. In patients receiving antibiotics IMS defined as the presence of six or more altered polymorphonuclear leukocytes per oil immersion with or without positive culture of the sinus aspirate.
Ventilator-associated pneumonia (VAP) was suspected in the presence of new and persistent pulmonary infiltrates in addition to two of the following criteria: body temperature higher than 38.3°C, leukocytosis (> 12,000 leukocytes/mm3) or leukopenia (< 4,000 leukocytes/mm3), and purulent tracheal secretions. The diagnosis of VAP was confirmed by quantitative cultures: a bronchoscopic or blind protected sample was taken on the day of suspicion of pneumonia by a specific double catheter set (Compicath Laboratoire Plastimed, Saint-Leu-La Foret, Cedex, France) as described in detail elsewhere [10]. A growth of 103 cfu/ml or more is considered significant, corresponding to an initial concentration of 105–106 bacteria/ml in the retrieved secretions.
All patients had a nasogastric or orogastric tube in place and the body position was, if possible, semirecumbent at a 30–45° angle. In the NDCA group 30 patients had a nasogastric tube and 9 an orogastric tube; in the control group 28 had a nasogastric tube and 12 an orogastric tube. Sixteen patients were extubated before the first week: six in the NDCA group and ten in the control group. The local ethics committees approved the study, and informed consent was obtained from the patients relatives before inclusion.
All data are expressed as mean ± SD. Qualitative variables were compared by χ2 test and continuous variables by Student's t test. We estimated that about 40 patients per group would be needed for a statistical significance between two groups, assuming at least a 10% reduction in NMS with a power of about 0.8 and α = 0.05. Differences were considered significant at p < 0.05.
Results
As shown in Table 2, RMS was detected in 53.8% of patients in the NDCA group (n = 21) and in 82.5% of those in the control group (n = 33; p < 0.01) and IMS, respectively, in 7.7% (n = 3) and 20% (n = 8; p = 0.11). Sixteen pathogen micro-organisms were isolated from maxillary aspirates in control patients but from only six of those in the NDCA group. The most commonly identified were Acinetobacter (32%), Staphylococcus aureus (21%), Pseudomonas aeruginosa (21%), and anaerobes (21%). No anaerobe micro-organisms were isolated from maxillary aspirates in patients in the NDCA group (Table 3). Following maxillary drainage the signs of sepsis resolved in 27.7% of patients with infectious maxillary sinusitis (n = 3). VAP was detected in 15.4% of patients in the NDCA group (n = 6) and in 27.5% of those in the control group (n = 11; NS). Two patients in the NDCA group and four in the control group developed VAP due to the same micro-organism as in infectious maxillary sinusitis (Fig. 1).
Discussion
This randomized open-label control study of mechanically ventilated patients with multiple traumas found that the combination of locally applied NDCA and corticosteroids reduced the incidence of RMS by 28.7% (p < 0.01) and that of IMS by 13.3% (p = 0.11). Although the altered incidence of IMS did not reach statistical significance, this could nevertheless be of clinical significance.
Nosocomial maxillary sinusitis is a closed-space infection that should be treated as such. The pathogenesis of NMS is probably multifactorial in many cases. As demonstrated in experimental sinusitis, the presence of a foreign body in the nostrils creates an ostial obstruction and an edema of the ostiomeatal complex that initiates sinus inflammation and mucus accumulation in the sinus cavity. In the absence of foreign body in the nostrils the ostial size varies from one patient to another [11]. Supine position reduces ostial patency by inducing swelling of the ostiomeatal complex [12]. A functional ostial area smaller than 5 mm2 induces hypoventilation of the maxillary cavity, local hypoxia, and decreased mucosal blood flow and predispose to maxillary sinusitis by impairing mucocilliary clearance [13]. Moreover, sepsis originating in other organs may lead to reduction in antral nitric oxide concentrations contributed to impaired mucocilliary clearance and decreased perfusion of the maxillary epithelium, both factors that facilitate infection of the maxillary cavity by impairing bacterial cleansing [14].
Rouby et al. [4] have suggested that NMS is secondary to ostial diminution caused by positioning or obstruction in cases of nasal intubation. Other reported risk factors include the presence of nasogastric tube, nasal packing, facial and cranial fractures, sedation, and unconsciousness [15, 16, 17]. In critically ill patients the mucus of the paranasal sinuses is directed toward the ostia for drainage against gravity because of the recumbent position. There have also been reports that limited head mobility may play a role in the development of sinusitis as evidenced by the higher incidence of this disease among patients with head trauma [18]. Moreover, drainage of the sinuses through the ostia in the lateral nasal walls is reduced as a result of physical irritation and local trauma causing inflammation and edema [8].
Our study used a combination of topical NDCAs xylometazoline hydrochloride and budesonide. Xylometazoline hydrochloride is an imidazoline derivative with marked α-adrenergic action acting as a vasoconstrictor when applied topically to mucous membrane [19]. It is commonly used in topical application to relieve nasal congestion associated with acute or chronic rhinitis, common cold, and hay fever and other allergies. It is also frequently used in treating community acquired sinusitis to reduce the mucosal edema and thus facilitate drainage of the sinuses. Xylomethazoline has also been hypothesized to have an additional beneficial effect in the topical treatment of nasal inflammation due to its antioxidant properties [20]. The effect of xylometazoline begins within 5–10 min of application and lasts for up to 10 h. When used for short periods of time at low doses, side effects are usually rare. However, it is well known that long-term use of topical vasoconstrictors for the nose may result in rhinitis medicamentosa, drug addiction, and tachyphylaxis [21]. Rhinitis medicamentosa, also known as rebound rhinitis, is characterized by increased nasal congestion as a result of prolonged daily use of topical nasal decongestant agents. We also used budesonide (a synthetic corticosteroid with strong anti-inflammatory action frequently used in the treatment of chronic allergic rhinitis) as a complementary agent to prevent the appearance of rhinitis medicamentosa. The results of a randomized double-blind controlled study carred out by Fergunson et al. [22] support the common clinical practice of nasal steroids to ameliorate rebound congestion concomitant with and after cessation of topical decongestant agents. Although we found a decreased incidence of NMS in the treated group, we must also consider the potential role of the nasal isotonic saline instillation followed by aspiration of nasal secretions.
One important finding of our study was the lack of a single case of NMS due to anaerobic micro-organism in patients receiving NDCA. Although isolation of anaerobic bacteria may be frequent if adequate transport conditions and special care in the laboratory are systematically achieved [3], most studies report low percentages of anaerobes in patients with NMS [1, 2, 6]. In our study the anaerobes were 21.05% of all isolated micro-organisms, but no anaerobe micro-organisms were isolated from maxillary aspirates of patients in the NDCA group. This could be an important support for the use of NDCA to prevent NMS in these patients if the incidence of anaerobe NMS is in fact as frequent as reported by Le Moal et al. [3]. Our explanation for anaerobic suppression in the treated group is the effect of NDCAs on ostial obstruction and edema of the ostiomeatal complex that initiates sinus inflammation and mucus accumulation within the sinus cavity, alleviating anaerobic conditions.
Another important question regarding NMS in patients treated in an ICU is whether NMS is an important cause of unexplained fever and, especially, whether it is a primary source of VAP. Several studies have demonstrated that NMS are frequently associated with nosocomial pneumonia, with similar micro-organisms often isolated from both sites [2, 5, 23]. The same micro-organisms were isolated from sinus and lungs in 6 of our 17 patients with VAP and IMS. Holzapfel et al. [4] showed that VAP in patients undergoing prolonged mechanical ventilation via a nasotracheal intubation can be prevented by the systemic search by CT and treatment of sinusitis by intravenous administration of antibiotics and sinus lavage. Although our data suggest that the decreased incidence of NMS in patients receiving NDCA is followed by a decreased incidence of VAP, further studies are needed to confirm this.
In conclusion, in mechanically ventilated patients with multiple traumas the combination of locally applied NDCA and corticosteroids reduces the incidence of RMS and may also reduce the incidence of NMS, especially that caused by anaerobes. This practice could be an integrated part of a prevention strategy against NMS.
References
Holzapfel L, Chervet S, Mandinier G, Madinier G, Ohen F, Demingeon G, Coupry A, Chaudet M (1993) Influence of long term oro or nasotracheal intubation on nosocomial maxillary sinusitis and pneumonia: a prospective randomized clinic trial. Crit Care Med 21:1132–1138
Rouby JJ, Laurent P, Gosnach M, Cambau E, Lamas G, Zouaoui A, Leguilou J-L, Bodin L, Khac TD, Marsault C, Poete P, Nikolas M-H, Jarlier V, Viars P (1994) Risk factors and clinical relevance of nosocomial maxillary sinusitis in the critically ill. Am J Respir Crit Care Med 150:776–783
Le Moal G, Lemerre D, Grollier G, Desmont C, Klossek J-M Robert R (1999) Nosocomial sinusitis with isolation of anaerobic bacteria in ICU patients. Intensive Care Med 25:1066–1071
Holzapfel L, Chastang C, Demingeon G, Bohe J, Piralla B, Coupry A (1999) A randomized study assessing the systematic search for maxillary sinusitis in nasotracheally mechanically ventilated patients. Am J Respir Crit Care Med 159:695–701
Sourweine B, Mom T, Traore O, Aublet-Cuvelier B, Bret l, Sirot J, Beteix P, Gilain L, Boyer L (2000) Ventilator associated sinusitis: microbiological results of sinus aspirates in patients on antibiotics. Anesthesiology 93:1255–1260
Bert F, Lambert -Zechovsky N (1995) Microbiology of nosocomial sinusitis in intensive care unit patients. J Infect 31:5–8
Westergren V, Lundbland L, Forsum U (1998) Ventilator-associated sinusitis: antroscopic findings and bacteriology when excluding contaminants. Acta Otolaryngol (Stockh) 118:547–580
Talmor M, Lip P, Barie PS (1997) Acute paranasal sinusitis in critically ill patients: Guidelines for prevention, diagnosis and treatment. Clin Infect Dis 24:1441–1446
Geiss HK (1999) Nosocomial sinusitis. Intensive Care Med 25:1037–1039
Pneumatikos I, Koulouras V, Nathanail C, Goe D, Nakos G (2002) Selective decontamination of subglottic area in mechanically ventilated patients with multiple trauma. Intensive Care Med 28:432–437
Aust R, Drettner B (1974) The functional size of the human maxillary ostium in vivo. Acta Otolaryngol (Stockh) 78:432–435
Aust R, Drettner B (1975) The patency of the maxillary ostium in relation to body posture. Acta Otolaryngol (Stockh) 80:443–446
Aust R, Stierna P, Drettner B (1994) Basic experimental studies of ostial patency and local metabolic environment of the maxillary sinus. Acta Otolaryngol Suppl (Stockh) 515:7–10
Deja M, Busch T, Bachmann S, Riskowski K, Campean V, Wiedmann B, Schwabe M, Hell B, Pheilschifter J, Falke K, Lewandowski K (2003) Reduced Nitric oxide in sinus epithelium of patients with radiologic sinusitis and sepsis. Am J Respir Crit Care Med 168:281–286
Arens JF, LeJeune FE, Webre DR (1974) Maxillary sinusitis a complication of nasotracheal intubation. Anesthesiology 40:415–416
Gallagher TJ, Civetta JM (1976) Acute maxillary sinusitis complicating nasotracheal intubation: a case report. Anesth Analg 55:885–886
Knodel AR, Beekaman JF (1982) Unexplained fever in patients with nasotracheal intubation. JAMA 248:868–870
Deutschman CS, Wilton PB, Snow J et al. (1985) Paranasal sinusitis: a common complication of nasotracheal intubation in neurosurgical patients. Neurosurgery 17:296–299
Castellano F, Mautone G (2002) Decongestant activity of a new formulation of xylometazoline nasal spray: a double-blind, randomized versus placebo and reference drugs controlled dose-effect study. Drugs Exp Clin Res 28:27–35
Westerveld GJ, Scheeren RA, Dekker I, Griffioen DH, Voss HP, Bast A (1995) Anti-oxidant actions of oxymethazoline and xylomethazoline. Eur J Pharmacol 291:27–31
Graf P, Juto JE (1995) Sustained use of xylometazoline nasal spray shortens the decongestive response and induces rebound swelling. Rhinology 33:14–17
Ferguson BJ, Paramaesvaran S, Rubinstein E (2001) A study of the effect of nasal steroid sprays in perennial allergic rhinitis patients with rhinitis medicamentosa. Otolaryngol Head Neck Surg 125:253–260
Bert F, Lambert-Zechovsky N (1996) Sinusitis in mechanically ventilated patients and its role in the pathogenesis of nosocomial pneumonia. Eur J Clin Microbiol Infect Dis 15:533–544
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by a grant from the Society for Pulmonary and Critical Care Research of East Macedonia and Thrace
Rights and permissions
About this article
Cite this article
Pneumatikos, I., Konstantonis, D., Tsagaris, I. et al. Prevention of nosocomial maxillary sinusitis in the ICU: the effects of topically applied α-adrenergic agonists and corticosteroids. Intensive Care Med 32, 532–537 (2006). https://doi.org/10.1007/s00134-006-0078-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0078-9