Abstract
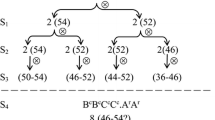
Allopolyploidy plays an important role in plant evolution and confers obvious advantages on crop growth and breeding compared to low ploidy levels. The present investigation was aimed at synthesising the first known chromosomally stable hexaploid Brassica with the genome constitution AABBCC. More than 2,000 putative hexaploid plants were obtained through large-scale hybridisation from various combinations of crosses between different cultivars of Brassica carinata (BBCC) and B. rapa (AA). The majority of plants after two generations of selfing within selected hexaploid plants (H2) were aneuploid, and only 80 plants (4.6%) had the expected hexaploid chromosome number (2n = 54). The hexaploid ratio increased to an average of 23.0 and 26.3% in the H3 and H4 generations, respectively, and was accompanied by an increase in pollen fertility. The appearance of aneuploid plants in each generation could be detected having various chromosomal abnormalities at meiosis. The frequency of hexaploid plants varied significantly among different cultivar combinations, from 0 to 56% in the H4 generation, and it showed a positive correlation with pollen fertility. The frequency of SSR allelic fragments lost or novel alleles gained was significantly lower in H4 than in H2 and H3, which reflects increasing genome stability in H4. The A and C genomes were significantly less stable than the B genome, which may mainly result from frequent homoeologous pairing and rearrangements between the A and C genomes. Methods to establish a stable hexaploid Brassica crop by intercrossing these lines followed by intensive selection are also discussed.





Similar content being viewed by others
References
Ammar K, Mergoum M, Rajaram S (2004) The history and evolution of triticale. In: Mergoum M, Macpherson HG (eds) Triticale improvement and production (FAO plant production and protection paper 179). Food and agriculture organization of the united nations, Rome, pp 1–9
Arumugam N, Mukhopadhyay A, Gupta V, Pental D, Pradhan AK (1996) Synthesis of hexaploid (AABBCC) somatic hybrids: a bridging material for transfer of ‘tour’ cytoplasmic male sterility to different Brassica species. Theor Appl Genet 92:762–768
Attia T, Röbbelen G (1986) Cytogenetic relationship within cultivated Brassica analyzed in amphidiploid from the three diploid ancestors. Can J Genet Cytol 28:323–329
Bennett MD (2004) Perspectives on polyploidy in plants-ancient and neo. Bio J Linn Soc 82:411–423
Burnham CR (1962) Discussions in cytogenetics. Burgess Publishing, Minneapolis
Chèvre AM, Eber F, Barret P, Brace J (1997) Identification of the different Brassica nigra chromosomes from both sets of B. oleracea-B. nigra and B. napus-B. nigra addition lines with special emphasis on chromosome transmission and self-incompatibility. Theor Appl Genet 94:603–611
Comai L (2000) Genetic and epigenetic interactions in allopolyploid plants. Plant Mol Biol 43:387–399
Comai L, Tyagi AP, Winter K, Davis RH, Reynolds SH, Stevens Y, Byers B (2000) Phenotypic instability and rapid gene silencing in newly formed Arabidopsis allotetraploids. Plant Cell 12:1551–1567
Doyle GG (1986) Aneuploidy and inbreeding depression in random mating and self-fertilizing autotetraploid populations. Theor Appl Genet 72:799–806
Doyle J, Doyle J (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Feldman M, Liu B, Sehgal G, Abbo S, Levy AA, Vega JM (1997) Rapid elimination of low copy DNA sequence in polyploid wheat: a possible mechanism for differentiation of homoeologous chromosomes. Genetics 147:1381–1387
Gerdemann KM, Sacristan MD, Braatz C, Schieder O (1994) Utilization of asymmetric somatic hybridization for the transfer of disease resistance from Brassica nigra to Brassica napus. Plant Breed 113:106–113
Howard HW (1942) The effect of polyploidy and hybridity on seed size in crosses between Brassica chinensis, B. carinata, amphidiploid B. chinensis-carinata, and autotetraploid B. chinensis. J Genet 43:105–119
Husband BC, Schemske DW (2000) Ecological mechanisms of reproductive isolation and coexistence of diploid and tetraploid Chamerion angustifolium. J Ecol 88:1–14
Iwasa S (1964) Cytogenetic studies on the artificially raised trigenomic hexaploid hybrid forms in the genus Brassica. J Fac Agr Kyushu Univ 13:309–318
Jenczewski E, Eber F, Grimaud A, Huet S, Lucas MO, Monod H, Chèvre AM (2003) PrBn, a major gene controlling homeologous pairing in oilseed rape (Brassica napus) haploids. Genetics 164:645–653
Jiang Y, Tian E, Li R, Chen L, Meng J (2007) Genetic diversity of Brassica carinata with emphasis on the interspecific crossability with B. rapa. Plant Breed 126:487–491
Kashkush K, Feldman M, Levy AA (2002) Gene loss, silencing and activation in a newly synthesized wheat allotetraploid. Genetics 160:1651–1659
Larter EN, Gustafson JP (1980) Triticale. In: Fehr WR, Hadley HH (eds) Hybridization of crop plants. American Society of Agronomy & Crop Science Society of America, Madison, pp 681–694
Leitch AR, Leitch IJ (2008) Genomic plasticity and the diversity of polyploid plants. Science 320:481–483
Levy AA, Feldman M (2004) Genetic and epigenetic reprogramming of the wheat genome upon allopolyploidization. Bio J Linn Soc 82:607–613
Li M, Cai D, Huang L (2001) Studies of the meiosis of 2n gamete apomictic wheat grass (Elymus rectisetus). Acta Genet Sin 28:939–946
Li M, Qian W, Meng J, Li Z (2004) Construction of novel Brassica napus genotypes through chromosomal substitution and elimination using interploid species hybridization. Chromosome Res 12:417–426
Li M, Chen X, Meng J (2006) Intersubgenomic heterosis in rapeseed production with a partial new-typed Brassica napus containing subgenome Ar from B. rapa and Cc from Brassica carinata. Crop Sci 46:234–242
Liu B, Vega JM, Feldman M (1998a) Rapid genomic changes in newly synthetized amphiploids of Triticum and Aegilops. II. changes in low-copy coding DNA sequences. Genome 41:535–542
Liu B, Vega JM, Segal G, Abbo S, Rodova M, Feldman M (1998b) Rapid genomic changes in newly synthesized amphiploids of Triticum and Aegilops. I. Changes in low-copy non-coding DNA sequences. Genome 41:272–277
Liu B, Brubaker CL, Mergeai G, Cronn RC, Wendel JF (2001) Ployploid formation in cotton is not accompanied by rapid genomic changes. Genome 44:321–330
Liu Z, Adamczyk K, Maria MD, Eber F, Lucas MO, Delourme R, Chévre AM, Jenczewski E (2006) Mapping PrBn and other quantitative trait loci responsible for the control of homeologous chromosome pairing in oilseed rape (Brassica napus L.) haploids. Genetics 174:1583–1596
Long Y, Shi J, Qiu D, Li R, Zhang C, Wang J, Hou J, Zhao J, Shi L, Choi SR, Park BS, Lim YP, Meng J (2007) Flowering time QTL analysis of oilseed Brassica in multiple environments and genome-wide alignment with Arabidopsis. Genetics 177:2433–2444
Lowe AJ, Moule C, Trick M, Edwards KJ (2004) Efficient large-scale development of microsatellites for marker and mapping applications in Brassica crop species. Theor Appl Genet 108:1103–1112
Lukens LN, Pires JC, Leon E, Vogelzang R, Oslach L, Osborn T (2006) Patterns of sequence loss and cytosine methylation within a population of newly resynthesized Brassica napus allopolyploids. Plant Physiol 140:336–348
Madlung A, Tyagi AP, Watson B, Jiang H, Kagochi T, Doerge RW, Martienssen R, Comai L (2005) Genomic changes in synthetic Arabidopsis polyploids. Plant J 41:221–230
Mahy G, Bruederle LP, Connors B, Hofwegen MV, Vorsa N (2000) Allozyme evidence for genetic autopolyploidy and high genetic diversity in tetraploid cranberry, Vaccinium oxycoccos (ericaceae). Am J Bot 87:1882–1889
Maich R, Ordóñez A (2003) Improved meiotic index in hexaploid triticale (Triticosecale Wittmack). Cytologia 3:303–306
Masterson J (1994) Stomatal size in fossil plants-evidence for polyploidy in majority of angiosperms. Science 264:421–424
Matzke MA, Scheid OM, Matzke AJM (1999) Rapid structural and epigenetic changes in polyploid and aneuploid genomes. BioEssays 21:761–767
Meng J, Shi S, Gan L, Li Z, Qu X (1998) The production of yellow-seeded Brassica napus (AACC) through crossing interspecific hybrids of B. campestris (AA) and B. carinata (BBCC) with B. napus. Euphytica 103:329–333
Merker A (1973a) Cytogenetic investigations in hexaploid Triticale I. Meiosis and fertility in F1 and F2. Hereditas 73:285–290
Merker A (1973b) Cytogenetics of hexaploid triticale. In Triticale: proceedings of an international symposium on cytogenetics of hexaploid triticale: 1–3 October, El Batan, Mexico, pp 167–172
Mizushima U (1950) On several artificial allopolyploids obtained in the tribe Brassiceae of Cruciferae. Tohoku J Agric Res 1:15–27
Nakajima G (1954) Genetical and cytological studies in the breeding of amphidiploid types between Triticum and Secale vii. external characters, fertility and somatic chromosomes of T. Pyramidale × S. cereale F2 plants. Jpn J Genet 29:202–204
Olsson G (1963) Induced polyploids in Brassica. In: Åkerberg E et al (eds) Recent research in plant breeding. Svalöf, New York, pp 1944–1961, 179–192
Osborn TC, Butruille DV, Sharpe AG, Pickering KJ, Parkin IAP (2003) Detection and effects of a homeologous reciprocal transposition in Brassica napus. Genetics 165:1569–1577
Ozkan H (2000) Genomic changes in newly synthesized amphiploids of Aegilops and Triticum. PhD Thesis University of Cukurova
Pires JC, Zhao JW, Schranz ME, Leon EJ, Quijiada PA, Lukens LN, Osborn TC (2004) Flowering time divergence and genomic rearrangements in resynthesized Brassica polyploids (Brassicaceae). Bio J Linn Soc 82:675–688
Pradhan A, Plummer JA, Nelson MN, Cowling WA, Yan G (2010) Trigenomic hybrids from interspecific crosses between Brassica napus and B. nigra. Crop Pasture Sci (accepted)
Prakash S, Hinata K (1980) Taxonomy, cytogenetics and origin of crop brassicas, a review. Opera Bot 55:1–57
Prakash S, Bhat SR, Quiros CF, Kirti PB, Chopra VL (2009) Brassica and its close allies: cytogenetics and evolution. Plant Breed Rev 31:21–187
Qian W, Chen X, Fu D, Zou J, Meng J (2005) Intersubgenomic heterosis in seed yield potential observed in a new type of Brassica napus introgressed with partial Brassica rapa genome. Theor Appl Genet 110:1187–1194
Qiu D, Morgan C, Shi J, Long Y, Liu J, Li R, Zhuang X, Wang Y, Tan X, Dietrich E, Weihmann T, Everett C, Vanstraelen S, Beckett P, Fraser F, Trick M, Barnes S, Wilmer J, Schmidt JR, Li J, Meng J, Bancroft I (2006) A comparative linkage map of oilseed rape and its use for QTL analysis of seed oil and erucic acid content. Theor Appl Genet 114:67–80
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst 29:467–501
Sacristán MD, Gerdemann KM, Schieder O (1989) Incorporation of hygromycin resistance in Brassica nigra and its transfer to B. napus through asymmetric protoplast fusion. Theor Appl Genet 78:194–200
Salmon A, Ainouche ML, Wendel JF (2005) Genetic and epigenetic consequences of recent hybridization and polyploidy in Spartina (Poaceae). Mol Ecol 14:1163–1175
SAS Institute Inc. (1999) SAS Online Doc®, version 8.0. Cary, NC
Shaked H, Kashkush K, Ozkan H, Feldman M, Levy AA (2001) Sequence elimination and cytosine methylation are rapid and reproducible responses of the genome to wide hybridization and allopolyploidy in wheat. Plant Cell 13:1749–1759
Sharpe AG, Parkin IAP, Keith DJ, Lydiate DJ (1995) Frequent nonreciprocal translocations in the amphidiploid genome of oilseed rape (Brassica napus). Genome 38:1112–1121
Singer T, Yordan C, Martienssen RA (2001) Robertson’s mutator transposons in A. thaliana are regulated by the chromatin-remodeling gene decrease in DNA methylation (DDM1). Genes Dev 15:591–602
Sjödin C, Glimelius K (1998) Brassica naponigra, a somatic hybrid resistant to Phoma lingam. Theor Appl Genet 77:651–656
Soltis DE, Soltis PS (1995) The dynamic nature of polyploidy genomes. Proc Nati Acad Sci USA 92:8089–8091
Soltis DE, Soltis PS, Tate JA (2004) Advances in the study of polyploidy since plant speciation. New Phytol 161:173–191
Song K, Lu P, Tang K, Osborn TC (1995) Rapid genome change in synthetic polyploids of Brassica and its implications for polyploid evolution. Proc Nati Acad Sci USA 92:7719–7723
SPSS (1999) SPSS (statistical product and service solutions) 10.0 for windows, , Chicago, IL. http://www.spss.com/spss
Takeda T (1967) Studies on the fertility of artificially synthesized trigenomic hexaploids in Brassicinae: Brassica carinata Harron × B. campestris L. and (B. nigra Koch × B. oleracea L.) × B. campestris var. sarson. Ann Rep Fac Agric Edu Iwate Univ 27:41–52
Thompson JD, Lumaret R (1992) The evolutionary dynamics of polyploid plants: origins, establishment and persistence. Trends Ecol Evol 7:302–307
Truco MJ, Hu J, Sadowsky J, Quiros CF (1996) Inter- and intra-genomic homology of the Brassica genomes: implications for their origin and evolution. Theor Appl Genet 93:1225–1233
Tsuchiya T, Larter EN (1971) Further results on chromosome stability of hexaploid Triticale. Euphytica 20:591–596
U N (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn J Bot 7:389–452
Udall JA, Quijada PA, Osborn TC (2005) Detection of chromosomal rearrangements derived from homeologous recombination in four mapping populations of Brassica napus L. Genetics 169:967–979
Wang J, Tian L, Madlung A (2004) Stochastic and epigenetic changes of gene expression in Arabidopsis polyploids. Genetics 167:1961–1973
Zhang LQ, Liu DC, Yan ZH, Lan XJ, Zheng YL, Zhou YH (2004) Rapid changes of microsatellite flanking sequence in the allopolyploidization of new synthesized hexaploid wheat. Sci China Ser C 47:553–561
Zou J, Zhu JL, Huang SM, Tian ET, Xiao Y, Fu DH, Tu JX, Fu TD, Meng JL (2010) Broadening the avenue of intersubgenomic heterosis in oilseed Brassica. Theor Appl Genet 120:283–290
Acknowledgments
The authors are grateful to Dr. Guijun Yan for critical reading of the manuscript. We appreciate to the reviewers for their critical comments, and to one of anonymous reviewers who edited the English across the whole manuscript. The study was supported by the Key Project of the National Natural Science Foundation of China (project code: 30830073).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Quiros.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tian, E., Jiang, Y., Chen, L. et al. Synthesis of a Brassica trigenomic allohexaploid (B. carinata × B. rapa) de novo and its stability in subsequent generations. Theor Appl Genet 121, 1431–1440 (2010). https://doi.org/10.1007/s00122-010-1399-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-010-1399-1