Abstract
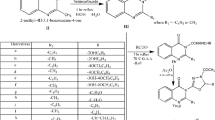
A variety of novel 3-(3-methoxyphenyl)-2-substituted amino-quinazolin-4(3H)-ones were synthesized by reacting the amino group of 2-hydrazino-3-(3-methoxyphenyl)-quinazolin-4(3H)-one with a variety of aldehydes and ketones. The starting material 2-hydrazino-3-(3-methoxyphenyl)-quinazolin-4(3H)-one was synthesized from 3-methoxy aniline. The title compounds were investigated for analgesic, anti-inflammatory, and ulcerogenic behavior. Among these the compound 2-(1-methyl butylidene-hydrazino)-3-(3-methoxyphenyl)-3H-quinazolin-4-one (AS3) emerged as the most active compound for the analgesic activity, while the compound 2-(1-ethyl propylidene-hydrazino)-3-(3-methyoxyphenyl)-3H-quinazolin-4-one (AS2) showed most potent anti-inflammatory activity of the series and these compounds are moderately more potent when compared to the reference standard diclofenac sodium. Interestingly the test compounds showed only mild ulcerogenic potential when compared to acetylsalicylic acid.


Similar content being viewed by others
References
Abdel-Rahman AE, Bakhite EA, Al-Taifi EA (2003) Synthesis and antimicrobial testing of some new S-substituted-thiopyridines, thienopyridines, pyridothienopyrimidines and pyridothienotriazines. Pharmazie 58:372–377
Alagarsamy V, Solomon VR, Vanikavitha G, Paluchamy V, Chandran MR, Sujin AA, Thangathiruppathy A, Amuthalakshmi S, Revathi R (2002) Synthesis, analgesic, anti-inflammatory and antibacterial activities of some novel 2-phenyl-3-substituted quinazolin-4(3H) ones. Biol Pharm Bull 25:1432–1435
Alagarsamy V, Murugananthan G, Venkateshperumal R (2003a) Synthesis, analgesic, anti-inflammatory and antibacterial activities of some novel 2-methyl-3-substituted quinazolin-4-(3H)-ones. Biol Pharm Bull 26:1711–1714
Alagarsamy V, Muthukumar V, Pavalarani N, Vasanthanathan P, Revathi R (2003b) Synthesis, analgesic and anti-inflammatory activities of some novel 2, 3-disubstituted quinazolin-4(3H)-ones. Biol Pharm Bull 26:557–559
Alagarsamy V, Revathi R, Vijayakumar S, Ramseshu KV (2003c) Synthesis and pharmacological investigation of some novel 2, 3-disubstituted quinazolin-4(3H)-ones as analgesic and antiinflammatory agents. Pharmazie 58:233–236
Alagarsamy V, Rajesh R, Meena R, Vijaykumar S, Ramseshu KV, Duraianandakumar T (2004) Synthesis, analgesic, anti-inflammatory and antibacterial activities of some novel 2-methylthio-3-substituted quinazolin-4(3H)-ones. Biol Pharm Bull 27:652–656
Alagarsamy V, Thangathirupathy A, Mandal SC, Rajasekaran S, Vijayakumar S, Revathi R, Anburaj J, Arunkumar S, Rajesh S (2006) Pharmacological evaluation of 2-substituted (1, 3, 4) thiadiazolo quinazolines. Indian J Pharm Sci 68:108–111
Amour RE, Smith DL (1941) A method for determinating loss of pain sensation. J Pharm Exp Ther 72:74–78
Beuck M (1999) Nonsteroidal antiinflammatory drugs: a new generation of cyclooxygenase inhibitors. Angew Chem Int Ed 38:631–633
Chambhare RV, Khadse BG, Bobde AS, Bahekar RH (2003) Synthesis and preliminary evaluation of some N-[5-(2-furanyl)-2-methyl-4-oxo-4H-thieno[2, 3-d]pyrimidin-3-yl]-carboxamide and 3-substituted-5-(2-furanyl)-2-methyl-3H-thieno[2, 3-d]pyrimidin-4-ones as antimicrobial agents. Eur J Med Chem 38:89–100
Ganguly AK, Bhatnagar OP (1973) Effect of bilateral adrenalectomy on production of restraint ulcers in the stomach of albino rats. Can J Physiol Pharmacol 51:748–750
Goyal RK, Chakrabarti A, Sanyal AK (1985) The effect of biological variables on the anti-ulcerogenic effect of vegetable plantain banana. Planta Med 29:85–88
Kulkarni SK (1980) Heat and other physiological stress-induced analgesia: catecholamine mediated and naloxone reversible response. Life Sci 27:185–188
Santagati NA, Caruso A, Cutuli VMC, Caccamo F (1995) Synthesis and pharmacological evaluation of thieno[2, 3-d]pyrimidin-2, 4-dione and 5H-pyrimido [5, 4-b]indol-2, 4-dione derivatives. Il Farmaco 50:689–695
Shay M, Komarov SA, Fels D, Meranze D, Grunstein H, Siplet H (1945) A simple method for the uniform production of gastric ulceration in the rats. Gastroenterology 5:43–61
Van Ryn J (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol 231:232–235
Van Ryn J, Botting RM (1995) New insights into the mode of action of anti-inflammatory drugs. Inflamm Res 44:1–10
Van Ryn J, Trummlitz G, Pairet M (2000) COX-2 selectivity and inflammatory processes. Curr Med Chem 7:1145–1161
Winter CA, Risely EA, Nuss GW (1962) Carregeenin induced oedema in hind paw of the rat as assay for antiinflammatory drugs. Exp Biol Med 111:544–547
Acknowledgments
The authors thank the head, Department of Chemistry, Indian Institute of Technology, Chennai for the mass spectral data and the head, sophisticated analytical instrument facilities, Central Drug Research Institute, Lucknow for NMR spectral data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alagarsamy, V., Gopinath, M., Parthiban, P. et al. Synthesis and analgesic, anti-inflammatory activities of 3-(3-methoxyphenyl)-2-substituted amino-quinazolin-4(3H)-ones. Med Chem Res 20, 946–954 (2011). https://doi.org/10.1007/s00044-010-9417-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-010-9417-z