Abstract
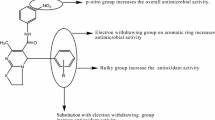
A series of 4,5-diaryl-2-[4-(t-amino)-2-butynyl]-2,4-dihydro-3H-1,2,4-triazol-3-ones were synthesized and characterized by infrared spectrum (IR), 1H-NMR spectra and elemental analyses. Investigation of their antimicrobial activity was performed. Antimicrobial activity profile of the title compounds were evaluated against Gram- positive bacteria, Gram-negative bacteria and fungi. The synthesized compounds displayed different degrees of antimicrobial activities as shown in Table 3. Compound 12 was the most active one. This may be attributed to 4,5-diphenylsubstituent on the triazoline-3-one ring and the nature of the amino groups at terminal acetylenic moiety.

Similar content being viewed by others
References
Asami T, Min YK, Nagata N, Yamagishi K, Takatsuto S, Fujioka S, Murofushi N, Yamaguchi I, Yoshida S (2000) Characterization of Brassinazole-type brassinosteroid biosynthesis inhibitor. Plant Physiol 123(1):93–100
Bhat AR, Bhat VG, Shenoy GG (2001) Synthesis and in vitro antimicrobial activity of new 1,2,4-triazoles. J Pharm Pharmacol 53:267–272
Chadha VK, Ranwa NS, Dadheech PK (1998) Synthesis and screening of biological activity of triazolo-thiadiazines and triazolo-thiadiazoles. J Phytol Res 11:201–202
Fujita M, Chiba K, Nakano J, Tominage Y, Matsumoto J (1998) Synthesis and structure-antibacterial activity relationships of 7-(3-amino-1-propynyl and 3-amino-1-propenyl)quinolones. Chem Pharm Bull 46(4):631–638
Gehlen H, Schade W (1964) Zur kenntnis der 2-amino-1.3.4-oxdiazole, XIV. 2-amino-5-arylamino-1.3.4-oxdiazole aus semicarbaziden und bromcyan. Justus Liebigs Ann Chem 675(1):176–179
Haasnoot J (2000) Mononuclear, oligonuclear and polynuclear metal coordination compounds with 1,2,4-trizole derivatives as ligands. Coord Chem Rev 200–202:131–185
Holla BS, Rao BS, Shridhara K, Akberali PM (2000) Studies on arylfuran derivatives—part XI. Synthesis, characterization and biological studies on some Mannich bases carrying 2,4-dichlorophenylfural moiety. Farmaco 55(5):338–344
Holla BS, Akberali PM, Shivananda MK (2001) Studies on nitrophenylfuran derivatives—part XII. Synthesis, characterization, antibacterial and antiviral activities of some nitrophenylfurfurylidene-1,2,4-triazolo[3,4-b]-1,3,4-thiadiazines. Farmaco 56(12):919–927
Holla BS, Rao BS, Sarojini BK, Akberali PM, Kumari NS (2006) Synthesis and studies on some new fluorine containing triazolothiazines as possible antibacterial, antifungal and anticancer agents. Eur J Med Chem 41(5):657–663
Hui XP, Zhang Y, Xu PF, Wang Q, Zhang Q, Zhang ZY (2005) Synthesis and antibacterial activities of novel 3-substituted-6-(4-chlorophenyl)-7-(1H–1,2,4-triazole-1-yl)-1′,2′,4′-triazolo[3,4-b]-1″,3″,4″-thiadiazines. Chin J Org Chem 25:700–704
Jin J, Zhang L, Zhang A, Lei X, Zhu J (2007) Synthesis and biological activity of some novel derivatives of 4-amino-(d-galactopentitol-1-yl)-5-mercapto-1,2,4-triazole. Molecules 12:1596–1605
Kim DK, Kim J, Park HJ (2004) Synthesis and biological evaluation of novel 2-pyridinyl-(1,2,3)triazoles and inhibitors of transforming growth factor beta1 type 1 receptor. Bioorg Med Chem Lett 14(10):2401–2405
Kritsanida M, Mouroutsou A, Marakos P, Pouli N, Papakonstantinou-Garoufalias S, Pannecouque C, Witvrouw M, De Clercq E (2002) Synthesis and antiviral activity evaluation of some new 6-substituted 3-(1-adamantyl)-1,2,4-triazolo[3,4–b][1,3,4]thiadiazoles. Farmaco 57(3):253–257
Liu J, Li L, Dai H, Liu Z, Fang J (2006) Synthesis and biological activity of new 1H–1,2,4-triazole derivatives containing ferrocenyl moity. Organomet Chem 691(12):2686–2690
Mali RK, Somani RR, Toraskar MP, Mali KK, Naik PP, Shirodkar PY (2009) Synthesis of some antifungal and anti-tubercular 1,2,4-triazole analogues. Int J Chem Tech Res 1:168–173
Modzelewska-Banachiewicz B, Banachiewicz J, Chodkowska A, Jagiello-Wojtowicz E, Mazur L (2004) Synthesis and biological activity of new derivatives of 3-(3,4-diaryl-1,2,4-triazole-5-yl) propenoic acid. Eur J Med Chem 39(10):873–877
Muhi-eldeen Z, Juma’a G, Al-kaissi E, Nouri L (2008) Antimicrobial activity of some new oxadiazole derivatives. Jord J Chem 3(30):233–243
Nadkarni BA, Kamat VR, Khadse BG (2001) Synthesis and anthelminthic activity of 3,6-disubstituted-7H-s-triazolo(3,4-b)(1,3,4,)thiadiazines. Arzneimitteforschung 51(7):569–573
National Committee for Clinical Laboratory Standards (NCCLS) (1998) Performance standards for antimicrobial susceptibility testing. standard (M100-128), 5th edn, Villanova
National Committee for Clinical Laboratory Standards (NCCLS) (2002) Reference method for broth dilution antifungal susceptibility testing for yeasts. approved standard (M27-A2), 2nd edn, Wayne
Sakata M, Shirakawa Y, Kamata N, Sakaguchi Y, Nishino H, Ouyang J, Kurosawa K (2001) Synthesis of triazole, oxazole and hetrocyclic ring-substituted 1,2-dioxanes. J Heterocyl Chem 37(2):269–274
Tatsumi Y, Yokoo M, Arika T, Yamaguchi H (2001) In vitro antifungal activity of KP-103, a novel triazole derivative, and its therapeutic efficacy against experimental plantar Tinea pedis and cutaneous candidiasis in Guinea pigs. Antimicrob Agents Chemother 45(5):1493–1499
Tian L, Sun Y, Li H, Zheng X, Cheng Y, Qian B, Liu X (2005) Synthesis and biological activity of triorganotin 2-phenyl-1,2,3-triazole-4-carboxylates. J Inorg Biochem 99(8):1646–1652
Ye X, Chen Z, Zhang A, Zhang L (2007) Synthesis and biological activity of some novel Schiff’s bases from 1,2,4-triazole. Molecules 12:1202–1209
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-kaissi, E.N., Al-Ghrary, N.F., Al-Kaisi, N.K. et al. Synthesis and antimicrobial evaluation of 4,5-diaryl-2-[4-(t-amino)-2-butynyl]-2,4-dihydro-3H-1,2,4-triazol-3-ones. Med Chem Res 21, 3390–3395 (2012). https://doi.org/10.1007/s00044-011-9840-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9840-9