Abstract
Human papillomavirus (HPV) is the most common sexually transmitted virus in the world. About 70% of cervical cancers are caused by the most oncogenic HPV genotypes of 16 and 18. Since available prophylactic vaccines do not induce immunity in those with established HPV infections, the development of therapeutic HPV vaccines using E6 and E7 oncogenes, or both as the target antigens remains essential. Also, knocking out the E6 and E7 oncogenes in host genome by genome-editing CRISPR/Cas system can result in tumor growth suppression. These methods have shown promising results in both preclinical and clinical trials and can be used for controlling the progression of HPV-related cervical diseases. This comprehensive review will detail the current treatment of HPV-related cervical precancerous and cancerous diseases. We also reviewed the future direction of treatment including different kinds of therapeutic methods and vaccines, genome-editing CRISPR/Cas system being studied in clinical trials. Although the progress in the development of therapeutic HPV vaccine has been slow, encouraging results from recent trials showed vaccine-induced regression in high-grade CIN lesions. CRISPR/Cas genome-editing system is also a promising strategy for HPV cancer therapy. However, its safety and specificity need to be optimized before it is used in clinical setting.
Similar content being viewed by others
Introduction
Human papillomavirus (HPV) is the most common sexually transmitted virus in the world [1]. HPV with more than 200 genotypes is associated with cervical cancers [2] and also anogenital and oropharyngeal cancers [3]. This gives rise to the nomenclature of high-risk types (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58 and 59) and low-risk types of HPVs (6, 22, 40, 42, 43, 44 and 54) based on the annual report of the International Agency for Research on Cancer evaluation (IARC) [1]. Cervical cancer remaining the fourth most common female malignancy and the second most common female cancer in woman aged 15 to 44 years is estimated to cause 91% of HPV-related cancer deaths [4]. Also, approximately 70% of cervical cancers are caused by the most oncogenic HPV genotypes of 16 and 18 [5].
This double-stranded DNA virus encodes eight open reading frames (ORFs) consisting of three functional parts: a) the early region (E1-E7) necessary for viral replications, cell cycle and signaling pathway, b) the late region (L1-L2) required for virion assembly and capsid formation, and c) a largely non-coding part referred to as the long control region (LCR) containing the Cis elements necessary for replication and transcription of viral DNA [6]. E1 and E2 proteins of HPV act as factors recognizing the origin of replication, E4 and E5 are believed to be involved in life cycles of virus, and E6 and E7, the main oncoprotein of HPVs, target the regulators of the cell cycle including p53 and retinoblastoma (Rb) proteins, respectively [7]. The E7 proteins of the high-risk HPV types such as HPV16 and HPV18 bind to Rb protein with a much higher affinity. One of the major biochemical functions of Rb is to bind E2F-family transcription factors mainly as repressors of the replication enzyme gene expression. This ability correlates with the tumor suppression function of Rb [8]. Finally, the L1 and L2 proteins form icosahedral capsids around the viral genome within the infected cells [6,7,8].
HPVs are perfectly adapted to the basal cells of cutaneous and mucosal epithelium of the host to exploit the cellular machinery for their own purposes. Infected cells act as the reservoir of the virus and during cell division, desquamation releases the viral particles as the viral replication cycle is completed [9]. As mentioned earlier, the viral proteins E1 and E2 are essential for basal DNA replication. Also, binding of E7 to Rb activates the E2F transcription factor leading to DNA replication [10]. Unscheduled S-phase would normally lead to apoptosis by the action of p53; however, in HPV-infected cells, the viral E6 protein targets p53 for proteolytic degradation [11]. Constant activity of viral oncoproteins E6 and E7 leads to increased genomic instability and accumulation of mutations and further loss of cell-growth control and cancer [12]. This constant level of E6 and E7 due to viral genome integration into the host chromosome is a hallmark of cervical cancer [13].
Today, the HPV vaccination program has been extended to prevent increasing incidence of HPV-related cancers especially cervical cancer. Three prophylactic FDA-approved vaccines including Gardasil and Gardasil 9 (Merck Sharp & Dhome-MSD) and Cervarix (Glaxo Smith Kline-GSK) are virus-like particles (VLP) based on L1 protein produced by recombinant technology which will prevent cervical cancer with almost 100% efficacy [14]. Tetravalent Gardasil protects against HPVs 6, 11, 16 and 18, bivalent Cervarix protects against HPVs 16 and 18 and nonavalant Gardasil 9 protects against HPVs 6, 11, 16, 18, 31, 33, 45, 52 and 58 [15]. These three HPV vaccines are highly immunogenic resulting in essentially 100% seroconversion. Gardasil, Gardasil 9 and Cervarix have shown to induce production of memory B cells and their protection against HPV 16/18 infections at least for 5 and 10 years, respectively. Extended age for vaccination (9–26 years in females) is also another advantage of these prophylactic vaccines [16, 17]. Major impediments such as economic barriers, storage and transportation, the so-called cold chain [18], lack of insurance coverage [19], lack of perceived benefit or need to vaccinate males [20] are hindering vaccine uptake around the world. Also, none of the prophylactic vaccines show therapeutic effect on existing HPV infection and related cervical disease [21].
According to IARC HPV information center at year 2021, the world has a population of 2,869 million woman aged above 15 years being at risk of the developing cervical cancer. Despite HPV vaccination, current estimates indicate that every year 604,127 women are diagnosed with cervical cancer and 341,831 die from the disease [5]. The disease endpoint of cervical intraepithelial neoplasia grade 2 or worse (CIN 2, CIN 3, adenocarcinoma in situ (AIS), adenocarcinoma and carcinoma) can be caused by high-risk HPV types. The most important aim of treatment of HPV-associated cervical lesions is to lower the chance of neoplasia. This review will summarize current and future direction in treatment of HPV-related cervical precancerous and cancerous diseases with the focus on novel strategies such as therapeutic vaccines and genome-editing method which are still under investigations in clinical trials.
Diagnostic consideration
As mentioned earlier, infection with HPVs can cause warts on cutaneous epithelium. Abnormal cervical epithelial cells can be detected either by examination or molecular tests. Genital warts can be flat, cauliflower-shaped, keratotic or dome-shaped. Accurate diagnosis of these morphological lesions is achieved by preparing the specimen for cervical cytology in two methods of Papanicolaou (Pap) test or the thin-layer liquid-based cytology method. For both methods, cells are obtained from the external surface of the cervix (ectocervix) and the cervical canal (endocervix) to evaluate the transformation zone (squamocolumnar junction), the area at greatest risk for neoplasia [22]. Cervical cytology can also be used to test for high-risk HPVs and whether the HPV has begun to cause dysplasia. Even with normal cytology on pap smear, 5.3% of samples will be positive for high-risk HPVs. High-grade squamous intraepithelial results over 90% positive for high-risk HPVs [23].
Since HPV early proteins including E6 and E7 have been shown to be positively related to the oncogenesis of HPV-induced cancers, these oncoproteins are promising biomarkers for diagnosis. More importantly, monoclonal antibodies (mAbs) as designed antibody mimics as well as new immunological kits have been developed for immunodiagnostic approaches using enzyme-linked immunosorbent assay (ELISA) method. For example, an IgG2a-type anti-HPV16 E7 mAb can be suitable for the detection of HPV16 E7 oncoprotein in HPV16-positive cervical carcinoma tissue [24]. In addition to oncogenic early proteins, HPV L1 and L2 late proteins can be also developed for the diagnosis of different HPV genotypes [25]. For example, a mAbs that specifically recognized HPV16 L1 can also be used for HPV18 L1, HPV31 L1, HPV45 L1 and HPV6 L1. In addition to detection of HPV protein antigens, there is an easier and more effective way of detection. Anti-HPV protein antibodies can be detected in exocrine samples of patients. For example, in cervical cancer patients, vaginal wash can be used to detect HPV protein antibodies. However, it is important to set up specific criteria for obtaining valid samples with high detection efficacy. Anti-HPV antibodies can be detected in the sera of patients underpinning the basis of the serological detection of anti-HPV antibodies [26]. However, serological testing for HPV has limited accuracy due to the lack of a robust host serological response [9, 27]. Thus, it is important to know that accurate diagnosis of HPV infection relies on the detection of viral nucleic acid. In situ hybridization being complementary to cytology including southern blot or dot spot hybridization, signal amplification systems and most importantly target amplification systems including polymerase chain reaction (PCR), real-time PCR and reverse transcriptase (RT) PCR are such molecular-based detection methods. PCR primer sets being used to detect a broad spectrum of HPV genotypes are aimed at L1 region of the virus since it’s the most conserved part of the genome [28]. Type-specific PCR primers combined with fluorescent probes can be used for quantification. Basically, PCR assays have high repeatability and reproducibility and they are very sensitive and specific. The increased information content, high throughput and low cost would facilitate the use of this real-time PCR-based assay in a variety of clinical settings [29].
Current methods of treatment
Although the ultimate goal of HPV vaccination is to prevent invasive cervical cancer, major impediments as mentioned above are hindering vaccine uptake around the world and therefore many women are diagnosed with cervical cancer. However, there are no data to indicate full treatment of HPV-related cervical diseases, the primary goal of the treatment is to ameliorate symptoms, remove the transformations zone of warts and reduce the risk of future invasive cervical cancer. Management and treatment of HPV-related disease are highly dependent on HPV types, available treatments and progression of the disease.
For treatment of external genital warts caused by non-oncogenic HPVs, recommended treatments includes Podophyllotoxin (an antimitotic agent destroying warts) [30,31,32,33,34,35,36,37,38,39,40,41,42,43,44], Imiquimod (an imidazoquinolinamine derivative inducing macrophages to secret IL-2 and IFN-α cytokines which were approved for HPV genital warts by the US Food and Drug Administration (FDA)) [45,46,47,48,49,50,51,52,53,54,55,56,57,58,59], and Sinecatechins (a compound derived from green tea leaves of the Camellia sinensis species containing active ingredient epigallocatechingallate with various immunomodulatory and antiproliferative properties activating the caspase pathway and inhibiting telomerase) [60,61,62,63,64,65] and Trichloroacetic acid 80–90%. [66,67,68]. Also, some limited therapies of external genital warts such as 5-fluorouarcil (an anti-metabolite blocking DNA synthesis) [69, 70], intralesional/topical interferon (pro-inflammatory cytokines with broad antiviral effects) [71,72,73,74], and photodynamic therapy (topical 5-aminolevulinic acid (ALA) as photosensitizer followed by irradiation) [75, 76] can be used in some circumstances.
For treatment of cervical precancerous lesions caused by oncogenic HPVs, surgical methods including excisional treatment with local anesthesia [77,78,79,80,81], cryosurgery(freezing) [77], electrosurgery also referred to as a cone biopsy or conization or loop electrosurgical excision procedure (LEEP) [82, 83] and laser therapy [84,85,86,87] is recommended. Actually, many studies suggest that the type of surgical approach does not influence the risk of recurrence [88,89,90,91]. So, depending on the availability of the method and the physician’s opinion, the treatment may vary.
Current treatments for cervical cancer contain adjuvant or neoadjuvant chemotherapy with radiation or total or radical hysterectomy. In the treatment of cervical cancer from locally to advanced and metastatic, chemotherapy is found to be efficacious. Women with distant metastatic and recurrent diseases traditionally have been treated with cisplatin-based chemotherapy [92]. Definitive radiation therapy with concurrent cisplatin-based chemotherapy (CRT), despite high recurrence rates (25–40%), is considered to be the standard in invasive cervical cancer [93]. Chemotherapy strategy can be conducted either as adjuvant chemotherapy after the surgery or as neoadjuvant chemotherapy (NACT) prior to the surgery with similar overall survival (OS) rates shown by meta-analyses [92, 94]. Also, recent studies and analyses have suggested that combination therapy with different chemotherapy agents such as Vinorelbine, Paclitaxel, Pemetrexed, Ifosfamide, Irinotecan, Topotecan, Capecitabine and S-1 increases the chance of treatment and can be prescribed based on the patient’s situation [95, 96].
Whether receiving therapy with CRT or NACT, many women undergo the surgery [97, 98]. Hysterectomy is a kind of surgery in which the uterus is removed. Among different types of hysterectomy, radical hysterectomy in which the uterus en bloc with the parametrium (i.e., round, broad, cardinal, and uterosacral ligaments) and the upper one-third to one-half of the vagina is removed has been confirmed to have the superiority [99]. Although, hysterectomy has a negative impact on fertility and can lead to many physical and mental disorders and thus affect the quality of life of the patients [100].
Recently, multiple studies have been published that investigate the effect of combining checkpoint inhibitors with current approaches. Immune checkpoint mechanism such as cytotoxic lymphocyte antigen 4 (CTLA-4) and programmed cell death protein 1 (PD-1) result in an immunosuppressive response leading to reduce T-cell activity. Multiple monoclonal antibodies (mAb) such as Ipilimumab, Pembrolizumab and Nivolumab have been developed and FDA-approved for targeting of the PD-1 axis in cervical cancer [101, 102].
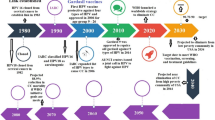
Summary of available treatments for external genital warts caused by non-oncogenic HPVs, cervical precancerous lesions and cervical cancer is shown in Table 1. In continue, we review the future direction in treatment of HPV-related cervical precancerous and cancerous lesions including therapeutic vaccines and genome-editing method which are still under investigation in clinical trials (Fig. 1).
Natural progress of human papilloma virus (HPV) infection and current and novel treatments: HPV establishes infection in the basal epithelial cells. A majority of infections are transient but about 10–20% of infections persist latently, leading to disease progression as illustrated by the red arrows. The lesions are known as cervical intraepithelial neoplasia (CIN) being classified based on severity. Low-grade squamous intraepithelial lesions (LSIL) will gradually advance to high-grade squamous intraepithelial lesions (HSIL) and ultimately leading to invasive carcinoma. The tumor regression in response to initial treatments is illustrated in green arrows. Current methods of treatment for each part are listed in green boxes. Different novel methods of treatment for LSIL, HSIL and cervical cancer are listed in blue box
Future direction in treatment of HPV-related cervical precancerous and cancerous lesions
As mentioned earlier, due to sub-optimal prophylactic HPV vaccination rates worldwide, HPV infections and subsequent development of HPV-associated malignancies are still public health issues. Thus, the development of therapeutic HPV vaccines and other therapies including genome-editing method represent a pressing public health concern. Most therapeutic vaccines and genome-editing method have used E6, E7, or a combination of both as the target antigens. Several different novel approaches for cervical precancerous and cancerous lesions will be discussed in this report with examples of vaccines at different stages of development. Current clinical trials for therapeutic vaccines and genome-editing methods were queried at the NIH ClinicalTrials.gov database. Terminated and withdrawn clinical studies were excluded in this report and the results are summarized in Tables 2–6.
Therapeutic vaccines
Limited global vaccine uptake, limited cross-protection, cost and requirement for a cold chain are such reasons that there is still a considerable population suffering from HPV infection despite vaccination. Here, therapeutic vaccines unlike prophylactic vaccines are aimed at generating cell-mediated immunity rather than neutralizing antibodies [104]. Thus, a therapeutic vaccine can have an immediate impact on patients who are already infected. Generally, HPV infection is an ideal target for therapeutic vaccination as the pre-invasive stages are well-known and progression to cancer takes up to 30 years. This window provides an ample opportunity for intervention [105]. Therapeutic vaccines require rational design to achieve concentrated antigen delivered to antigen-presenting cells (APCs) such as dendritic cells (DCs) to efficiently prime and activate cellular immune response [106]. A number of therapeutic vaccines have been developed targeting E6 and E7 including live vector vaccines, peptide/protein-based vaccines, nucleic acid-based vaccines. These vaccines have shown promising results in both preclinical and clinical trials and can be a novel option to control the progression of HPV-related cervical precancerous and cancerous diseases. However, HPV E5 oncoprotein was used in some preclinical studies, but it was not efficient enough in clinical trials, up to now. For instance, injection of E5 peptide combined with CpG led to strong cell-mediated immunity and protected mice from tumor growth [107]. Yet, further study on preclinical levels is needed to investigate the efficacy of E5. In continue, different therapeutic vaccines are reviewed as follows.
Live vector-based vaccines:
Live vector-based vaccines encompass bacterial and viral vectors as follows. These vectors are highly immunogenic and as they replicate within host cells, they facilitate the spread of antigens. These vectors deliver the E6 and E7 antigens to the DCs which stimulate antigen expression through major histocompatibility complex (MHC) class I (cytotoxic T-cell (CD8+)) or class II (helper T-cell (CD4+)) [108, 109]. However, these vectors pose a potential safety risk to immunocompromised patients and the immune response efficacy after repeated immunization using the same vector is also limited [110, 111].
-
a)
Bacterial vectors-based vaccines: Several bacteria including Listeria monocytogenes, Lactobacillus Lactis, Lactobacillus plantarum and Lactobacillus casei can deliver genes or proteins of interest such as E6 and E7 to antigen-presenting cells [112,113,114,115]. Listeria is a food-borne pathogen that is typically ingested in unpasteurized dairy products. It is able to evade phagosomal lysis due to secretion of listeriolysin O (LLO) which is a pore-forming toxin [116]. This feature allows the antigen peptides in the bacteria to be presented on both MHC class I and II pathway since Listeria can be present in both cell cytoplasm and endosomal compartments [117, 118]. The first clinical use of live-attenuated Listeria-based therapeutic HPV vaccine (Lm-LLO-E7 also known as ADXs11-001 or ADXs-HPV) was reported in 2009 [119]. The phase I trial tested the safety of the vaccine in 15 patients with metastatic or refractory advanced squamous cell carcinoma of the cervix. The Lm-LLO-E7 vaccine was well tolerated by patients. Thus, scientists planed and designed additional clinical trials to determine the efficacy of the vaccine in phase II trials including NCT02853604, NCT02399813, NCT02002182, NCT02291055 and NCT01266460. Clinical trials for therapeutic HPV bacterial-vectored vaccines are summarized in Table 2.
-
b)
Viral vectors-based vaccines: Several viruses including adenoviruses, adeno-associated viruses, alphaviruses, lentiviruses and vaccinia viruses have also been examined to deliver HPV E6 and E7 antigens [120,121,122,123]. Among these viruses, vaccinia is a promising vector for vaccine delivery due to its large genome, highly infectious nature and low likelihood of unregulated integration of foreign DNA into its genome [124]. Vaccinia-based vaccines include vaccinia encoding fusion of E7 and Calreticulin (CRT) to enhance MHC class I processing in DCs and vaccinia encoding E7 and listeriolysin O to facilitate MHC class I and II presentation [125]. The recombinant vaccinia virus vaccine known as TA-HPV (NCT00002916) was first used in patients with advanced-stage cervical cancer. Phase I and II clinical trials have shown that the TA-HPV induces potent antigen-directed antibody and cytotoxic responses in patients with cervical cancer [126]. One challenge of using live vector-based therapeutic HPV vaccine is the generation of antibacterial or antiviral immune response through expression of cytokines or co-stimulatory molecules. Another hurdle to overcome is the generation of neutralizing antibodies after initial vaccine exposure which prevents the effective administration. In this regard, studies show that cyclooxygenase 2 (COX-2) inhibitors such as Celecoxib prevent the production of neutralizing antibodies to vaccinia virus allowing repeated administration without losing infectivity a promising advance [127]. The immunological activity, safety and tolerability of an alphavirus-based therapeutic vaccine (Vvax001) against HPV-induced cancers were investigated in human Phase I trial. Vvax001 consists of replication-incompetent Semliki Forest virus replicon particles encoding HPV16 E6 and E7 antigens [128]. Harper et al. showed that the Tipapkinogen Sovacivec (TS) therapeutic HPV vaccine provides histologic clearance of CIN2/3 irrespective of high-risk HPV type in one third of subjects and is generally safe through 30 months. TS is a highly attenuated replication-deficient strain of vaccinia virus (MVA) encoding three proteins including human cytokine IL-2 and modified forms of HPV16 E6 and E7 proteins as non-oncogenic [129]. Clinical trials for therapeutic HPV viral-vectored vaccines are summarized in Table 2.
Peptide/protein-based vaccines:
Peptide/protein-based vaccines are safe, stable and easy to produce and are derived from HPV antigens which can be processed by DCs and presented on either MHC I or II [130]. In continue, we investigate these types of vaccines. Clinical trials for therapeutic HPV peptide/protein-based vaccines are summarized in Table 3.
-
a)
Peptide-based vaccines: In spite of being safe and stable and easy to produce, peptide vaccines are restricted by the necessity to identify immunogenic epitopes corresponding to the polymorphic MHC molecules within populations. In this case, in silico approaches such as epitope prediction and mapping, molecular modeling and structural vaccinology can predict the highly immunogenic epitopes in a short time with high specificity and can be used for development of an effective vaccine [21]. The peptide vaccines also have poor immunogenicity and require adjuvants to enhance the vaccine potency [130]. HPV16 synthetic long-peptide vaccine (HPV16-SLP) consists of both E6 and E7 overlapping peptides with Montanide ISA-51 as an adjuvant is a peptide-based vaccine that has been studied extensively [131]. The therapeutic potential of HPV16-SLP vaccine has been evaluated in NCT01923116, NCT02426892 and NCT02128126. The ISA101 in phase I/II clinical trial (NCT02128126) has been studied in combination with chemotherapy agents such as carboplatin and paclitaxel with or without bevacizumab as an immunotherapy strategy.
-
b)
Protein-based vaccines: Like peptide-based vaccines, protein-based vaccines suffer from low immunogenicity. Strategies to improve their potency are similar to those employed in peptide-based vaccine. One benefit to using protein-based vaccine is that they contain all epitopes of human leukocyte antigen (HLA) avoiding the limitation of MHC restriction. Although, protein-based vaccines are presented via MHC class II pathway which means they activate the production of antibodies rather than generating a T-cell response [132]. Strategies to overcome this problem focus on enhancing the MHC class I presentation. For example, adjuvants and immunostimulant molecules such as liposome-polycationic-DNA carrier particle [133] and the saponin-based ISCOMATRIX [134] can increase endogenous processing and further MHC class I expression of antigen. A protein-based vaccine that has progressed to clinical trials is TA-CIN which contains a fusion protein composed of HPV16 L2, E6 and E7 [135]. The safety and efficacy of TA-CIN in combination with adjuvant GPI-0100 are currently investigated in HPV16 associated cervical cancer in clinical trial NCT02405221. GTL001 (ProCervix) is another protein-based vaccine composed of both HPV 16 and 18 [136] E7 proteins. However, in phase II clinical trial NCT01957878, the GTL001 wasn’t superior to placebo in viral clearance. In general, the future of protein-based vaccines relies upon the enhancement of immunogenicity and T-cell response through adjuvant and fusion protein strategies.
Nucleic acid-based vaccines:
Nucleic acid-based vaccines are safe and easy to manufacture and purify. These types of therapeutic vaccines do not produce neutralizing antibodies allowing for repeated vaccination [137]. In continue, we go deeper and investigate the pros and cons of these vaccines. Clinical trials for therapeutic HPV nucleic acid-based vaccines are summarized in Table 4.
-
a)
DNA-based vaccines: DNA vaccines have been studied extensively and proven to be safe in clinical trials [138, 139]. This involves the delivery of plasmid DNA encoding a protein of interest into the host’s tissue leading to expression and production of protein. The plasmid is often a bacterial plasmid containing a strong viral promoter which controls the expression of the protein of interest. The bacterial DNA backbone can induce innate immune response through recognition of CpG motifs being a ligand for toll-like receptor 9 (TLR9) on dendritic cells (DCs) and B-cells and natural killers (NKs) [140]. DNA vaccines are believed to induce adaptive cellular immune responses via MHC class I or II-associated antigen presentation. DNA vaccines have benefit of simplicity, thermostability, low cost of manufacture, potential to elicit both cellular and humoral immunity, absence of anti-vector immunity, excellent safety and tolerability in humans. Although, DNA vaccines have limited immunogenicity, thus should be administered repeatedly. Immunogenicity of DNA vaccines can be improved by using them in a heterologous prime-boost vaccination strategy. Using both traditional and novel immunization approaches provides exciting opportunities to elicit unique immune responses to allow for improved immunogenicity [141]. Cellular uptake of naked DNA is inefficient in vivo and the bulk of injected DNA remains extracellular [142]. Codon optimization can improve the expression [143] and the addition of a leader sequence can target the antigens into the endoplasmic reticulum [144]. Also, electroporation (EP) can greatly enhance vaccine uptake. However, a variety of DNA-based vaccines were developed in preclinical studies which some of them were achieved to clinical trials. Recently, DNA vaccines using doggybone DNA (dbDNA) have been developed without the use of bacteria. Allen et al. showed that linear doggybone DNA vaccine induces similar immunological responses to conventional plasmid DNA. It is interesting that these responses were independent of immune recognition by TLR9 in a preclinical model [145].
An important example of therapeutic DNA-vectored HPV vaccine is VGX-3100 (Inovio Biomedical Corp/VGX pharmaceutical) containing two DNA plasmids encoding optimized synthetic consensus E6 and E7 genes of HPV16/18. This trial addressed two important issues that a therapeutic vaccine could induce adaptive immune response in patients with existing disease and systemically administration of the vaccine can elicit adaptive immune responses. The phase II study of VGX-3100 (NCT03180684) was followed by electroporation alone or in combination with imiquimod and phase III study of VGX-3100 (REVEAL 1, NCT03185013) is currently underway to confirm the efficacy and safety of the vaccine followed by electroporation.
-
b)
RNA-based vaccines: The RNA replicons are naked RNA sustaining cellular antigen expression due to self-limiting replication. Thus, RNA vaccines produce more antigenic protein than DNA vaccines. Also, in contrast to DNA vaccines, RNA vaccines only have to cross the plasma membrane but not the nuclear membrane since the mRNA transcripts are translated directly in the cytoplasm once the vaccine is administrated and internalized by host cells resulting in an improved probability of successful transfection. RNA vaccines can also activate innate immune system since the mRNA is a natural TRL7/8 ligand [146]. Although, delivery systems in vivo are still being tested and optimized since this technology is new. There is also potential for RNA vaccines to cause toxicity due to the inherent inflammatory activity of mRNA [147]. RNA vaccines have been pioneered in many other malignancies, but few are available for HPV as they are notoriously unstable. HPV 16 RNA-LPX is one of the studies using the mRNA encapsulated in lipoplex so that it was selectively taken up by DC in lymphoid compartments [148]. Encapsulating can protect the mRNA from nuclease degradation resulting in enhanced cell uptake and delivery efficiency. The phase I HARE-40 (NCT03418480) evaluating the HPV16 RNA-LPX vaccine with and without anti-CD40 is the only RNA vaccine for HPV-associated disease that has been reached in clinical trials.
Whole cell-based vaccines:
Whole cell-based vaccines involve the patient’s APCs being directly loaded with HPV antigens and infused back into the patient. This platform has the advantage for antigen-specific cells being introduced directly to the patient which eliminates the trial and error associated with generating antigen-specific cells within the tumor microenvironment via vaccine. Although, this approach has HLA restriction and generating personalized whole cell-based therapies is time-consuming. In continue, we further investigate these vaccines. Clinical trials for therapeutic HPV cell-based vaccines are summarized in Table 5.
-
a)
DC-based vaccines: This strategy is created to enhance T-cell mediated immunity by loading DCs with HPV antigens and deliver them to the patients. DCs can be prepared ex vivo by the physical loading of MHC I and II molecules and antigen loading can be accomplished by pulsing the DCs with antigenic peptides or proteins or transfecting with DNA or RNA encoding HPV antigens [149]. Effective loading of tumor antigen into DCs can be achieved through gene delivery to DCs by targeting adenoviral vectors to CD40 with specific antibodies. NCT0015766 and NCT03870113 are two studies on DC-based vaccines that have been reached the clinical trials and yet are being investigated for safety, immunologic and clinical responses.
-
b)
Tumor cell-based vaccines: Manipulating tumor cells ex vivo being used to express immunomodulatory proteins such as IL-2, IL-12 and GM-CSF cytokines can enhance immunogenicity and be used as an approach for whole-cell immunization [150]. The use of tumor cell-based vaccine is an interesting strategy if the tumor antigens are unknown. However, there is always a safety concern about injecting tumor cells into patients. Tumor cell-based HPV vaccines have been tested in preclinical models but not yet in clinical trials. For example, GM-CSF-transduced autologous or allogenic tumor cells have been used in clinical trials for many other cancers. Also, tumor cell-based vaccines have limited scope for HPV vaccine development since it’s difficult to be produced on large scale [111, 151].
The genome-editing by CRISPR/Cas
Among different gene-editing strategies, clustered regularly interspaced short palindromic repeats (CRISPR) in combination with CRISPR-associated protein (Cas) are an interesting platform. Generally, gene-editing generate pre-designed alterations in the genome by inducing double-strand breaks (DSBs) or single-strand breaks (SSBs) (also termed ‘nicks’) resulting in the activation of endogenous repair mechanism [152]. CRISPR/Cas, the 2020 noble prize winner in chemistry, is a fast, cheap and efficient genome-editing method [153] which has been rooted in adaptive immunity in prokaryotes. Short stretches of invading foreign nucleic acids, so-called protospacers, are incorporated into the CRISPR locus of the bacterial or archaeal genome. For genome-editing process, a single guide RNA (sgRNA) binds complementary to DNA target and guides the Cas protein to the desired target site to create a DSB [153]. The protospacer adjacent motif (PAM) is the absolute prerequisite for Cas protein to induce a DSB. In cancer diseases which are driven by the overexpression of key oncogenes, a knock-out CRISPR/Cas system can be designed to overcome future malignancies.
As mentioned earlier, persistent infection with high-risk HPVs, especially types 16 and 18, is due to overexpression of E6 and E7 oncogenes. Indeed, knocking out the E6 and E7 oncogenes by CRISPR/Cas system can result in tumor growth suppression [154,155,156]. Targeting E6 oncogene can result in reactivation of p53 tumor suppressor pathway and targeting E7 oncogene can result in restoration of the retinoblastoma protein (Rb) tumor suppressor pathway. In one in vitro study, Zheng et al. designed an E7 sgRNA that targeted HPV16-E7 knocking down E7 oncogene, upregulating pRB, and inhibiting HPV cancer cell growth [156]. In another study, Zhen et al. designed a few sgRNAs that targeted E6 and E7 oncogene promoters and transcripts resulting in pronounced expression of p53 and pRB [154]. In one valuable in vivo study in 2019, Jubair et al.delivered the E6 and E7 sgRNA in CRISPR/Cas9 vector systemically into mice and explored the post-gene editing events in tumors thereby showed the effective cell death induced by apoptosis [155].
Despite success in targeting E6/E7 with CRISPR/Cas in pre-clinical models, previous efforts failed to address a range of issues. First, the delivery of the treatment vector has proven challenging owing to the large size of the CRISPR/Cas vector. To overcome this challenge, CRISPR/Cas vectors can be packaged into liposomes or adenovirus-associated vectors (AAV). Second, the development of precise gene-editing tools in the clinic requires careful consideration of the medical implications of permanent modification in the genome. Thus, the on-target and off-target effects of designed CRISPR/Cas vector should be investigated to provide sufficient targeted editing [157]. This issue is almost resolved by evolution of bioinformatics software tools. Recently, there are many simple and functional servers for designing CRISPR/Cas gRNAs with reduced off-target sites [158]. Also, easy monitoring methods based on polymerase chain reaction (PCR) or sanger sequencing or even next-generation sequencing (NGS) are other solutions to detect the off or on-target effects of designed CRISPR/Cas. Third, due to the role of p53 in multiple DNA damage response mechanisms, it’s hard to investigate the exact influence of p53. Although recently there are guidelines for CRISPR screening to ensure the quality of screening performance [159]. Finally, the ability and specificity of the Cas9, the most commonly used CRISPR/Cas system, is limited. Recently, many engineered forms of Cas9 proteins with improved targeting specificities have been developed to generate increasing gene-targeting efficiency and fidelity [160].
The enormous volume of study over CRISPR/Cas indicates that this new treatment strategy is the future of cancer treatment if the studies effectively addressed the mentioned challenges. Now, some trials are underway in five treatment areas including blood disorders, cancers, eye disease, chronic infections and protein-folding disorders. For example, CRISPR-based technology for sickle cell disease (SCD) and transfusion-dependent betta-thalassemia (TDT) aims to increase levels of fetal hemoglobin which can take place of defective adult hemoglobin in red blood cells [161]. The first in vivo CRISPR-based therapy trial, meaning to edit a gene within patient’s own body, is for treatment of Leber Congenital Amaurosis-10 (LCA10). In this case, CRISPR edits the patient’s mutated photoreceptor gene. If enough cells are edited, patients are expected to regain vision [162]. Also, the first trial to use CRISPR/Cas3-edited bacteriophages is for treatment of urinary tract infection (UTI) by Escherichia coli (E. coli). In this case, Cas3 protein, which targets longer stretches of DNA for destruction, rather than Cas9, makes a more precise cut at one location [163]. As resistance to traditional antibiotics like penicillin becomes a major public health threat, there is growing interest in developing bacteriophage therapy. At last, the first trial using lipid nanoparticles to deliver the genome-editing treatment systemically is for treatment of hereditary transthyretin amyloidosis (hATTR). In this case, researchers try to reduce the amount of mutated TTR proteins. Less amount of mutated TTR proteins decrease the chance of formation and accumulation amyloidosis [164]. Although many CRISPR-based therapies for different genetic diseases are now in clinical trials, there is only one phase I clinical trial for HPV. The NCT03057912 is the only HPV-related CIN I treatment that investigate the safety and efficacy of CRISPR/Cas9 for editing the E6 and E7 oncogenes of HPV16 and 18. Table 6 has listed all available clinical trials based on CRISPR/Cas technology.
Other advanced therapeutic methods
Besides novel therapeutic vaccines, there are few advanced methods yet need to be expanded. Recently, adoptive T-cell therapy (ATC) using engineered T-cells redirected by genes that encode for tumor-specific T-cell receptors (TCRs) or chimeric antigen receptors (CARs) has demonstrated a delightful potency in cancer treatment. In chimeric antigen receptor (CAR)-T immunotherapy, researchers genetically engineer patient’s T-cells to have a receptor that recognizes the patient’s cancer cells. Along with CAR-T immunotherapy, researchers use CRISPR-based technology as an immunotherapy approach known as checkpoint inhibition which edits the PD-1 gene in T-cells [165]. The landmark of CAR-T therapy is the commercial CD19 specific approved by FDA for acute lymphocytic leukemia (ALL). In HPV-positive cervical cancer, few clinical studies including NCT02280811, NCT02858310, NCT03578406, NCT03356795 and NCT04556669 have been in progress but more clinical evidence regarding the efficiency of CAR-T therapy for cervical cancer is required [166].
Another advanced therapeutic method is radioimmunotherapy (RIT) which uses tumor antigen-specific mAb for targeted delivery of cytocidal ionizing radiation to tumor cells [103]. In this method, radiolabeled mAbs systematically are administered to bind to specific tumor-associated antigens [104]. Radiolabeled E6 or E7-specific mAbs with a beta-emitter 188-Rhenium or beta-emitters 177-lutetium bind to extracellular E6 and E7, and deliver cytotoxic radiation to the area [105, 106]. Surviving tumor cells with weak or no E6 or E7 expression are also killed by radiation via the “cross-fire” effect produced by radiation in 360° spheres [107]. This potentially makes RIT of HPV-related cervical cancer a clinically important therapeutic modality in near future.
Conclusion and future perspectives
With increasing prophylactic HPV vaccination rates among the general population, the rates of HPV infection are set to drop. Cervarix, Gardasil and Gardasil 9 had shown to be highly immunogenic resulting in essentially 100% seroconversion. Yet there is an unmet need for HPV therapeutic vaccination as the burden of HPV malignancies will remain high for years to come. The available treatments for HPV-related cervical disease usually work on topical genital warts and no gold standard exists for the treatment. Furthermore, treatment choice depends on the severity of the disease, type of HPV infection and patient’s preferences. This article aims to offer available and the future direction in treatment of HPV-related cervical precancerous and cancerous lesions. Although the progress in the development of targeted therapeutic HPV vaccine has been slow, efforts to increase immunogenicity of T-cell response including varying delivery systems, vaccine composition, routes of delivery and different kinds of adjuvants should be made to promote treatment. Indeed, encouraging results from recent trials showed vaccine-induced regression in high-grade CIN lesions. For example, viral vector-based vaccines have high immunogenicity and can facilitate intracellular antigen spreading. Bacterial vector-based vaccines can deliver either engineered plasmids or HPV tumor proteins to APCs. Peptide and protein-based vaccines are stable, safe and easy to produce. DNA-based vaccines have a sustained expression of antigen on MHC-peptide complex. RNA-based vaccines have enhanced antigen expression, and whole cell-based vaccine potency can be enhanced by cytokine treatment. These features of therapeutic vaccines provide opportunities for broad, more effective and less toxic treatment strategy. At least for now, to advance the field, precise design of clinical trials evaluating different side effects and dosing of interventions, and investigating intended effects in the intended target tissues at a molecular level is necessary. Understanding the molecular biology of the tumor may favor the development of more effective therapeutic vaccines. Also, further clinical trials are needed to improve the clinical efficacy of therapeutic vaccines. As HPV-related cervical cancer express both viral and neo-antigens, the potential for synergy between these agents and therapeutic vaccines should be investigated in future clinical trials. Also, the CRISPR/Cas genome-editing system is a viable route of treatment that could supplement or potentially replace the current treatments of surgery, chemo and radiation therapy. The enormous volume of study over CRISPR/Cas indicates that this new treatment strategy is the future of cancer treatment. There are also few advanced methods like CAR-T therapy and radioimmunotherapy showing promising results. However, these methods will still be necessary to optimize their efficacy, safety and specificity before they are used in clinical practice. We believe that with continued understanding of the nature and function of programmable Cas nuclease, the outlook is bright for a precise gene therapy in cervical cancer.
Availability of data and materials
Not applicable.
References
Brüggmann D, Kayser L, Jaque J, Bundschuh M, Klingelhöfer D, Groneberg DA (2018) Human papilloma virus: Global research architecture assessed by density-equalizing mapping. Oncotarget 9:21965
Wiley D, Douglas J, Beutner K, Cox T, Fife K, Moscicki A-B, Fukumoto L (2002) External genital warts: diagnosis, treatment, and prevention. Clin Infect Dis 35:S210–S224
Ljubojevic S, Skerlev M (2014) HPV-associated diseases. Clin Dermatol 32:227–234
Organization WH (2021) WHO guideline for screening and treatment of cervical pre-cancer lesions for cervical cancer prevention: web annex A: syntheses of evidence
Bruni LAG, Serrano B, Mena M, Collado JJ, Gómez D, Muñoz J, Bosch FX, de Sanjosé S (2021) ICO/IARC Information Centre on HPV and Cancer (HPV Information Centre). Human papillomavirus and related diseases in the world. Summ Rep 22 October 2021
Liu Y, Pan Y, Gao W, Ke Y, Lu Z (2017) Whole-genome analysis of human papillomavirus types 16, 18, and 58 isolated from cervical Precancer and Cancer samples in Chinese women. Sci Rep 7:1–9
Yim E-K, Park J-S (2005) The role of HPV E6 and E7 oncoproteins in HPV-associated cervical carcinogenesis. Cancer Res Treat: Official J Korean Cancer Assoc 37:319
Lipinski MM, Jacks T (1999) The retinoblastoma gene family in differentiation and development. Oncogene 18:7873–7882
Hathaway JK (2012) HPV: diagnosis, prevention, and treatment. Clin Obstet Gynecol 55:671–680
Münger K, Baldwin A, Edwards KM, Hayakawa H, Nguyen CL, Owens M, Grace M, Huh K (2004) Mechanisms of human papillomavirus-induced oncogenesis. J Virol 78:11451–11460
Thomas M, Pim D, Banks L (1999) The role of the E6–p53 interaction in the molecular pathogenesis of HPV. Oncogene 18:7690–7700
Duensing S, Münger K (2004) Mechanisms of genomic instability in human cancer: insights from studies with human papillomavirus oncoproteins. Int J Cancer 109:157–162
Bernard H-U (2002) Gene expression of genital human papillomaviruses and considerations on potential antiviral approaches. Antivir Ther 7:219–237
White MD (2014) Pros, cons, and ethics of HPV vaccine in teens—Why such controversy? Translational andrology and urology 3:429
Toh ZQ, Kosasih J, Russell FM, Garland SM, Mulholland EK, Licciardi PV (2019) Recombinant human papillomavirus nonavalent vaccine in the prevention of cancers caused by human papillomavirus. Infection and drug resistance 12:1951
Madrid-Marina V, Olea EG, Torres-Poveda KJ (2015) 8. Impact of prophylactic HPV vaccination on the cervical cancer incidence and mortality. Perspectives for development of therapeutic HPV vaccines. Research Signpost 155–188
Arbyn M, Xu L (2018) Efficacy and safety of prophylactic HPV vaccines. A Cochrane review of randomized trials. Expert Rev Vaccines 17:1085–1091
Namvar A, Bolhassani A, Javadi G, Noormohammadi Z (2019) In silico/In vivo analysis of high-risk papillomavirus L1 and L2 conserved sequences for development of cross-subtype prophylactic vaccine. Sci Rep 9:1–22
Dorell CG, Yankey D, Santibanez TA, Markowitz LE (2011) Human papillomavirus vaccination series initiation and completion, 2008–2009. Pediatrics 128:830–839
Gutierrez B Jr, Leung A, Jones KT, Smith P, Silverman R, Frank I, Leader AE (2013) Acceptability of the human papillomavirus vaccine among urban adolescent males. Am J Mens Health 7:27–36
Namvar A, Panahi HA, Agi E, Bolhassani A (2020) Development of HPV 16, 18, 31, 45 E5 and E7 peptides-based vaccines predicted by immunoinformatics tools. Biotech Lett 42:403–418
Bongaerts O, Clevers C, Debets M, Paffen D, Senden L, Rijks K, Ruiten L, Sie-Go D, Van Diest PJ, Nap M (2018) Conventional microscopical versus digital whole-slide imaging-based diagnosis of thin-layer cervical specimens: A validation study. J Pathol Inform 9
Mayrand MH, Duarte-Franco E, Coutlée F, Rodrigues I, Walter SD, Ratnam S, Franco EL, Group CS (2006) Randomized controlled trial of human papillomavirus testing versus Pap cytology in the primary screening for cervical cancer precursors: design, methods and preliminary accrual results of the Canadian cervical cancer screening trial (CCCaST). Int J Cancer 119:615–623
Salazar-Piña DA, Pedroza-Saavedra A, Cruz-Valdez A, Ortiz-Panozo E, Maldonado-Gama M, Chihu-Amparan L, Rodriguez-Ocampo AN, Orozco-Fararoni E, Esquivel-Guadarrama F, Gutierrez-Xicotencatl L (2016) Validation of serological antibody profiles against human papillomavirus type 16 antigens as markers for early detection of cervical cancer. Medicine 95
Le Cann P, Touze A, Enogat N, Leboulleux D, Mougin C, Legrand MC, Calvet C, Afoutou JM, Coursaget P (1995) Detection of antibodies against human papillomavirus (HPV) type 16 virions by enzyme-linked immunosorbent assay using recombinant HPV 16 L1 capsids produced by recombinant baculovirus. J Clin Microbiol 33:1380–1382
Ramezani A, Aghakhani A, Soleymani S, Bavand A, Bolhassani A (2019) Significance of serum antibodies against HPV E7, Hsp27, Hsp20 and Hp91 in Iranian HPV-exposed women. BMC Infect Dis 19:1–8
Dong Z, Hu R, Du Y, Tan L, Li L, Du J, Bai L, Ma Y, Cui H (2021) Immunodiagnosis and immunotherapeutics based on human papillomavirus for HPV-induced cancers. Front Immunol 11:3408
Molijn A, Kleter B, Quint W, van Doorn L-J (2005) Molecular diagnosis of human papillomavirus (HPV) infections. J Clin Virol 32:43–51
Liao Y, Zhou Y, Guo Q, Xie X, Luo E, Li J, Li Q (2013) Simultaneous detection, genotyping, and quantification of human papillomaviruses by multicolor real-time PCR and melting curve analysis. J Clin Microbiol 51:429–435
Beutner KR, Wiley DJ, Douglas JM, Tyring SK, Fife K, Trofatter K, Stone KM (1999) Genital warts and their treatment. Clin Infect Dis 28:S37–S56
Greenberg MD, Rutledge LH, Reid R, Berman NR, Precop SL, Elswick RK Jr (1991) A double-blind, randomized trial of 0.5% podofilox and placebo for the treatment of genital warts in women. Obstet Gynecol 77:735–739
Von Krogh G, Lacey C, Gross G, Barrasso R, Schneider A (2000) European course on HPV associated pathology: guidelines for primary care physicians for the diagnosis and management of anogenital warts. Sexually transmitted infections 76:162–168
Kirby P, Dunne A, King DH, Corey L (1990) Double-blind randomized clinical trial of self-administered podofilox solution versus vehicle in the treatment of genital warts. Am J Med 88:465–469
Claesson U, Lassus A, Happonen H, Hogstrom L, Siboulet A (1996) Topical treatment of venereal warts: a comparative open study of podophyllotoxin cream versus solution. Int J STD AIDS 7:429–434
Edwards A, Atma-Ram A, Thin RN (1988) Podophyllotoxin 0.5% v podophyllin 20% to treat penile warts. Sex Transm Infect 64:263–265
Beutner K, Friedman-Kien A, Artman N, Conant M, Illeman M, Thisted R, King D (1989) Patient-applied podofilox for treatment of genital warts. The Lancet 333:831–834
Mazurkiewicz W, Jablońska S (1990) Clinical efficacy of Condyline (0.5% podophyllotoxin) solution and cream versus podophyllin in the treatment of external condylomata acuminata. J Dermatol Treat 1:123–125
von Krogh G, Szpak E, Andersson M, Bergelin I (1994) Self-treatment using 0.25%-0.50% podophyllotoxin-ethanol solutions against penile condylomata acuminata: a placebo-controlled comparative study. Sex Transm Infect 70:105–109
Strand A, Brinkeborn R, Siboulet A (1995) Topical treatment of genital warts in men, an open study of podophyllotoxin cream compared with solution. Sex Transm Infect 71:387–390
Lacey C, Goodall R, Tennvall GR, Maw R, Kinghorn G, Fisk P, Barton S, Byren I (2003) Randomised controlled trial and economic evaluation of podophyllotoxin solution, podophyllotoxin cream, and podophyllin in the treatment of genital warts. Sex Transm Infect 79:270–275
Komericki P, Akkilic-Materna M, Strimitzer T, Aberer W (2011) Efficacy and safety of imiquimod versus podophyllotoxin in the treatment of anogenital warts. Sex Transm Dis 38:216–218
Werner RN, Westfechtel L, Dressler C, Nast A (2017) Self-administered interventions for anogenital warts in immunocompetent patients: a systematic review and meta-analysis. Sex Transm Infect 93:155–161
Syed T, Lundin S, Ahmad M (1994) Topical 0.3% and 0.5% podophyllotoxin cream for self-treatment of molluscum contagiosum in males. Dermatology 189:65–68
Von Krogh G, Hellberg D (1992) Self-treatment using a 0.5% podophyllotoxin cream of external genital condylomata acuminata in women. A placebo-controlled, double-blind study. Sex Transm Dis 19:170–174
Gilson R, Nugent D, Werner R, Ballesteros J, Ross J (2020) 2019 IUSTI-Europe guideline for the management of anogenital warts. J Eur Acad Dermatol Venereol 34:1644–1653
Beutner KR, Tyring SK, Trofatter KF, Douglas JM, Spruance S, Owens ML, Fox TL, Hougham AJ, Schmitt KA (1998) Imiquimod, a patient-applied immune-response modifier for treatment of external genital warts. Antimicrob Agents Chemother 42:789–794
Edwards L, Ferenczy A, Eron L, Baker D, Owens ML, Fox TL, Hougham AJ, Schmitt KA, Group HS (1998) Self-administered topical 5% imiquimod cream for external anogenital warts. Arch Dermatol 134:25–30
Beutner KR, Spruance SL, Hougham AJ, Fox TL, Owens ML, Douglas JM Jr (1998) Treatment of genital warts with an immune-response modifier (imiquimod). J Am Acad Dermatol 38:230–239
Baker DA, Ferris DG, Martens MG, Fife KH, Tyring SK, Edwards L, Nelson A, Ault K, Trofatter KF, Liu T (2011) Imiquimod 3.75% cream applied daily to treat anogenital warts: combined results from women in two randomized, placebo-controlled studies. Infect Dis Obstet Gynecol 2011
Harrison C, Jenski L, Voychehovski T, Bernstein D (1988) Modification of immunological responses and clinical disease during topical R-837 treatment of genital HSV-2 infection. Antiviral Res 10:209–223
Sidky YA, Borden EC, Weeks CE, Reiter MJ, Hatcher JF, Bryan GT (1992) Inhibition of murine tumor growth by an interferon-inducing imidazoquinolinamine. Can Res 52:3528–3533
Bernstein DI, Miller RL, Harrison CJ (1993) Effects of therapy with an immunomodulator (imiquimod, R-837) alone and with acyclovir on genital HSV-2 infection in guinea-pigs when begun after lesion development. Antiviral Res 20:45–55
Tyring SK, Arany I, Stanley MA, Tomai MA, Miller RL, Smith MH, McDermott DJ, Slade HB (1998) A randomized, controlled, molecular study of condylomata acuminata clearance during treatment with imiquimod. J Infect Dis 178:551–555
Grillo‐Ardila CF, Angel‐Müller E, Salazar‐Díaz LC, Gaitan HG, Ruiz‐Parra AI, Lethaby A (2014) Imiquimod for anogenital warts in non‐immunocompromised adults. Cochrane Database Syst Rev
Schöfer H, Van Ophoven A, Henke U, Lenz T, Eul A (2006) Randomized, comparative trial on the sustained efficacy of topical imiquimod 5% cream versus conventional ablative methods in external anogenital warts. Eur J Dermatol 16:642–648
Arican O, Guneri F, Bilgic K, Karaoglu A (2004) Topical imiquimod 5% cream in external anogenital warts: a randomized, double-blind, placebo-controlled study. J Dermatol 31:627–631
Einarson A, Costei A, Kalra S, Rouleau M, Koren G (2005) The use of topical 5% imiquimod during pregnancy: a case series. Reprod Toxicol (Elmsford, NY) 21:1–2
Audisio T, CRESPO ROCA F, Piatti C (2008) Topical imiquimod therapy for external anogenital warts in pregnant women. Int J Gynaecol Obstet 100:275–276
Ciavattini A, Tsiroglou D, Vichi M, Di Giuseppe J, Cecchi S, Tranquilli AL (2012) Topical Imiquimod 5% cream therapy for external anogenital warts in pregnant women: Report of four cases and review of the literature. J Matern Fetal Neonatal Med 25:873–876
Lin J-K, Liang Y-C (2000) Cancer chemoprevention by tea polyphenols. Proc Natl Sci Counc Repub China B 24:1–13
Kuo C-L, Chen T-S, Liou S-Y, Hsieh C-C (2014) Immunomodulatory effects of EGCG fraction of green tea extract in innate and adaptive immunity via T regulatory cells in murine model. Immunopharmacol Immunotoxicol 36:364–370
Stockfleth E, Meyer T (2014) Sinecatechins (Polyphenon E) ointment for treatment of external genital warts and possible future indications. Expert Opin Biol Ther 14:1033–1043
Tzellos T, Sardeli C, Lallas A, Papazisis G, Chourdakis M, Kouvelas D (2011) Efficacy, safety and tolerability of green tea catechins in the treatment of external anogenital warts: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol 25:345–353
Stockfleth E, Meyer T (2012) The use of sinecatechins (polyphenon E) ointment for treatment of external genital warts. Expert Opin Biol Ther 12:783–793
Tatti S, Swinehart JM, Thielert C, Tawfik H, Mescheder A, Beutner KR (2008) Sinecatechins, a defined green tea extract, in the treatment of external anogenital warts: a randomized controlled trial. Obstet Gynecol 111:1371–1379
Godley M, Bradbeer C, Gellan M, Thin R (1987) Cryotherapy compared with trichloroacetic acid in treating genital warts. Sex Transm Infect 63:390–392
Sherrard J, Riddell L (2007) Comparison of the effectiveness of commonly used clinic-based treatments for external genital warts. Int J STD AIDS 18:365–368
Lotfabadi P, Maleki F, Gholami A, Yazdanpanah MJ (2015) Liquid nitrogen cryotherapy versus 70% trichloroacetic acid in the treatment of anogenital warts: a randomized controlled trial. Iran J Dermatol 18:151–155
Krebs H-B (1990) Treatment of extensive vulvar condylomata acuminata with topical 5-fluorouracil. South Med J 83:761–764
Haye K (1974) Treatment of condyloma acuminata with 5 per cent. 5-fluorouracil (5-FU) cream. Br J Vener Dis 50:466
Westfechtel L, Werner RN, Dressler C, Gaskins M, Nast A (2018) Adjuvant treatment of anogenital warts with systemic interferon: a systematic review and meta-analysis. Sex Transm Infect 94:21–29
Yang J, Pu Y-g, Zeng Z-m, Yu Z-j, Huang N, Deng Q-w (2009) Interferon for the treatment of genital warts: a systematic review. BMC Infect Dis 9:1–9
Eron LJ, Judson F, Tucker S, Prawer S, Mills J, Murphy K, Hickey M, Rogers M, Flannigan S, Hien N (1986) Interferon therapy for condylomata acuminata. N Engl J Med 315:1059–1064
Syed TA, Ahmadpour OA (1998) Human leukocyte derived interferon-alpha in a hydrophilic gel for the treatment of intravaginal warts in women: a placebo-controlled, double-blind study. Int J STD AIDS 9:769–772
Hu Z, Liu L, Zhang W, Liu H, Li J, Jiang L, Zeng K (2018) Dynamics of HPV viral loads reflect the treatment effect of photodynamic therapy in genital warts. Photodiagn Photodyn Ther 21:86–90
Hu Z, Li J, Liu H, Liu L, Jiang L, Zeng K (2018) Treatment of latent or subclinical Genital HPV Infection with 5-aminolevulinic acid-based photodynamic therapy. Photodiagn Photodyn Ther 23:362–364
Martin‐Hirsch PP, Paraskevaidis E, Bryant A, Dickinson HO, Keep SL (2010) Surgery for cervical intraepithelial neoplasia. Cochrane Database Syst Rev
Kyrgiou M, Athanasiou A, Paraskevaidi M, Mitra A, Kalliala I, Martin-Hirsch P, Arbyn M, Bennett P, Paraskevaidis E (2016) Adverse obstetric outcomes after local treatment for cervical preinvasive and early invasive disease according to cone depth: systematic review and meta-analysis. bmj 354
Jensen SL (1985) Comparison of podophyllin application with simple surgical excision in clearance and recurrence of perianal condylomata acuminata. The Lancet 326:1146–1148
Reichman RC, Oakes D, Bonnez W, Greisberger C, Tyring S, Miller L, Whitley R, Carveth H, Weidner M, Krueger G (1988) Treatment of Condyloma Acuminatum with Three Different Interferons Administered lntralesionally: A Double-Blind, Placebo-Controlled Trial. Ann Intern Med 108:675–679
Khawaja H (1989) Podophyllin versus scissor excision in the treatment of perianal condylomata acuminata: a prospective study. J Br Surg 76:1067–1068
Stone K, Becker T, Hadgu A, Kraus S (1990) Treatment of external genital warts: a randomised clinical trial comparing podophyllin, cryotherapy, and electrodesiccation. Sex Transm Infect 66:16–19
Scambia G, Baiocchi G, Perrone L, Pintus C, Mancuso S (1989) Randomized clinical trial comparing systemic interferon with diathermocoagulation in primary multiple and widespread anogenital condyloma. Obstet Gynecol 74:393–397
Azizjalali M, Ghaffarpour G, Mousavifard B (2012) CO2 Laser therapy versus cryotherapy in treatment of genital warts; a Randomized Controlled Trial (RCT). Iran J Microbiol 4:187
Chen K, Chang B, Ju M, Zhang X, Gu H (2007) Comparative study of photodynamic therapy vs. CO2 laser vaporization in treatment of condylomata acuminata, a randomized clinical trial. Br J Dermatol 156:516–520
Liang J, Lu XN, Tang H, Zhang Z, Fan J, Xu JH (2009) Evaluation of photodynamic therapy using topical aminolevulinic acid hydrochloride in the treatment of condylomata acuminata: a comparative, randomized clinical trial. Photodermatol Photoimmunol Photomed 25:293–297
SZEIMIES RM, Schleyer V, Moll I, Stocker M, Landthaler M, Karrer S (2009) Adjuvant Photodynamic Therapy Does Not Prevent Recurrence of Condylomata Acuminata After Carbon Dioxide Laser Ablation—A Phase III, Prospective, Randomized, Bicentric, Double-Blind Study. Dermatol Surg 35:757–764
Bogani G, Ditto A, Martinelli F, Mosca L, Chiappa V, Rossetti D, Leone Roberti Maggiore U, Sabatucci I, Lorusso D, Raspagliesi F (2018) LASER treatment for women with high-grade vaginal intraepithelial neoplasia: A propensity-matched analysis on the efficacy of ablative versus excisional procedures. Lasers Surg Med 50:933–939
Spitzer M (2007) Screening and management of women and girls with human papillomavirus infection. Gynecol Oncol 107:S14–S18
Kyrgiou M, Valasoulis G, Founta C, Koliopoulos G, Karakitsos P, Nasioutziki M, Navrozoglou I, Dalkalitsis N, Paraskevaidis E (2010) Clinical management of HPV-related disease of the lower genital tract. Ann N Y Acad Sci 1205:57–68
Mitra A, Tzafetas M, Lyons D, Fotopoulou C, Paraskevaidis E, Kyrgiou M (2016) Cervical intraepithelial neoplasia: screening and management. Br J Hosp Med 77:C118–C123
Marchetti C, Fagotti A, Tombolini V, Scambia G, De Felice F (2020) Survival and toxicity in neoadjuvant chemotherapy plus surgery versus definitive chemoradiotherapy for cervical cancer: a systematic review and meta-analysis. Cancer Treat Rev 83: 101945
Marth C, Landoni F, Mahner S, McCormack M, Gonzalez-Martin A, Colombo N (2018) Cervical cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 29: iv262. https://doi.org/10.1093/annonc/mdy160
Gadducci A, Cosio S (2020) Neoadjuvant chemotherapy in locally advanced cervical cancer: review of the literature and perspectives of clinical research. Anticancer Res 40:4819–4828
Barra F, Lorusso D, Leone Roberti Maggiore U, Ditto A, Bogani G, Raspagliesi F, Ferrero S (2017) Investigational drugs for the treatment of cervical cancer. Expert Opin Investig Drugs 26:389–402
Movva S, Rodriguez L, Arias‐Pulido H, Verschraegen C (2009) Novel chemotherapy approaches for cervical cancer. Cancer: Interdisciplinary Int J Am Cancer Soc 115: 3166–3180
Melamed A, Margul DJ, Chen L, Keating NL, Del Carmen MG, Yang J, Seagle B-LL, Alexander A, Barber EL, Rice LW (2018) Survival after minimally invasive radical hysterectomy for early-stage cervical cancer. N Engl J Med 379:1905–1914
Marchetti C, De Felice F, Di Pinto A, Romito A, Musella A, Palaia I, Monti M, Tombolin V, Muzii L, Panici PB (2018) Survival nomograms after curative neoadjuvant chemotherapy and radical surgery for stage IB2-IIIB cervical cancer. Cancer Res Treat: Official J Korean Cancer Assoc 50:768
Trimbos J, Franchi M, Zanaboni F, Velden J, Vergote I (2004) ‘State of the art’of radical hysterectomy; current practice in European oncology centres. Eur J Cancer 40:375–378
Uppal S, Gehrig PA, Peng K, Bixel KL, Matsuo K, Vetter MH, Davidson BA, Cisa MP, Lees BF, Brunette LL (2020) Recurrence rates in patients with cervical cancer treated with abdominal versus minimally invasive radical hysterectomy: a multi-institutional retrospective review study. J Clin Oncol 38:1030–1040
De Felice F, Marchetti C, Palaia I, Ostuni R, Muzii L, Tombolini V, Panici PB (2018) Immune check-point in cervical cancer. Crit Rev Oncol Hematol 129:40–43
Liu C, Lu J, Tian H, Du W, Zhao L, Feng J, Yuan D, Li Z (2017) Increased expression of PD-L1 by the human papillomavirus 16 E7 oncoprotein inhibits anticancer immunity. Mol Med Rep 15:1063–1070
Keay S, Teng N, Eisenberg M, Story B, Sellers PW, Merigan TC (1988) Topical interferon for treating condyloma acuminata in women. J Infect Dis 158:934–939
Clark KT, Trimble CL (2020) Current status of therapeutic HPV vaccines. Gynecol Oncol 156:503–510
Farmer E, Cheng MA, Hung C-F, Wu T-C (2021) Vaccination Strategies for the Control and Treatment of HPV Infection and HPV-Associated Cancer. Virus Hum Cancer 157–195
Hancock G, Hellner K, Dorrell L (2018) Therapeutic HPV vaccines. Best Pract Res Clin Obstet Gynaecol 47:59–72
Liao S-j, Deng D-r, Zeng D, Zhang L, Hu X-j, Zhang W-n, Li L, Jiang X-f, Wang C-y, Zhou J-f (2013) HPV16 E5 peptide vaccine in treatment of cervical cancer in vitro and in vivo. J Huazhong Univ Sci Technol [Medical Sciences] 33:735–742
Ma B, Maraj B, Tran NP, Knoff J, Chen A, Alvarez RD, Hung C-F, Wu T-C (2012) Emerging human papillomavirus vaccines. Expert Opin Emerg Drugs 17:469–492
Yang A, Jeang J, Cheng K, Cheng T, Yang B, Wu T-C, Hung C-F (2016) Current state in the development of candidate therapeutic HPV vaccines. Expert Rev Vaccines 15:989–1007
Mahdavi A, Monk BJ (2005) Vaccines against human papillomavirus and cervical cancer: promises and challenges. Oncologist 10:528–538
Bergot A-S, Kassianos A, Frazer IH, Mittal D (2011) New approaches to immunotherapy for HPV associated cancers. Cancers 3:3461–3495
Adachi K, Kawana K, Yokoyama T, Fujii T, Tomio A, Miura S, Tomio K, Kojima S, Oda K, Sewaki T (2010) Oral immunization with a Lactobacillus casei vaccine expressing human papillomavirus (HPV) type 16 E7 is an effective strategy to induce mucosal cytotoxic lymphocytes against HPV16 E7. Vaccine 28:2810–2817
Bermúdez-Humarán LG, Cortes-Perez NG, Le Loir Y, Alcocer-González JM, Tamez-Guerra RS, de Oca-Luna RM, Langella P (2004) An inducible surface presentation system improves cellular immunity against human papillomavirus type 16 E7 antigen in mice after nasal administration with recombinant lactococci. J Med Microbiol 53:427–433
Cortes-Perez NG, Azevedo V, Alcocer-González JM, Rodriguez-Padilla C, Tamez-Guerra RS, Corthier G, Gruss A, Langella P, Bermúdez-Humarán LG (2005) Cell-surface display of E7 antigen from human papillomavirus type-16 in Lactococcus lactis and in Lactobacillus plantarum using a new cell-wall anchor from lactobacilli. J Drug Target 13:89–98
Sewell DA, Pan ZK, Paterson Y (2008) Listeria-based HPV-16 E7 vaccines limit autochthonous tumor growth in a transgenic mouse model for HPV-16 transformed tumors. Vaccine 26:5315–5320
Schnupf P, Portnoy DA (2007) Listeriolysin O: a phagosome-specific lysin. Microbes Infect 9:1176–1187
Peters C, Paterson Y (2003) Enhancing the immunogenicity of bioengineered Listeria monocytogenes by passaging through live animal hosts. Vaccine 21:1187–1194
Chen Z, Ozbun L, Chong N, Wallecha A, Berzofsky JA, Khleif SN (2014) Episomal Expression of Truncated Listeriolysin O in LmddA-LLO–E7 Vaccine Enhances Antitumor Efficacy by Preferentially Inducing Expansions of CD4+ FoxP3− and CD8+ T Cells. Cancer Immunol Res 2:911–922
Maciag PC, Radulovic S, Rothman J (2009) The first clinical use of a live-attenuated Listeria monocytogenes vaccine: a Phase I safety study of Lm-LLO-E7 in patients with advanced carcinoma of the cervix. Vaccine 27:3975–3983
Cassetti MC, McElhiney SP, Shahabi V, Pullen JK, Le Poole IC, Eiben GL, Smith LR, Kast WM (2004) Antitumor efficacy of Venezuelan equine encephalitis virus replicon particles encoding mutated HPV16 E6 and E7 genes. Vaccine 22:520–527
Gomez-Gutierrez JG, Elpek KG, de Oca-Luna RM, Shirwan H, Zhou HS, McMasters KM (2007) Vaccination with an adenoviral vector expressing calreticulin-human papillomavirus 16 E7 fusion protein eradicates E7 expressing established tumors in mice. Cancer Immunol Immunother 56:997–1007
Walczak M, Regts J, Van Oosterhout A, Boon L, Wilschut J, Nijman HW, Daemen T (2011) Role of regulatory T-cells in immunization strategies involving a recombinant alphavirus vector system. Antivir Ther 16:207
Grasso F, Negri DR, Mochi S, Rossi A, Cesolini A, Giovannelli A, Chiantore MV, Leone P, Giorgi C, Cara A (2013) Successful therapeutic vaccination with integrase defective lentiviral vector expressing nononcogenic human papillomavirus E7 protein. Int J Cancer 132:335–344
Borysiewicz L, Fiander A, Nimako M, Man S, Wilkinson GWG, Westmoreland D, Evans A, Adams M, Stacey SN, Boursnell M (1996) A recombinant vaccinia virus encoding human papillomavirus types 16 and 18, E6 and E7 proteins as immunotherapy for cervical cancer. The Lancet 347:1523–1527
Lamikanra A, Pan Z-K, Isaacs SN, Wu T-C, Paterson Y (2001) Regression of established human papillomavirus type 16 (HPV-16) immortalized tumors in vivo by vaccinia viruses expressing different forms of HPV-16 E7 correlates with enhanced CD8+ T-cell responses that home to the tumor site. J Virol 75:9654–9664
Kaufmann AM, Stern PL, Rankin EM, Sommer H, Nuessler V, Schneider A, Adams M, Onon TS, Bauknecht T, Wagner U (2002) Safety and immunogenicity of TA-HPV, a recombinant vaccinia virus expressing modified human papillomavirus (HPV)-16 and HPV-18 E6 and E7 genes, in women with progressive cervical cancer. Clin Cancer Res 8:3676–3685
Chang C-L, Ma B, Pang X, Wu TC, Hung C-F (2009) Treatment with cyclooxygenase-2 inhibitors enables repeated administration of vaccinia virus for control of ovarian cancer. Mol Ther 17:1365–1372
Komdeur FL, Singh A, van de Wall S, Meulenberg JJ, Boerma A, Hoogeboom BN, Paijens ST, Oyarce C, de Bruyn M, Schuuring E (2021) First-in-human phase I clinical trial of an SFV-based RNA replicon cancer vaccine against HPV-induced cancers. Mol Ther 29:611–625
Harper DM, Nieminen P, Donders G, Einstein MH, Garcia F, Huh WK, Stoler MH, Glavini K, Attley G, Limacher J-M (2019) The efficacy and safety of Tipapkinogen Sovacivec therapeutic HPV vaccine in cervical intraepithelial neoplasia grades 2 and 3: Randomized controlled phase II trial with 2.5 years of follow-up. Gynecol Oncol 153:521–529
Lin K, Doolan K, Hung C-F, Wu T (2010) Perspectives for preventive and therapeutic HPV vaccines. J Formos Med Assoc 109:4–24
Zwaveling S, Mota SCF, Nouta J, Johnson M, Lipford GB, Offringa R, van der Burg SH, Melief CJ (2002) Established human papillomavirus type 16-expressing tumors are effectively eradicated following vaccination with long peptides. J Immunol 169:350–358
Su J-H, Wu A, Scotney E, Ma B, Monie A, Hung C-F, Wu T-C (2010) Immunotherapy for cervical cancer. BioDrugs 24:109–129
Cui Z, Huang L (2005) Liposome-polycation-DNA (LPD) particle as a carrier and adjuvant for protein-based vaccines: therapeutic effect against cervical cancer. Cancer Immunol Immunother 54:1180–1190
Stewart TJ, Drane D, Malliaros J, Elmer H, Malcolm KM, Cox JC, Edwards SJ, Frazer IH, Fernando GJ (2004) ISCOMATRIX™ adjuvant: an adjuvant suitable for use in anticancer vaccines. Vaccine 22:3738–3743
De Jong A, O’Neill T, Khan A, Kwappenberg K, Chisholm S, Whittle N, Dobson J, Jack L, Roberts JSC, Offringa R (2002) Enhancement of human papillomavirus (HPV) type 16 E6 and E7-specific T-cell immunity in healthy volunteers through vaccination with TA-CIN, an HPV16 L2E7E6 fusion protein vaccine. Vaccine 20:3456–3464
Van Damme P, Bouillette-Marussig M, Hens A, De Coster I, Depuydt C, Goubier A, Van Tendeloo V, Cools N, Goossens H, Hercend T (2016) GTL001, A therapeutic vaccine for women infected with human papillomavirus 16 or 18 and normal cervical cytology: results of a phase I clinical trial. Clin Cancer Res 22:3238–3248
Lee S-J, Yang A, Wu T-C, Hung C-F (2016) Immunotherapy for human papillomavirus-associated disease and cervical cancer: review of clinical and translational research. J Gynecol Oncol 27
Vici P, Pizzuti L, Mariani L, Zampa G, Santini D, Di Lauro L, Gamucci T, Natoli C, Marchetti P, Barba M (2016) Targeting immune response with therapeutic vaccines in premalignant lesions and cervical cancer: hope or reality from clinical studies. Expert Rev Vaccines 15:1327–1336
Yang A, Farmer E, Wu T, Hung C-F (2016) Perspectives for therapeutic HPV vaccine development. J Biomed Sci 23:1–19
Barber GN (2011) Cytoplasmic DNA innate immune pathways. Immunol Rev 243:99–108
Fiander AN, Tristram AJ, Davidson EJ, Tomlinson AE, Man S, Baldwin P, Sterling JC, Kitchener HC (2006) Prime-boost vaccination strategy in women with high-grade, noncervical anogenital intraepithelial neoplasia: clinical results from a multicenter phase II trial. Int J Gynecol Cancer 16
Dupuis M, Denis-Mize K, Woo C, Goldbeck C, Selby MJ, Chen M, Otten GR, Ulmer JB, Donnelly JJ, Ott G (2000) Distribution of DNA vaccines determines their immunogenicity after intramuscular injection in mice. J Immunol 165:2850–2858
Stratford R, Douce G, Zhang-Barber L, Fairweather N, Eskola J, Dougan G (2000) Influence of codon usage on the immunogenicity of a DNA vaccine against tetanus. Vaccine 19:810–815
Sher Y-P, Lin S-I, Chai KM, Chen I-H, Liu S-J (2019) Endoplasmic reticulum-targeting sequence enhanced the cellular immunity of a tumor-associated antigen L6-based DNA vaccine. Am J Cancer Res 9:2028
Allen A, Wang C, Caproni LJ, Sugiyarto G, Harden E, Douglas LR, Duriez PJ, Karbowniczek K, Extance J, Rothwell PJ (2018) Linear doggybone DNA vaccine induces similar immunological responses to conventional plasmid DNA independently of immune recognition by TLR9 in a pre-clinical model. Cancer Immunol Immunother 67:627–638
Diebold SS, Kaisho T, Hemmi H, Akira S, e Sousa CR (2004) Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 303:1529–1531
Liu MA (2019) A comparison of plasmid DNA and mRNA as vaccine technologies. Vaccines 7:37
Pardi N, Hogan MJ, Porter FW, Weissman D (2018) mRNA vaccines—a new era in vaccinology. Nat Rev Drug Discovery 17:261
Santin AD, Bellone S, Roman JJ, Burnett A, Cannon MJ, Pecorelli S (2005) Therapeutic vaccines for cervical cancer: dendritic cell-based immunotherapy. Curr Pharm Des 11:3485–3500
Ma B, Xu Y, Hung C-F, Wu T-C (2010) HPV and therapeutic vaccines: where are we in 2010? Curr Cancer Ther Rev 6:81–103
Bolhassani A, Mohit E, Rafati SR (2009) Different spectra of therapeutic vaccine development against HPV infections. Hum Vaccin 5:671–689
Lau C-H, Suh Y (2017) Genome and epigenome editing in mechanistic studies of human aging and aging-related disease. Gerontology 63:103–117
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E (2012) A programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821
Zhen S, Hua L, Takahashi Y, Narita S, Liu Y-H, Li Y (2014) In vitro and in vivo growth suppression of human papillomavirus 16-positive cervical cancer cells by CRISPR/Cas9. Biochem Biophys Res Commun 450:1422–1426
Jubair L, Fallaha S, McMillan NA (2019) Systemic delivery of CRISPR/Cas9 targeting HPV oncogenes is effective at eliminating established tumors. Mol Ther 27:2091–2099
Hu Z, Yu L, Zhu D, Ding W, Wang X, Zhang C, Wang L, Jiang X, Shen H, He D (2014) Disruption of HPV16-E7 by CRISPR/Cas system induces apoptosis and growth inhibition in HPV16 positive human cervical cancer cells. BioMed Res Int 2014
Cho SW, Kim S, Kim Y, Kweon J, Kim HS, Bae S, Kim J-S (2014) Analysis of off-target effects of CRISPR/Cas-derived RNA-guided endonucleases and nickases. Genome Res 24:132–141
Naito Y, Hino K, Bono H, Ui-Tei K (2015) CRISPRdirect: software for designing CRISPR/Cas guide RNA with reduced off-target sites. Bioinformatics 31:1120–1123
Brown KR, Mair B, Soste M, Moffat J (2019) CRISPR screens are feasible in TP 53 wild‐type cells. Mol Syst Biol 15:e8679
Wang D, Zhang C, Wang B, Li B, Wang Q, Liu D, Wang H, Zhou Y, Shi L, Lan F (2019) Optimized CRISPR guide RNA design for two high-fidelity Cas9 variants by deep learning. Nat Commun 10:1–14
Frangoul H, Bobruff Y, Cappellini MD, Corbacioglu S, Fernandez C, De La Fuente J, Grupp S, Handgretinger R, Ho TW, Imren S (2020) Safety and efficacy of CTX001 in patients with transfusion-dependent β-thalassemia or sickle cell disease: Early results from the CLIMB THAL-111 and CLIMB SCD-121 studies of autologous CRISPR-CAS9-modified CD34+ hematopoietic stem and progenitor cells. Proc 62nd Am Soc Hematol Ann Meeting, Online, pp. 5–8
Philippidis A (2020) One Small Dose, One Giant Leap for CRISPR Gene Editing. Hum Gene Ther 31:402–404
Barnard AM, Fairhead HI (2021) A commentary on the development of engineered phage as therapeutics. Drug Discovery Today
Gillmore JD, Gane E, Taubel J, Kao J, Fontana M, Maitland ML, Seitzer J, O’Connell D, Walsh KR, Wood K (2021) CRISPR-Cas9 in vivo gene editing for transthyretin amyloidosis. New England J Med
Lu Y, Xue J, Deng T, Zhou X, Yu K, Deng L, Huang M, Yi X, Liang M, Wang Y (2020) Safety and feasibility of CRISPR-edited T cells in patients with refractory non-small-cell lung cancer. Nat Med 26:732–740
Xu Y, Jiang J, Wang Y, Wang W, Li H, Lai W, Zhou Z, Zhu W, Xiang Z, Wang Z (2021) Engineered T cell therapy for gynecologic malignancies: challenges and opportunities. Front Immunol 3035
Acknowledgements
Not applicable.
Funding
The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
NK wrote the manuscript. NK, AB and RN reviewed the current state of the manuscript and prepared the tables and figure. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khairkhah, N., Bolhassani, A. & Najafipour, R. Current and future direction in treatment of HPV-related cervical disease. J Mol Med 100, 829–845 (2022). https://doi.org/10.1007/s00109-022-02199-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-022-02199-y