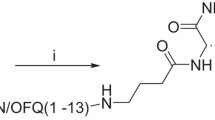
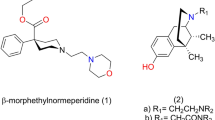
Abstract
A series of N-substituted analogs based upon the spiropiperidine core of the lead compound Spiroxatrine was synthesized. In particular, the new compounds were obtained by replacing the benzodioxane moiety of the Spiroxatrine with several 2-substituted 1,3-dioxolanes. Thus the designed derivatives were synthesized and evaluated as possible NOP receptor ligands. As a conclusion of these studies, the new triazaspirodecanone derivatives showed unique and significant SAR as NOP receptor agonists. In particular, the present study demonstrated that 1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one portion together with appropriate 1,3-dioxolane substituents could lead to a new promising class of NOP receptor ligands.





Similar content being viewed by others
References
Ambo A, Hamazaki N, Yamada Y, Nakata E, Sasaki Y (2001) Structure–activity studies on nociceptin analogues: ORL1 receptor binding and biological activity of cyclic disulfide-containing analogues of nociceptin peptides. J Med Chem 44:4015–4018
Andoh T, Yaqeta Y, Takeshima H, Kuraishi YI (2004) Intradermal nociceptin elicits itch-associated responses through leukotriene B-4 in mice. J Invest Dermatol 123:196–201
Brasili L, Sorbi C, Franchini S, Manicardi M, Angeli P, Marucci G, Leonardi A, Poggesi E (2003) 1,3-dioxolane-based ligands as a novel class of α 1-adrenoceptor antagonists. J Med Chem 46:1504–1511
Calò G, Guerrini R, Bigoni R (1998) Structure–activity study of the nociceptin(1–13)-NH2 N-terminal tetrapeptide and discovery of a nociceptin receptor antagonist. J Med Chem 41:3360–3366
Camarda V, Fischetti C, Anzellotti N, Molinari P, Ambrosio C, Kostenis E, Regoli D, Trapella C, Guerrini R, Salvadori S, Calò G (2009) Pharmacological profile of NOP receptors coupled with calcium signaling via the chimeric protein G alpha(qi5). Naunyn Schmiedebergs Arch Pharmacol 379:599–610
Chen Y, Fan Y, Liu J, Mestek A, Tian M, Kozak CA, Yu L (1994) Molecular-cloning, tissue distribution and chromosomal localization of novel member of the opioid receptor gene family. FEBS Lett 347:279–283
Fischetti C, Camarda V, Rizzi A, Pelà M, Trapella C, Guerrini R, McDonald J, Lambert DG, Salvadori S, Regoli D, Calò G (2009) Pharmacological characterization of the nociceptin/orphanin FQ receptor non peptide antagonist Compound 24. Eur J Pharmacol 614:50–57
Granata F, Potenza RL, Fiori A, Strom R, Caronti B, Molinari P, Donsante S, Citro G, Iacovelli L, de Blasi A (2003) Expression of OP4 (ORL1, NOP1) receptors in vascular endothelium. Eur J Pharmacol 482:1–3
Guerrini R, Calò G, Bigoni R, Rizzi D, Rizzi A, Zucchini M, Varani K, Hashiba E, Lambert DG, Toth G (2001) Structure-activity studies of the Phe4 residue of nociceptin(1–13)-NH2: identification of highly potent agonists of the nociceptin/orphanin FQ receptor. J Med Chem 44:3956–3964
Jacoby E, Bouhelal R, Gerspacher M, Seuwen K (2006) The 7TM G-protein-coupled receptor target family. ChemMedChem 1:761–782
Kawamoto H, Ozaki S, Itoh Y, Miyaji M, Arai S, Nakashima H, Kato T, Ohta H, Iwasawa Y (1999) Discovery of the first potent and selective small molecule opioid receptor-like (ORL1) antagonist: 1-[(3R,4R)-1-cyclooctylmethyl-3- hydroxymethyl-4-piperidyl]-3-ethyl- 1,3-dihydro-2H-benzimidazol-2-one (J-113397). J Med Chem 42:5061–5063
Lambert DG (2008) The nociceptin/orphanin FQ receptor: a target with broad therapeutic potential. Nat Rev Drug Discov 7:694–710
Meunier JC, Mollereau C, Toll L, Suaudeau C, Moisand C, Alvineria P, Butour JL, Guillemot JC, Ferrara P, Monsarrat B, Mazargull H, Vassart G, Parmentier M, Costantin J (1995) Isolation and structure of the endogenous agonist of opioid receptor-like ORL1 receptor. Nature 377:532–535
Mollereau C, Parmentier M, Mailleux P, Butour JL, Moisand C, Chalon P, Caput D, Vassart G, Meunier C (1994) ORL1, a novel member of the opioid receptor family: cloning, functional expression and localization. FEBS Lett 341:33–38
Mollereau C, Mouledous L (2000) Tissue distribution of the opioid receptor-like (ORL1) receptor. Peptides 21:907–917
Mustazza C, Bastanzio G (2011) Development of nociceptin receptor (NOP) agonists and antagonists. Med Res Rev 31:605–648
Okada K, Sujaku T, Chuman Y, Nakashima R, Nose T, Costa T, Yamada Y, Yokoyama M, Nagahisa A, Shimohigashi Y (2000) Highly potent nociceptin analog containing the Arg-Lys triple repeat. Biochem Biophys Res Commun 278:493–498
Reinscheid RK, Nothacker HP, Bourson A, Ardati A, Henningsen RA, Bunzow JR, Gandy K, Langen H, Monsma FJ, Civelli O (1995) Orphanin FQ: a neuropeptide that activates an opioid like G protein-coupled receptor. Science 270:792–794
Sorbi C, Franchini S, Tait A, Prandi A, Gallesi R, Angeli P, Marucci G, Pirona L, Poggesi E, Brasili L (2009) 1,3-dioxolane-based ligands as rigid analogues of naftopidil: structure-affinity/activity relationships at alpha(1) and 5-HT1A receptors. ChemMedChem 4:393–399
Thomsen C, Hohlweg R (2000) (8-naphthalen-1-ylmethyl-4-oxo-1-phenyl-1,3,8-triaza-spiro[4.5]-dec-3-yl)-acetic acid methyl ester (NNC 63-0532) is a novel potent nociceptin receptor agonist. Br J Pharmacol 131:903–908
Zaveri N (2003) Peptide and nonpeptide ligands for the nociceptin/orphanin FQ receptor ORL1: research tools and potential therapeutic agents. Life Sci 73:663–678
Zhang C, Miller W, Valenzano KJ, Kyle DJ (2002) Novel, potent ORL-1 receptor agonist peptides containing α-Helix-promoting conformational constraints. J Med Chem 45:5280–5286
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Corrado, S., Sorbi, C., Tait, A. et al. 1,4-Dioxolane-triazaspirodecanone derivatives as nociceptin/orphanin FQ receptor ligands. Med Chem Res 23, 4642–4656 (2014). https://doi.org/10.1007/s00044-014-1032-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1032-y