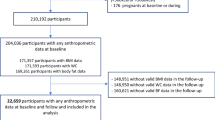
Abstract
Aims/hypothesis
Diets with higher inflammatory and insulinaemic potential have been associated with an increased risk of type 2 diabetes. However, it remains unknown whether plasma metabolomic profiles related to proinflammatory/hyperinsulinaemic diets and to inflammatory/insulin biomarkers are associated with type 2 diabetes risk.
Methods
We analysed 6840 participants from the Nurses’ Health Study and Health Professionals Follow-up Study to identify the plasma metabolome related to empirical dietary inflammatory pattern (EDIP), empirical dietary index for hyperinsulinemia (EDIH), four circulating inflammatory biomarkers and C-peptide. Dietary intakes were assessed using validated food frequency questionnaires. Plasma metabolomic profiling was conducted by LC-MS/MS. Metabolomic signatures were derived using elastic net regression. Multivariable Cox regression was used to examine associations of the metabolomic profiles with type 2 diabetes risk.
Results
We identified 27 metabolites commonly associated with both EDIP and inflammatory biomarker z score and 21 commonly associated with both EDIH and C-peptide. Higher metabolomic dietary inflammatory potential (MDIP), reflecting higher metabolic potential of both an inflammatory dietary pattern and circulating inflammatory biomarkers, was associated with higher type 2 diabetes risk. The HR comparing highest vs lowest quartiles of MDIP was 3.26 (95% CI 2.39, 4.44). We observed a strong positive association with type 2 diabetes risk for the metabolomic signature associated with EDIP-only (HR 3.75; 95% CI 2.71, 5.17) or inflammatory biomarkers-only (HR 4.07; 95% CI 2.91, 5.69). In addition, higher metabolomic dietary index for hyperinsulinaemia (MDIH), reflecting higher metabolic potential of both an insulinaemic dietary pattern and circulating C-peptide, was associated with greater type 2 diabetes risk (HR 3.00; 95% CI 2.22, 4.06); further associations with type 2 diabetes were HR 2.79 (95% CI 2.07, 3.76) for EDIH-only signature and HR 3.89 (95% CI 2.82, 5.35) for C-peptide-only signature. The diet scores were significantly associated with risk, although adjustment for the corresponding metabolomic signature scores attenuated the associations with type 2 diabetes, these remained significant.
Conclusions/interpretation
The metabolomic signatures reflecting proinflammatory or hyperinsulinaemic diets and related biomarkers were positively associated with type 2 diabetes risk, supporting that these dietary patterns may influence type 2 diabetes risk via the regulation of metabolism.
Graphical Abstract



Similar content being viewed by others
Abbreviations
- BIOM-only:
-
Metabolomic profile score comprised of metabolites associated mainly with inflammatory biomarkers, not considering their associations with EDIP
- CPEP-only:
-
Metabolomic profile score comprised of metabolites associated mainly with C-peptide, not considering their associations with EDIH
- CRP:
-
C-reactive protein
- EDIH:
-
Empirical dietary index for hyperinsulinemia
- EDIP:
-
Empirical dietary inflammatory pattern
- FDR:
-
False discovery rate
- FFQ:
-
Food frequency questionnaire
- HPFS:
-
Health Professionals Follow-up Study
- MDIH:
-
Metabolomic dietary index for hyperinsulinemia
- MDIP:
-
Metabolomic dietary inflammatory potential
- NHS:
-
Nurses’ Health Study
- PC:
-
Phosphatidylcholine
- PE:
-
Phosphatidylethanolamine
- TAG:
-
Triacylglycerol
- TNF-α-R2:
-
TNF-α receptor 2
- WHI:
-
Women’s Health Initiative
References
Harmon BE, Boushey CJ, Shvetsov YB et al (2015) Associations of key diet-quality indexes with mortality in the Multiethnic Cohort: the Dietary Patterns Methods Project. Am J Clin Nutr 101(3):587–597. https://doi.org/10.3945/ajcn.114.090688
Liese AD, Krebs-Smith SM, Subar AF et al (2015) The Dietary Patterns Methods Project: synthesis of findings across cohorts and relevance to dietary guidance. J Nutr 145(3):393–402. https://doi.org/10.3945/jn.114.205336
Cespedes EM, Hu FB, Tinker L et al (2016) (2016) Multiple healthful dietary patterns and type 2 diabetes in the Women’s Health Initiative. Am J Epidemiol 183:622–633. https://doi.org/10.1093/aje/kwv241
World Cancer Research Fund/American Institute for Cancer Research (2018) Diet, nutrition, physical activity and cancer: a global perspective. Continuous Update Project Expert Report 2018. Available from www.wcrf.org/wp-content/uploads/2021/02/Summary-of-Third-Expert-Report-2018.pdf
Chiuve SE, Fung TT, Rimm EB et al (2012) Alternative dietary indices both strongly predict risk of chronic disease. J Nutr 142(6):1009–1018. https://doi.org/10.3945/jn.111.157222
Schulze MB, Hoffmann K, Kroke A, Boeing H (2003) An approach to construct simplified measures of dietary patterns from exploratory factor analysis. Br J Nutr 89(3):409–418. https://doi.org/10.1079/BJN2002778
Varraso R, Garcia-Aymerich J, Monier F et al (2012) Assessment of dietary patterns in nutritional epidemiology: principal component analysis compared with confirmatory factor analysis. Am J Clin Nutr 96(5):1079–1092. https://doi.org/10.3945/ajcn.112.038109
Tabung FK, Giovannucci EL, Giulianini F et al (2018) An empirical dietary inflammatory pattern score is associated with circulating inflammatory biomarkers in a multi-ethnic population of postmenopausal women in the United States. J Nutr 148(5):771–780. https://doi.org/10.1093/jn/nxy031
Tabung FK, Smith-Warner SA, Chavarro JE et al (2017) An empirical dietary inflammatory pattern score enhances prediction of circulating inflammatory biomarkers in adults. J Nutr 147(8):1567–1577. https://doi.org/10.3945/jn.117.248377
Tabung FK, Smith-Warner SA, Chavarro JE et al (2016) Development and validation of an empirical Dietary Inflammatory Index. J Nutr 146(8):1560–1570. https://doi.org/10.3945/jn.115.228718
Tabung FK, Wang W, Fung TT et al (2016) Development and validation of empirical indices to assess the insulinaemic potential of diet and lifestyle. Br J Nutr 116(10):1787–1798. https://doi.org/10.1017/S0007114516003755
Liu L, Nishihara R, Qian ZR et al (2017) Association between inflammatory diet pattern and risk of colorectal carcinoma subtypes classified by immune responses to tumor. Gastroenterology 153(6):1517–1530. https://doi.org/10.1053/j.gastro.2017.08.045. (e1514)
Tabung FK, Liu L, Wang W et al (2018) Association of dietary inflammatory potential with colorectal cancer risk in men and women. JAMA Oncol 4(3):366–373. https://doi.org/10.1001/jamaoncol.2017.4844
Tabung FK, Wang W, Fung TT et al (2018) Association of dietary insulinemic potential and colorectal cancer risk in men and women. Am J Clin Nutr 108(2):363–370. https://doi.org/10.1093/ajcn/nqy093
Li J, Lee DH, Hu J et al (2020) Dietary inflammatory potential and risk of cardiovascular disease among men and women in the U.S. J Am Coll Cardiol 76(19):2181–2193. https://doi.org/10.1016/j.jacc.2020.09.535
Lee DH, Li J, Li Y et al (2020) Dietary inflammatory and insulinemic potential and risk of type 2 diabetes: results from three prospective US cohort studies. Diabetes Care 43(11):2675–2683. https://doi.org/10.2337/dc20-0815
Guasch-Ferre M, Bhupathiraju SN, Hu FB (2018) Use of metabolomics in improving assessment of dietary intake. Clin Chem 64(1):82–98. https://doi.org/10.1373/clinchem.2017.272344
Brennan L, Hu FB (2019) Metabolomics-based dietary biomarkers in nutritional epidemiology—current status and future opportunities. Mol Nutr Food Res 63(1):e1701064. https://doi.org/10.1002/mnfr.201701064
Johnson CH, Ivanisevic J, Siuzdak G (2016) Metabolomics: beyond biomarkers and towards mechanisms. Nat Rev Mol Cell Biol 17(7):451. https://doi.org/10.1038/nrm.2016.25
Colditz GA, Manson JE, Hankinson SE (1997) The Nurses’ Health Study: 20-year contribution to the understanding of health among women. J Women’s Health 6(1):49–62. https://doi.org/10.1089/jwh.1997.6.49
Rimm EB, Giovannucci EL, Willett WC et al (1991) Prospective study of alcohol consumption and risk of coronary disease in men. Lancet 338(8765):464–468. https://doi.org/10.1016/0140-6736(91)90542-W
Hankinson SE, Willett WC, Manson JE et al (1995) Alcohol, height, and adiposity in relation to estrogen and prolactin levels in postmenopausal women. J Natl Cancer Inst 87(17):1297–1302. https://doi.org/10.1093/jnci/87.17.1297
Wittenbecher C, Guasch-Ferre M, Haslam DE et al (2022) Changes in metabolomics profiles over ten years and subsequent risk of developing type 2 diabetes: results from the Nurses’ Health Study. EBioMedicine 75:103799. https://doi.org/10.1016/j.ebiom.2021.103799
Feskanich D, Rimm EB, Giovannucci EL et al (1993) Reproducibility and validity of food intake measurements from a semiquantitative food frequency questionnaire. J Am Dietetic Assoc 93(7):790–796. https://doi.org/10.1016/0002-8223(93)91754-E
Rimm EB, Giovannucci EL, Stampfer MJ, Colditz GA, Litin LB, Willett WC (1992) Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am J Epidemiol 135(10):1114–1126. https://doi.org/10.1093/oxfordjournals.aje.a116211
Willett WC, Sampson L, Stampfer MJ et al (1985) Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol 122(1):51–65. https://doi.org/10.1093/oxfordjournals.aje.a114086
Yuan C, Spiegelman D, Rimm EB et al (2018) Relative validity of nutrient intakes assessed by questionnaire, 24-hour recalls, and diet records as compared with urinary recovery and plasma concentration biomarkers: findings for women. Am J Epidemiol 187(5):1051–1063. https://doi.org/10.1093/aje/kwx328
Yuan C, Spiegelman D, Rimm EB et al (2017) Validity of a dietary questionnaire assessed by comparison with multiple weighed dietary records or 24-hour recalls. Am J Epidemiol 185(7):570–584. https://doi.org/10.1093/aje/kww104
Hu FB, Stampfer MJ, Rimm E et al (1999) Dietary fat and coronary heart disease: a comparison of approaches for adjusting for total energy intake and modeling repeated dietary measurements. Am J Epidemiol 149(6):531–540. https://doi.org/10.1093/oxfordjournals.aje.a009849
Pai JK, Pischon T, Ma J et al (2004) Inflammatory markers and the risk of coronary heart disease in men and women. N Engl J Med 351(25):2599–2610. https://doi.org/10.1056/NEJMoa040967
Cheng S, Larson MG, McCabe EL et al (2015) Distinct metabolomic signatures are associated with longevity in humans. Nat Commun 6(1):1–10. https://doi.org/10.1038/ncomms7791
Mascanfroni ID, Takenaka MC, Yeste A et al (2015) Metabolic control of type 1 regulatory T cell differentiation by AHR and HIF1-α. Nat Med 21(6):638–646. https://doi.org/10.1038/nm.3868
O’Sullivan JF, Morningstar JE, Yang Q et al (2017) Dimethylguanidino valeric acid is a marker of liver fat and predicts diabetes. J Clin Investig 127(12):4394–4402. https://doi.org/10.1172/JCI95995
Paynter NP, Balasubramanian R, Giulianini F et al (2018) Metabolic predictors of incident coronary heart disease in women. Circulation 137(8):841–853. https://doi.org/10.1161/CIRCULATIONAHA.117.029468
Friedman J, Hastie T, Tibshirani R (2010) Regularization paths for generalized linear models via coordinate descent. J Stat Softw 33(1):1–22
Li J, Guasch-Ferré M, Chung W et al (2020) The Mediterranean diet, plasma metabolome, and cardiovascular disease risk. Eur Heart J 41(28):2645–2656. https://doi.org/10.1093/eurheartj/ehaa209
Wang F, Baden MY, Guasch-Ferré M et al (2022) Plasma metabolite profiles related to plant-based diets and the risk of type 2 diabetes. Diabetologia 65(7):1119–1132. https://doi.org/10.1007/s00125-022-05692-8
Lee DH, Jin Q, Shi N et al (2023) Dietary inflammatory and insulinemic potentials, plasma metabolome and risk of colorectal cancer. Metabolites 13(6):744. https://doi.org/10.3390/metabo13060744
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodological) 57(1):289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Tabung FK, Liang L, Huang T et al (2019) Identifying metabolomic profiles of inflammatory diets in postmenopausal women. Clin Nutr. https://doi.org/10.1016/j.clnu.2019.06.010
Bene J, Hadzsiev K, Melegh B (2018) Role of carnitine and its derivatives in the development and management of type 2 diabetes. Nutr Diabetes 8(1):8. https://doi.org/10.1038/s41387-018-0017-1
Bruce CR, Hoy AJ, Turner N et al (2009) Overexpression of carnitine palmitoyltransferase-1 in skeletal muscle is sufficient to enhance fatty acid oxidation and improve high-fat diet-induced insulin resistance. Diabetes 58(3):550–558. https://doi.org/10.2337/db08-1078
Paumen MB, Ishida Y, Muramatsu M, Yamamoto M, Honjo T (1997) Inhibition of carnitine palmitoyltransferase I augments sphingolipid synthesis and palmitate-induced apoptosis. J Biol Chem 272(6):3324–3329. https://doi.org/10.1074/jbc.272.6.3324
Hang D, Zeleznik OA, He X et al (2020) Metabolomic signatures of long-term coffee consumption and risk of type 2 diabetes in women. Diabetes Care 43(10):2588–2596. https://doi.org/10.2337/dc20-0800
Wang DD, Hu FB (2018) Precision nutrition for prevention and management of type 2 diabetes. Lancet Diabetes Endocrinol 6(5):416–426. https://doi.org/10.1016/S2213-8587(18)30037-8
Townsend MK, Clish CB, Kraft P et al (2013) Reproducibility of metabolomic profiles among men and women in 2 large cohort studies. Clin Chem 59(11):1657–1667. https://doi.org/10.1373/clinchem.2012.199133
Jin Q, Shi N, Aroke D et al (2021) Insulinemic and inflammatory dietary patterns show enhanced predictive potential for type 2 diabetes risk in postmenopausal women. Diabetes Care 44(3):707–714. https://doi.org/10.2337/dc20-2216
Shi N, Aroke D, Jin Q et al (2021) Proinflammatory and hyperinsulinemic dietary patterns are associated with specific profiles of biomarkers predictive of chronic inflammation, glucose-insulin dysregulation, and dyslipidemia in postmenopausal women. Front Nutr 8:690428. https://doi.org/10.3389/fnut.2021.690428
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Acknowledgements
The authors thank the participants and staff of the NHS and HPFS for their valuable contributions. The graphical abstract was created using BioRender.com.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on request.
Funding
This work was supported by the National Institutes of Health research grants UM1 CA186107, U01 CA176726, U01 CA167552, P01 CA87969, R01 CA50385 and R00 CA207736. ELG is funded as an American Cancer Society Clinical Research Professor (CRP-23-1014041). This research was also supported by the Yonsei Signature Research Cluster Project (2021-22-0009) and the Yonsei University Research Fund of 2023-22-0159.
Authors’ relationships and activities
JAM has served as an advisor/consultant to Merck Pharmaceutical and COTA Healthcare. KW is currently an employee and stockholder of Vertex Pharmaceuticals. This work was not funded by this commercial entity. The other authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
DHL, ELG, JL and FKT were involved in the study concept and design. FBH, AHE, CC, ELG and FKT participated in acquisition of data. DHL, QJ, NS, FW, LL, OAZ, JL and FKT were involved in statistical analysis. All authors participated in interpretation of data. DHL prepared the first draft of the manuscript. All authors participated in critical revision of the manuscript and approved the final version of the manuscript. The corresponding author (FKT) takes full responsibility for the work and/or the conduct of the study, had access to the data and controlled the decision to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jun Li and Fred K. Tabung contributed equally as co-senior authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, D.H., Jin, Q., Shi, N. et al. The metabolic potential of inflammatory and insulinaemic dietary patterns and risk of type 2 diabetes. Diabetologia 67, 88–101 (2024). https://doi.org/10.1007/s00125-023-06021-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-023-06021-3