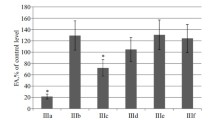
Summary
Aminoguanidine, a nucleophilic hydrazine, has been shown to be capable of blocking the formation of advanced glycation end products. It reduces the development of atherosclerotic plaques and prevents experimental diabetic nephropathy. We have found that aminoguanidine is also quite potent at inhibiting semicarbazide-sensitive amine oxidase (SSAO) both in vitro and in vivo. The inhibition is irreversible. This enzyme catalyses the deamination of methylamine and aminoacetone, which leads to the production of cytotoxic formaldehyde and methylglyoxal, respectively. Serum SSAO activity was reported to be increased in diabetic patients and positively correlated with the amount of plasma glycated haemoglobin. Increased SSAO has also been demonstrated in diabetic animal models. Urinary excretion of methylamine is substantially increased in the rats following acute or chronic treatment with aminoguanidine. Urinary methylamine levels were substantially increased in streptozotocin (STZ)-induced diabetic rats following administration of aminoguanidine. The non-hydrazine SSAO inhibitor (E)-2-(4-fluorophenethyl)-3-fluoroallylamine hydrochloride (MDL-72974A) has been shown to reduce urinary excretion of lactate dehydrogenase (an indicator of nephropathy) in STZ-induced diabetic rats. Formaldehyde not only induces protein crosslinking, but also enhances the advanced glycation of proteins in vitro. The results support the hypothesis that increased SSAO-mediated deamination may be involved in structural modification of proteins and contribute to advanced glycation in diabetes. The clinical implications for the use of aminoguanidine to prevent glycoxidation have been discussed.
Article PDF
Similar content being viewed by others
Abbreviations
- SSAO:
-
Semicarbazide-sensitive amine oxidase
- STZ:
-
streptozotocin
- AGEs:
-
advanced glycation end products
- LDH:
-
lactate dehydrogenase
- LDL:
-
low density lipoprotein
- MDL-72974A:
-
(E)-2-(4-fluoro-phenethyl)-3-fluoroallylamine hydrochloride.
References
Brownlee M, Cerami A, Vlassara H (1988) Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications. N Engl J Med 318:1315–1321
Brownlee M (1992) Non-enzymatic glycosylation of macro-molecules: prospects of pharmacological modulation. Diabetes 41 [Suppl 2]: 57–60
Fu MX, Knecht KJ, Thorpe SR, Baynes JW (1992) Role of oxygen in cross linking and chemical modification of collagen by glucose. Diabetes 41[Suppl 2]: 42–48
Baynes JW (1991) Role of oxidated stress in development of complications in diabetes. Diabetes 40: 405–412
Hunt JV, Bottoms MA, Mitchinson MJ (1993) Oxidative alteration in the experimental glycation model of diabetes mellitus are due to protein-glucose adduct oxidation. Bio-chem J 291: 529–535
Hunt JV, Smith CCT, Wolff SP (1990) Autooxidative glycosylation and possible involvement of peroxides and free radicals in LDL modification by glucose. Diabetes 39: 1420–1424
Monnier VM, Kohn RR, Cerami A (1984) Accelerated age related browning of human collagen in diabetes mellitus. Proc Natl Acad Sci USA 81: 583–587
Lo SK, Janakidevi K, Lai L, Malik AB (1993) Hydrogen peroxide-induced increase in endothelial adhesiveness is dependent on ICAM-1 activation. Am J Physiol 264: L406-L412
Uusitupa MIJ, Niskanen LK, Sittonen O, Voutilainen E, Pyorala K (1993) Ten-year cardiovascular mortality in relation to risk factors and abnormalities in lipoprotein composition in type 2 (non-insulin-dependent) diabetic and nondiabetic subjects. Diabetologia 36:1175–1184
Brownlee M (1985) Microvascular disease and related abnormalities: Their relation to control of diabetes. In: Marble A et al. (eds) Joslin’s diabetes mellitus. Lea & Febiger, Philadelphia pp 185–216
Mullarkey CJ, Edelstein D, Brownlee M (1990) Free radical generation by early glycation products: a mechanism for accelerated atherogenesis in diabetes. Biochem Bio-phys Res Comm 173: 932–939
Brett J, Ogawa S, Kirstein M, Radoff S, Vlassara H, Stem D (1990) Advanced glycosylation end products selectively attract monocytes to migrate across endothelial cell monolayers and induce activation and growth factor elaboration. Circulation 82[Suppl 3]: 92 (Abstract)
Vlassara H, Makita Z, Rayfield E, Freidman E, Cerami A, Morgelo S (1990) In vitro advanced glycation as a signal for monocyte migration in vessel wall: role in diabetes and aging. Circulation 82[Suppl 3]: 92 (Abstract)
Edelstein D, Brownlee M (1992) Mechanistic studies of advanced glycosylation end product inhibition by aminoguanidine. Diabetes 41: 26–29
Soulis-Liparota T, Cooper M, Papazoglou D, Clarke B, Jerums G (1991) Retardation by aminoguanidine of development of albuminuria, mesangial expansion and tissue fluorescence in streptozotocin induced diabetic rats. Diabetes 40:1328–1335
Bucala R, Makita Z, Koschinsky T, Cerami A, Vlassara H (1993) Lipid advanced glycation: pathway for lipid oxidation in vivo. Proc Natl Acad Sci USA 90: 6434–6438
Picard S, Parathasarathy S, Fruebis J, Witzum JL (1992) Aminoguanidine inhibits oxidative modification of low density lipoprotein and the subsequent increase in uptake by macrophage scavenger receptors. Proc Natl Acad Sci USA 89: 6876–6880
O’Brien RE, Panangiotopoulos S, Cooper MS, Jerums G (1992) Anti-atherogenic effect of aminoguanidine, an inhibitor of advanced glycation. Diabetes 4[Suppl 1] 16A (Abstract)
Itakura M, Yoshikawa H, Bannai C et al. (1992) Aminoguanidine decreases urinary albumin and high-molecular weight proteins in diabetic rats. Life Sci 49: 889–897
Huijiberts MSP, Wolffenbuttel BHR, Crijns FRL et al. (1994) Aminoguanidine reduces regional albumin clearance but not urinary albumin excretion in streptozotocin-diabetic rats. Diabetologia 37:10–14
Yu PH (1990) Oxidative deamination of aliphatic amines by rat aorta semicarbazide-sensitive amine oxidase. J Pharm Pharmacol 42: 882–884
Boor PJ, Trent MB, Lyles GA, Tao M, Ansari GAS (1992) Methylamine metabolism to formaldehyde by vascular semicarbazide-sensitive amine oxidase. Toxicology 73: 251–258
McEwen CM Jr, Castell DO (1967) Abnormalities of serum monoamine oxidase in chronic liver disease. J Lab Clin Med 70: 36–47
Nilsson SE, Tryding N, Tufvesson G (1968) Serum monoamine oxidase in diabetes mellitus and some other internal diseases. Acta Med Scand 184:105–108
Tryding N, Nilsson SE, Tufvesson G et al. (1969) Physiological and pathological influences on serum monoamine oxidase level. Scan J Clin Lab Invest 23: 79–84
Boomsma F, Derkx FHM, van den Meiracker AH, Veld AJM, Schalekamp MADH (1995) Plasma semicarbazide-sensitive amine oxidase activity is elevated in diabetes mellitus and correlates with glycosylated haemoglobin. Clin Sci 88: 675–679
Hayes BE, Clarke DE (1990) Semicarbazide-sensitive amine oxidase activity in streptozotocin diabetic rats. Res Comm Chem Pathol Pharmacol 69: 71–83
Elliott J, Fowden AL, Callingham BA, Sharman DF, Silver M (1991) Physiological and pathological influences on sheep blood plasma amine oxidase: effect of pregnancy and experimental alloxan-induced diabetes mellitus. Res Vet Science 50: 334–339
Yu PH, Zuo DM (1993) Methylamine, a potential endogenous toxin for vascular tissues: formation of formaldehyde via enzymatic deamination and the cytotoxic effects on endothelial cells. Diabetes 42: 594–603
Yu PH, Zuo DM (1991) (E)-4-Fluoro-beta-fluoroethylene benzene butamine (MDL-72974A) as highly potent inhibitor for semicarbazide-sensitive amine oxidase from vascular tissues and serum of different species. Biochem Pharmacol 43: 307–312
Chouinard S, Viau C (1992) Reversibility of renal tubular dysfunction in streptozotocin-induced diabetes in the rat. Can J Physiol Pharmacol 70: 977–982
Jane SM, Mu D, Wemmer D et al. (1990) A new redox cofactor in eukaryotic enzymes: 6-hydroxydopa at the active site of bovine serum amine oxidase. Science 248: 981–987
Jane SM, Klinman JP (1991) An investigation of bovine serum amine oxidase active site stoichiometry: evidence for an aminotransferase mechanism involving two carbonyl cofactors per enzyme dimer. Am Chem Soc 30: 4599–4605
Nicholis K, Mandel TE (1989) Advanced glycosylation end-products in experimental murine diabetic nephropathy: effect of islet isografting and of aminoguanidine. Lab Invest 60: 486–491
Corbett JA, Tilton RG, Chang K et al. (1992) Aminoguanidine, a novel inhibitor of nitric oxide formation, prevents diabetic vascular dysfunction. Diabetes 41: 552–556
Tilton RG, Chang K, Hason KS et al. (1993) Prevention of diabetic vascular dysfunction by guanidines; inhibition of nitric oxide synthase versus advanced glycation end-product formation. Diabetes 42: 221–232
Tamura H, Horiike K, Fukura H, Watanabe T (1989) Kinetic studies on the inhibition mechanism of diamine oxidase from porcine kidney by aminoguanidine. J Biochem (Tokyo) 105: 299–306
Seiler N (1995) Polyamine oxidase, properties and functions. Prog Brain Res 106: 333–344
Mathison BH, Murphy SE, Shank R (1994) Hydralazine and other hydrazine derivatives and the formation of DNA adducts. Toxicol Appl Pharmacol 127: 91–98
Bosan WS, Shank RC, MacEven JD, Gaworske CL, Newberne PM (1987) Methylation of DNA guanine during the course of induction of liver cancer in hamsters by hydrazine or dimethylnitrosamine. Carcinogenesis 8: 439–444
Helander A, Tottmar O (1987) Metabolism of biogenic aldehydes in isolated human blood cells, platelets and in plasma. Biochem Pharmacol 36: 1077–1082
Sato Y, Hotta N, Sakamonto N, Matsuoka S, Ohishin N, Yagi IK (1979) Lipid peroxide levels in plasma of diabetic patients. Biochem Med 21: 104–107
Jennings PE, Jones AF, Florkouski CM, Lunic J, Barnett AH (1987) Increased diene conjugates in diabetic subjects with microangiopathy. Diabet Med 4: 452–456
Ohkuwa T, Sato Y, Naoi M (1995) Hydroxyl radical formation in diabetic rats induced by streptozotocin. Life Sci 56: 1789–1798
Gutteridge JMC (1994) Hydroxyl radicals, iron, oxidative stress and neurodegeneration. Ann N Y Acad Sci 738: 201–213
Lyles GA, Singh I (1985) Vascular smooth muscle cells: a major source of the semicarbazide-sensitive amine oxidase of the rat aorta. J Pharm Pharmacol 37: 637–643
Wibo M, Duong AT, Godfraind T (1980) Subcellular location of semicarbazide-sensitive amine oxidase in rat aorta. Eur J Biochem 112: 87–94
Lyles GA, Bertie KH (1987) Properties of a semicarbazide-sensitive amine oxidase in rat articular cartilage. Pharmacol Toxicol [Suppl] 1: 33
Zuo, DM, Yu, PH (1993) Semicarbazide-sensitive amine oxidase and monoamine oxidase in rat brain microvessels, meninges, retina and eye sciera. Brain Research Bull 33: 307–311
Lyles GA, Chalmers J (1992) The metabolism of aminoacetone to methylglyoxal by semicarbazide-sensitive amine oxidase in human umbilical artery. Biochem Pharmacol 31: 1417–1424
Yu PH, Zuo DM (1995) Formaldehyde produced endogenously via deamination of methylamine; a potential risk factor for initiation of endothelial injury. Atherosclerosis 120: 189–197
Kapeller-Adler R, Toda K (1932) Über das Vorkommen von Monomethylamine im Harn. Biochem Zeischrift 248: 403–425
McLellan AC, Phillips SA, Thornalley PJ (1992) The assay of methylglyoxal in biological systems by derivatization with 1,2-diamino-4,5-dimethoxybenzene. Anal Biochem 206: 17–23
Lo TWC, Westwood ME, McLellan AC Seiwood T, Thornalley PJ (1994) Binding and modification of proteins by methylglyoxal and physiological conditions. J Biol Chem 269: 32299–32305
Thornalley PJ (1990) The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem J 269: 1–11
Brownlee M, Vlassara H, Kooney P, Ulrich P, Cerai A (1986) Aminoguanidine prevents diabetes-induced arterial wall protein cross-linking. Science 232: 1629–1632
Lo TWC, Selwood T, Thornalley PJ (1994) The reaction of methylglyoxal with aminoguanidine under physiological conditions and prevention of methylglyoxal binding to plasma proteins. Biochem Pharmacol 48: 1865–1870
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yu, P.H., Zuo, D.M. Aminoguanidine inhibits semicarbazide-sensitive amine oxidase activity: implications for advanced glycation and diabetic complications. Diabetologia 40, 1243–1250 (1997). https://doi.org/10.1007/s001250050816
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s001250050816