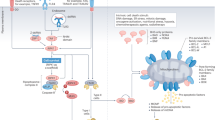
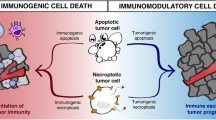
Abstract
The mode of tumor cell death has significant effects on anti-tumor immunity. Although, previously it was thought that cell death is an inert effect, different investigators have clearly shown that dying tumors can attract, activate and mature professional antigen presenting cells and dendritic cells. In addition, others and we have shown that the type of tumor cell death not only controls the presence or absence of specific tumor antigens, but also can result in immunological responses ranging from immunosuppression to anti-tumor immunity. More importantly, it is possible to enhance anti-tumor immunity both in vitro and in vivo by targeting specific molecular mechanisms such as oligopeptidases and the proteasome. These studies not only extend our knowledge on basic immunological questions and the induction of anti-tumor immunity, but also have implications for all types of cancer treatments, in which rapid tumor cell death is induced. This review is a comprehensive summary of cell death and particularly necrosis and the pivotal role it plays in anti-tumor immunity.

Similar content being viewed by others
Abbreviations
- DAMP:
-
Damage-associated molecular pattern molecules
- PAMP:
-
Pathogen-associated molecular pattern molecules
- IFN:
-
Interferon
- HMGB1:
-
High mobility group box 1 protein
- TNF:
-
Tumor necrosis factor
- TLR:
-
Toll-like receptor
- RIPK:
-
Receptor-interacting protein kinase
- APC:
-
Antigen presenting cell
- ICD:
-
Immunogenic cell death
- ER:
-
Endoplasmic reticulum
- ROS:
-
Reactive oxygen species
- CRT:
-
Calreticulin
- HSP:
-
Heat shock protein
- LPS:
-
Lipopolysaccharide
- RAGE:
-
Receptor for advanced glycosylation end product
- IL:
-
Interleukin
- RNP:
-
Ribonucleoproteins
- PRR:
-
Pattern recognition receptor
- DC:
-
Dendritic cell
- DRiP:
-
Defective ribosomal product
- OVA:
-
Ovalbumin
- LAP:
-
Leuzin aminopeptidase
- PSA:
-
Puromycine-sensitive aminopeptidase
- BH:
-
Bleomycin hydrolase
- TOP-1:
-
Thimet oligopeptidase 1
- TPPII:
-
Tripeptidyl peptidase II
- ERAP:
-
ER aminopeptidase
- ERAAP:
-
ER aminopeptidase associated with antigen processing
- DRibbles:
-
DRiPs-containing blebs
- DPP-3:
-
Dipeptidyl peptidase 3
- PEP:
-
Prolyl endopeptidase
- Ag:
-
Antigen
References
Ahrens S, Zelenay S et al (2012) F-Actin Is an Evolutionarily Conserved Damage-Associated Molecular Pattern Recognized by DNGR-1, a Receptor for Dead Cells. Immunity 36:635–645
Albert ML, Sauter B et al (1998) Dendritic cells acquire antigen from apoptotic cells and induce class I-restricted CTLs. Nature 392(6671):86–89
Andersson U, Wang H et al (2000) High mobility group 1 protein (HMG-1) stimulates proinflammatory cytokine synthesis in human monocytes. J Exp Med 192(4):565–570
Apetoh L, Ghiringhelli F et al (2007) Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 13(9):1050–1059
Ashkenazi A, Dixit VM (1999) Apoptosis control by death and decoy receptors. Curr Opin Cell Biol 11(2):255–260
Bartholomae WC, Rininsland FH et al (2004) T cell immunity induced by live, necrotic, and apoptotic tumor cells. J Immunol 173(2):1012–1022
Bartlett DL, Liu Z et al (2013) Oncolytic viruses as therapeutic cancer vaccines. Mol Cancer 12(1):103
Basta S, Stoessel R et al (2005) Cross-presentation of the long-lived lymphocytic choriomeningitis virus nucleoprotein does not require neosynthesis and is enhanced via heat shock proteins. J Immunol 175(2):796–805
Basu S, Binder RJ et al (2000) Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-kappa B pathway. Int Immunol 12(11):1539–1546
Beavis PA, Stagg J et al (2012) CD73: a potent suppressor of antitumor immune responses. Trends Immunol 33(5):231–237
Beninga J, Rock KL et al (1998) Interferon-gamma can stimulate post-proteasomal trimming of the N terminus of an antigenic peptide by inducing leucine aminopeptidase. J Biol Chem 273(30):18734–18742
Berti DA, Morano C et al (2009) Analysis of intracellular substrates and products of thimet oligopeptidase in human embryonic kidney 293 cells. J Biol Chem 284(21):14105–14116
Binder RJ, Srivastava PK (2005) Peptides chaperoned by heat-shock proteins are a necessary and sufficient source of antigen in the cross-priming of CD8+ T cells. Nat Immunol 6(6):593–599
Boldin MP, Varfolomeev EE et al (1995) A novel protein that interacts with the death domain of Fas/APO1 contains a sequence motif related to the death domain. J Biol Chem 270(14):7795–7798
Bustin M, Hopkins RB et al (1978) Immunological relatedness of high mobility group chromosomal proteins from calf thymus. J Biol Chem 253(5):1694–1699
Camargo AC, Gomes MD et al (1997) Structural features that make oligopeptides susceptible substrates for hydrolysis by recombinant thimet oligopeptidase. Biochem J 324(Pt 2):517–522
Camargo AC, Shapanka R et al (1973) Preparation, assay, and partial characterization of a neutral endopeptidase from rabbit brain. Biochemistry 12(9):1838–1844
Casares N, Pequignot MO et al (2005) Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J Exp Med 202(12):1691–1701
Cascio P, Hilton C et al (2001) 26S proteasomes and immunoproteasomes produce mainly N-extended versions of an antigenic peptide. EMBO J 20(10):2357–2366
Cavassani KA, Ishii M et al (2008) TLR3 is an endogenous sensor of tissue necrosis during acute inflammatory events. J Exp Med 205(11):2609–2621
Chen G, Ward MF et al (2004) Extracellular HMGB1 as a proinflammatory cytokine. J Interferon Cytokine Res 24(6):329–333
Chiba S, Baghdadi M et al (2012) Tumor-infiltrating DCs suppress nucleic acid-mediated innate immune responses through interactions between the receptor TIM-3 and the alarmin HMGB1. Nat Immunol 13(9):832–842
Cho YS, Challa S et al (2009) Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137(6):1112–1123
Deretic V, Levine B (2009) Autophagy, immunity, and microbial adaptations. Cell Host Microbe 5(6):527–549
Eigenbrod T, Park JH et al (2008) Cutting edge: critical role for mesothelial cells in necrosis-induced inflammation through the recognition of IL-1 alpha released from dying cells. J Immunol 181(12):8194–8198
Elliott MR, Chekeni FB et al (2009) Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461(7261):282–286
Gallucci S, Lolkema M et al (1999) Natural adjuvants: endogenous activators of dendritic cells. Nat Med 5(11):1249–1255
Galluzzi L, Vitale I et al (2012) Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 19(1):107–120
Gamrekelashvili J, Kapanadze T et al (2013) Peptidases released by necrotic cells control CD8+ T cell cross-priming. J Clin Invest 123(11):4755–4768
Gamrekelashvili J, Kruger C et al (2007) Necrotic tumor cell death in vivo impairs tumor-specific immune responses. J Immunol 178(3):1573–1580
Gamrekelashvili J, Ormandy LA et al (2012) Primary sterile necrotic cells fail to cross-prime CD8(+) T cells. Oncoimmunology 1(7):1017–1026
Gardai SJ, McPhillips KA et al (2005) Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell 123(2):321–334
Garg AD, Krysko DV et al (2012) A novel pathway combining calreticulin exposure and ATP secretion in immunogenic cancer cell death. EMBO J 31(5):1062–1079
Garg AD, Nowis D et al (2010) Immunogenic cell death, DAMPs and anticancer therapeutics: an emerging amalgamation. Biochim Biophys Acta 1805(1):53–71
Gebhardt C, Riehl A et al (2008) RAGE signaling sustains inflammation and promotes tumor development. J Exp Med 205(2):275–285
Gelebart P, Opas M et al (2005) Calreticulin, a Ca2+-binding chaperone of the endoplasmic reticulum. Int J Biochem Cell Biol 37(2):260–266
Ghiringhelli F, Apetoh L et al (2009) Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors. Nat Med 15(10):1170–1178
Gold LI, Eggleton P et al (2010) Calreticulin: non-endoplasmic reticulum functions in physiology and disease. FASEB J 24(3):665–683
Goldberg AL, Cascio P et al (2002) The importance of the proteasome and subsequent proteolytic steps in the generation of antigenic peptides. Mol Immunol 39(3–4):147–164
Goldszmid RS, Idoyaga J et al (2003) Dendritic cells charged with apoptotic tumor cells induce long-lived protective CD4+ and CD8+ T cell immunity against B16 melanoma. J Immunol 171(11):5940–5947
Holler N, Zaru R et al (2000) Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol 1(6):489–495
Inoue H, Tani K (2014) Multimodal immunogenic cancer cell death as a consequence of anticancer cytotoxic treatments. Cell Death Differ 21(1):39–49
Iwasaki A, Medzhitov R (2004) Toll-like receptor control of the adaptive immune responses. Nat Immunol 5(10):987–995
Iyer SS, Pulskens WP et al (2009) Necrotic cells trigger a sterile inflammatory response through the Nlrp3 inflammasome. Proc Natl Acad Sci USA 106(48):20388–20393
Janssen E, Tabeta K et al (2006) Efficient T cell activation via a toll-interleukin 1 receptor-independent pathway. Immunity 24(6):787–799
Joubert PE, Albert ML (2012) Antigen cross-priming of cell-associated proteins is enhanced by Macroautophagy within the antigen donor cell. Front Immunol 3:61
Kaczmarek A, Vandenabeele P et al (2013) Necroptosis: the release of damage-associated molecular patterns and its physiological relevance. Immunity 38(2):209–223
Kariko K, Ni H et al (2004) mRNA is an endogenous ligand for toll-like receptor 3. J Biol Chem 279(13):12542–12550
Kazama H, Ricci JE et al (2008) Induction of immunological tolerance by apoptotic cells requires caspase-dependent oxidation of high-mobility group box-1 protein. Immunity 29(1):21–32
Kerr JF, Wyllie AH et al (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26(4):239–257
Kessler JH, Khan S et al (2011) Antigen processing by nardilysin and thimet oligopeptidase generates cytotoxic T cell epitopes. Nat Immunol 12(1):45–53
Kisselev AF, Akopian TN et al (1999) The sizes of peptides generated from protein by mammalian 26 and 20 S proteasomes. Implications for understanding the degradative mechanism and antigen presentation. J Biol Chem 274(6):3363–3371
Kloetzel PM (2004) Generation of major histocompatibility complex class I antigens: functional interplay between proteasomes and TPPII. Nat Immunol 5(7):661–669
Kroemer G, Galluzzi L et al (2007) Mitochondrial membrane permeabilization in cell death. Physiol Rev 87(1):99–163
Kroemer G, Galluzzi L et al (2013) Immunogenic cell death in cancer therapy. Annu Rev Immunol 31:51–72
Kroemer G, Galluzzi L et al (2009) Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 16(1):3–11
Krysko DV, Garg AD et al (2012) Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer 12(12):860–875
Kunisawa J, Shastri N (2006) Hsp90alpha chaperones large C-terminally extended proteolytic intermediates in the MHC class I antigen processing pathway. Immunity 24(5):523–534
Lev A, Takeda K et al (2008) The exception that reinforces the rule: crosspriming by cytosolic peptides that escape degradation. Immunity 28(6):787–798
Li M, Davey GM et al (2001) Cell-associated ovalbumin is cross-presented much more efficiently than soluble ovalbumin in vivo. J Immunol 166(10):6099–6103
Li MO, Sarkisian MR et al (2003) Phosphatidylserine receptor is required for clearance of apoptotic cells. Science 302(5650):1560–1563
Li Y, Wang L-X et al (2009) Cross-presentation of tumor associated antigens through tumor-derived autophagosomes. Autophagy 5(4):576–577
Li Y, Wang LX et al (2011) Tumor-derived autophagosome vaccine: mechanism of cross-presentation and therapeutic efficacy. Clin Cancer Res 17(22):7047–7057
Lieberman J (2003) The ABCs of granule-mediated cytotoxicity: new weapons in the arsenal. Nat Rev Immunol 3(5):361–370
Liu K, Iyoda T et al (2002) Immune tolerance after delivery of dying cells to dendritic cells in situ. J Exp Med 196(8):1091–1097
Luthi AU, Cullen SP et al (2009) Suppression of interleukin-33 bioactivity through proteolysis by apoptotic caspases. Immunity 31(1):84–98
Matzinger P (1994) Tolerance, danger, and the extended family. Annu Rev Immunol 12:991–1045
Michaud M, Martins I et al (2011) Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 334(6062):1573–1577
Muller S, Ronfani L et al (2004) Regulated expression and subcellular localization of HMGB1, a chromatin protein with a cytokine function. J Intern Med 255(3):332–343
Neufeld TP, Baehrecke EH (2008) Eating on the fly: function and regulation of autophagy during cell growth, survival and death in drosophila. Autophagy 4(5):557–562
Norbury CC, Basta S et al (2004) CD8+ T cell cross-priming via transfer of proteasome substrates. Science 304(5675):1318–1321
Obeid M, Tesniere A et al (2007) Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med 13(1):54–61
Oerlemans MI, Liu J et al (2012) Inhibition of RIP1-dependent necrosis prevents adverse cardiac remodeling after myocardial ischemia-reperfusion in vivo. Basic Res Cardiol 107(4):270
Panaretakis T, Kepp O et al (2009) Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. EMBO J 28(5):578–590
Paz P, Brouwenstijn N et al (1999) Discrete proteolytic intermediates in the MHC class I antigen processing pathway and MHC I-dependent peptide trimming in the ER. Immunity 11(2):241–251
Pellegatti P, Raffaghello L et al (2008) Increased level of extracellular ATP at tumor sites: in vivo imaging with plasma membrane luciferase. PLoS One 3(7):e2599
Princiotta MF, Finzi D et al (2003) Quantitating protein synthesis, degradation, and endogenous antigen processing. Immunity 18(3):343–354
Rapaport E, Fontaine J (1989) Anticancer activities of adenine nucleotides in mice are mediated through expansion of erythrocyte ATP pools. Proc Natl Acad Sci USA 86(5):1662–1666
Reits E, Griekspoor A et al (2003) Peptide diffusion, protection, and degradation in nuclear and cytoplasmic compartments before antigen presentation by MHC class I. Immunity 18(1):97–108
Rock KL, York IA et al (2004) Post-proteasomal antigen processing for major histocompatibility complex class I presentation. Nat Immunol 5(7):670–677
Rovere-Querini P, Capobianco A et al (2004) HMGB1 is an endogenous immune adjuvant released by necrotic cells. EMBO Rep 5(8):825–830
Samuels MA (2007) The brain-heart connection. Circulation 116(1):77–84
Sancho D, Joffre OP et al (2009) Identification of a dendritic cell receptor that couples sensing of necrosis to immunity. Nature 458(7240):899–903
Saric T, Beninga J et al (2001) Major histocompatibility complex class I-presented antigenic peptides are degraded in cytosolic extracts primarily by thimet oligopeptidase. J Biol Chem 276(39):36474–36481
Saric T, Graef CI et al (2004) Pathway for degradation of peptides generated by proteasomes: a key role for thimet oligopeptidase and other metallopeptidases. J Biol Chem 279(45):46723–46732
Saveanu L, Carroll O et al (2005) Concerted peptide trimming by human ERAP1 and ERAP2 aminopeptidase complexes in the endoplasmic reticulum. Nat Immunol 6(7):689–697
Savill J, Dransfield I et al (2002) A blast from the past: clearance of apoptotic cells regulates immune responses. Nat Rev Immunol 2(12):965–975
Scaffidi P, Misteli T et al (2002) Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418(6894):191–195
Scheffer SR, Nave H et al (2003) Apoptotic, but not necrotic, tumor cell vaccines induce a potent immune response in vivo. Int J Cancer 103(2):205–211
Seong SY, Matzinger P (2004) Hydrophobicity: an ancient damage-associated molecular pattern that initiates innate immune responses. Nat Rev Immunol 4(6):469–478
Serwold T, Gaw S et al (2001) ER aminopeptidases generate a unique pool of peptides for MHC class I molecules. Nat Immunol 2(7):644–651
Shen L, Rock KL (2004) Cellular protein is the source of cross-priming antigen in vivo. Proc Natl Acad Sci USA 101(9):3035–3040
Shi Y, Evans JE et al (2003) Molecular identification of a danger signal that alerts the immune system to dying cells. Nature 425(6957):516–521
Sigman JA, Patwa TH et al (2005) Flexibility in substrate recognition by thimet oligopeptidase as revealed by denaturation studies. Biochem J 388(Pt 1):255–261
Silva CL, Portaro FC et al (1999) Thimet oligopeptidase (EC 3.4.24.15), a novel protein on the route of MHC class I antigen presentation. Biochem Biophys Res Commun 255(3):591–595
Somersan S, Larsson M et al (2001) Primary tumor tissue lysates are enriched in heat shock proteins and induce the maturation of human dendritic cells. J Immunol 167(9):4844–4852
Sparvero LJ, Asafu-Adjei D et al (2009) RAGE (receptor for advanced glycation endproducts), RAGE ligands, and their role in cancer and inflammation. J Transl Med 7:17
Srivastava P (2002) Roles of heat-shock proteins in innate and adaptive immunity. Nat Rev Immunol 2(3):185–194
Stoltze L, Schirle M et al (2000) Two new proteases in the MHC class I processing pathway. Nat Immunol 1(5):413–418
Thorburn J, Horita H et al (2009) Autophagy regulates selective HMGB1 release in tumor cells that are destined to die. Cell Death Differ 16(1):175–183
Uhl M, Kepp O et al (2009) Autophagy within the antigen donor cell facilitates efficient antigen cross-priming of virus-specific CD8 + T cells. Cell Death Differ 16(7):991–1005
Vanden Berghe T, Kalai M et al (2006) Necrosis is associated with IL-6 production but apoptosis is not. Cell Signal 18(3):328–335
Vanden Berghe T, Linkermann A et al (2014) Regulated necrosis: the expanding network of non-apoptotic cell death pathways. Nat Rev Mol Cell Biol 15(2):135–147
Venereau E, Casalgrandi M et al (2012) Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release. J Exp Med 209(9):1519–1528
Wang H, Bloom O et al (1999) HMG-1 as a late mediator of endotoxin lethality in mice. Science 285(5425):248–251
Weber CK, Adler G (2003) Acute pancreatitis. Curr Opin Gastroenterol 19(5):447–450
Wolfers J, Lozier A et al (2001) Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat Med 7(3):297–303
Woo M, Hakem R et al (2000) Executionary pathway for apoptosis: lessons from mutant mice. Cell Res 10(4):267–278
Yang H, Lundback P et al (2012) Redox modification of cysteine residues regulates the cytokine activity of high mobility group box-1 (HMGB1). Mol Med 18:250–259
Yewdell JW (2011) DRiPs solidify: progress in understanding endogenous MHC class I antigen processing. Trends Immunol 32(11):548–558
Yewdell JW, Anton LC et al (1996) Defective ribosomal products (DRiPs): a major source of antigenic peptides for MHC class I molecules? J Immunol 157(5):1823–1826
Yoon TJ, Kim JY et al (2008) Anti-tumor immunostimulatory effect of heat-killed tumor cells. Exp Mol Med 40(1):130–144
York IA, Bhutani N et al (2006) Tripeptidyl peptidase II is the major peptidase needed to trim long antigenic precursors, but is not required for most MHC class I antigen presentation. J Immunol 177(3):1434–1443
York IA, Chang SC et al (2002) The ER aminopeptidase ERAP1 enhances or limits antigen presentation by trimming epitopes to 8–9 residues. Nat Immunol 3(12):1177–1184
York IA, Mo AX et al (2003) The cytosolic endopeptidase, thimet oligopeptidase, destroys antigenic peptides and limits the extent of MHC class I antigen presentation. Immunity 18(3):429–440
Zhang Q, Raoof M et al (2010) Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464(7285):104–107
Acknowledgments
This work was supported by the Intramural Research Program of the NCI, NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gamrekelashvili, J., Greten, T.F. & Korangy, F. Immunogenicity of necrotic cell death. Cell. Mol. Life Sci. 72, 273–283 (2015). https://doi.org/10.1007/s00018-014-1741-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-014-1741-x