Abstract
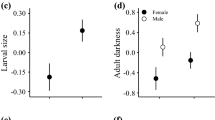
A resource allocation trade-off is expected when resources from a common pool are allocated to two or more traits. In holometabolous insects, resource allocation to different functions during metamorphosis relies completely on larval-derived resources. At adult eclosion, resource allocation to the abdomen at the expense of other body parts can be seen as a rough estimate of resource allocation to reproduction. Theory suggests geographic variation in resource allocation to the abdomen, but there are currently no empirical data on it. We measured resource allocation to the abdomen at adult eclosion in four geometrid moths along a latitudinal gradient. Resource (total dry material, carbon, nitrogen) allocation to the abdomen showed positive allometry with body size. We found geographic variation in resource allocation to the abdomen in each species, and this variation was independent of allometry in three species. Geographic variation in resource allocation to the abdomen was complex. Resource allocation to the abdomen was relatively high in partially bivoltine populations in two species, which fits theoretical predictions, but the overall support for theory is weak. This study indicates that the geographic variation in resource allocation to the abdomen is not an allometric consequence of geographic variation in resource acquisition (i.e., body size). Thus, there is a component of resource allocation that can evolve independently of resource acquisition. Our results also suggest that there may be intraspecific variation in the degree of capital versus income breeding.




Similar content being viewed by others
References
Arrese EL, Soulages JL (2010) Insect fat body: energy, metabolism, and regulation. Annu Rev Entomol 55:207–225. doi:10.1146/annurev-ento-112408-085356
Bernardo J (1996) Maternal effects in animal ecology. Am Zool 36:83–105. doi:10.1093/icb/36.2.83
Blanckenhorn WU (1994) Fitness consequences of alternative life histories in water striders, Aquarius remigis (Heteroptera: Gerridae). Oecologia 97:354–365. http://www.jstor.org/stable/4220627
Blanckenhorn WU, Demont M (2004) Bergmann and converse Bergmann latitudinal clines in arthropods: two ends of a continuum? Integr Comp Biol 44:413–424. doi:10.1093/icb/44.6.413
Blanckenhorn WU, Fairbairn DJ (1995) Life history adaptation along a latitudinal cline in the water strider Aquarius remigis (Heteroptera: Gerridae). J Evol Biol 8:21–41. doi:10.1046/j.1420-9101.1995.8010021.x
Boggs CL (1981) Nutritional and life-history determinants of resource allocation in holometabolous insects. Am Nat 117:692–709. http://www.jstor.org/stable/2460754
Briegel H (1990) Metabolic relationship between female body size, reserves, and fecundity of Aedes aegypti. J Insect Physiol 36:165–172
Burke S, Pullin AS, Wilson RJ, Thomas CD (2005) Selection for discontinuous life-history traits along a continuous thermal gradient in the butterfly Aricia agestis. Ecol Entomol 30:613–619. doi:10.1111/j.0307-6946.2005.00741.x
Chown SL, Gaston KJ (1999) Exploring links between physiology and ecology at macro-scales: the role of respiratory metabolism in insects. Biol Rev 74:87–120. doi:10.1111/j.1469-185X.1999.tb00182.x
Christians JK (2000) Trade-offs between egg size and number in waterfowl: an interspecific test of the van Noordwijk and de Jong model. Funct Ecol 14:497–501. doi:10.1046/j.1365-2435.2000.00444.x
de Jong G, van Noordwijk AJ (1992) Acquisition and allocation of resources: genetic (co)variances, selection, and life histories. Am Nat 139:749–770. http://www.jstor.org/stable/2462620
Diss AL, Kunkel JG, Montgomery ME, Leonard DE (1996) Effects of maternal nutrition and egg provisioning on parameters of larval hatch, survival and dispersal in the gypsy moth, Lymantria dispar L. Oecologia 106:470–477. http://www.jstor.org/stable/4221287
Ellers J (1996) Fat and eggs: an alternative method to measure the trade-off between survival and reproduction in insect parasitoids. Neth J Zool 46:227–235
Ellers J, van Alphen JJM (1997) Life history evolution in Asobara tabida: plasticity in allocation of fat reserves to survival and reproduction. J Evol Biol 10:771–785. doi:10.1046/j.1420-9101.1997.10050771.x
Fairbairn DJ (2005) Allometry for sexual size dimorphism: testing two hypotheses for Rench’s rule in the water strider Aquarius remigis. Am Nat 166:S69–S84. http://www.jstor.org/stable/3473067
Hatle JD, Crowley MC, Andrews AL, Juliano SA (2002) Geographic variation of reproductive tactics in lubber grasshoppers. Oecologia 132:517–523. doi:10.1007/s00442-002-0994-5
Hughes CL, Dytham C, Hill JK (2007) Modelling and analysing evolution of dispersal in populations at expanding range boundaries. Ecol Entomol 32:437–445. doi:10.1111/j.1365-2311.2007.00890.x
Hyönteistietokanta (2009) Database of entomological observations by Finnish Museum of Natural History, Helsinki. http://www.fmnh.helsinki.fi/insects/main/EntDatabase.html. Accessed 22 December 2009
Iwasa Y, Ezoe H, Yamauchi A (1994) Evolutionary stable seasonal timing of univoltine and bivoltine insects. In: Danks HV (ed) Insect life-cycle polymorphism. Kluwer Academic, Dordrecht, pp 69–89
Javoiš J, Molleman F, Tammaru T (2011) Quantifying income breeding: using geometrid moths as an example. Ent Exp Appl 139:187–196. doi:10.1111/j.1570-7458.2011.01120.x
Jervis MA, Boggs CL, Ferns PN (2005) Egg maturation strategy and its associated trade-offs: a synthesis focusing on Lepidoptera. Ecol Entomol 30:359–375. doi:10.1111/j.0307-6946.2005.00712.x
Jervis MA, Ellers J, Harvey JA (2008) Resource acquisition, allocation, and utilization in parasitoid reproductive strategies. Annu Rev Entomol 53:361–385. doi:10.1146/annurev.ento.53.103106.093433
Karl I, Lorenz MW, Fischer K (2007) Energetics of reproduction: consequences of divergent selection on egg size, food limitation, and female age for egg composition and reproductive effort in a butterfly. Biol J Linn Soc 91:403–418. doi:10.1111/j.1095-8312.2007.00806.x
Karlsson B, Johansson A (2008) Seasonal polyphenism and developmental trade-offs between flight ability and egg laying in a pierid butterfly. Proc R Soc B 275:2131–2136. doi:10.1098/rspb.2008.0404
Karlsson B, Wikman P-O (1989a) Abdomen size, body size and the reproductive effort of insects. Oikos 56:209–214. http://www.jstor.org/stable/3565338
Karlsson B, Wikman P-O (1989b) The cost of prolonged life: an experiment on a nymphalid butterfly. Funct Ecol 3:399–405. http://www.jstor.org/stable/2389613
Karlsson B, Wikman P-O (1990) Increase in reproductive effort as explained by body size and resource allocation in the speckled wood butterfly, Pararge aegeria (L.). Funct Ecol 4:609–617. http://www.jstor.org/stable/2389728
Karlsson B, Stjernholm F, Wiklund C (2008) Test of a developmental trade-off in a polyphenic butterfly: direct development favours reproductive output. Funct Ecol 22:121–126. doi:10.1111/j.1365-2435.2007.01334.x
King EG, Roff DA, Fairbairn DJ (2011) Trade-off acquisition and allocation in Gryllus firmus: a test of the Y model. J Evol Biol 24:256–264. doi:10.1111/j.1420-9101.2010.02160.x
Kivelä SM, Välimäki P, Oksanen J, Kaitala A, Kaitala V (2009) Seasonal clines of evolutionarily stable reproductive effort in insects. Am Nat 174:526–536. http://www.jstor.org/stable/27735863
Kivelä SM, Välimäki P, Carrasco D, Mäenpää MI, Oksanen J (2011) Latitudinal insect body size clines revisited: a critical evaluation of the saw-tooth model. J Anim Ecol 80:1184–1195. doi:10.1111/j.1365-2656.2011.01864.x
Mikkola K, Jalas I, Peltonen O (1989) Suomen perhoset, Mittarit 2. Recallmed/Lepidopterological Society of Finland, Hanko
Mousseau TA, Dingle H (1991) Maternal effects in insect life histories. Annu Rev Entomol 36:511–534. doi:10.1146/annurev.en.36.010191.002455
Mousseau TA, Fox CW (1998) The adaptive significance of maternal effects. Trends Ecol Evol 13:403–407
Mousseau TA, Roff DA (1989) Adaptation to seasonality in a cricket: patterns of phenotypic and genotypic variation in body size and diapause expression along a cline in season length. Evolution 43:1483–1496. http://www.jstor.org/stable/2409463
Pinheiro JC, Bates DM (2000) Mixed-effects models in S and S-plus. Springer, New York
Pinheiro J, Bates D, DebRoy S, Sarkar D, the R Core team (2009) nlme: linear and nonlinear mixed effects models. R package version 3.1-96. http://cran.r-project.org/. Accessed 15 Jan 2010
R Development Core Team (2009) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Roff D (1980) Optimizing development time in a seasonal environment: the ‘ups and downs’ of clinal variation. Oecologia 45:202–208. http://www.jstor.org/stable/4216080
Roff DA (2002) Life history evolution. Sinauer Associates, Sunderland
Tammaru T, Javoiš J (2000) Responses of ovipositing moths (Lepidoptera: Geometridae) to host plant deprivation: life-history aspects and implications for population dynamics. Environ Entomol 29:1002–1010. doi:10.1603/0046-225X-29.5.1002
van Noordwijk AJ, de Jong G (1986) Acquisition and allocation of resources: their influence on variation in life history tactics. Am Nat 128:137–142. http://www.jstor.org/stable/2461293
Warton D, Ormerod J (2007) smatr: (Standardised) Major Axis Estimation and Testing Routines. R package version 2.1. http://cran.r-project.org/. Accessed 15 Jan 2010
Warton DI, Wright IJ, Falster DS, Westoby M (2006) Bivariate line-fitting methods for allometry. Biol Rev 81:259–291. doi:10.1017/S1464793106007007
Wiklund C, Nylin S, Forsberg J (1991) Sex-related variation in growth rate as a result of selection for large size and protandry in a bivoltine butterfly, Pieris napi. Oikos 60:241–250. http://www.jstor.org/stable/3544871
Worley AC, Houle D, Barrett SCH (2003) Consequences of hierarchical allocation for the evolution of life-history traits. Am Nat 161:153–167
Acknowledgments
We thank W. Blanckenhorn, H. Kokko, M. Mutanen, K. Ronkainen, J. Oksanen, H. Pöykkö, T. Tammaru, and two anonymous reviewers for commenting on an earlier version of this manuscript. We are grateful to H. Pöykkö for help both in the field and in the lab, H. Huiskonen for help in rearing the larvae, M. Mutanen for providing us with some moths, J. Oksanen for processing the climate data, T. Pakonen for help in the elemental analysis, M. A. Jervis for proposing the protein and lipid analyses and giving instructions for them, and M.-L. Martimo-Halmetoja and M. Orreveteläinen for help in the protein and lipid analyses. The study was financed by the Ella and Georg Ehrnrooth foundation (grants to SMK and PV), Societas pro Fauna et Flora Fennica (SMK), Societas Biologica Fennica Vanamo (SMK), the Jenny and Antti Wihuri foundation (SMK), the Finnish Cultural Foundation (PV), and the Oskar Öflund foundation (PV). All Finland’s guidelines and legal requirements for the use of animals in research were followed.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Rights and permissions
About this article
Cite this article
Kivelä, S.M., Välimäki, P., Carrasco, D. et al. Geographic variation in resource allocation to the abdomen in geometrid moths. Naturwissenschaften 99, 607–616 (2012). https://doi.org/10.1007/s00114-012-0940-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-012-0940-2