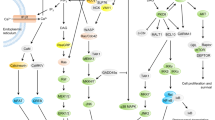
Abstract
Over the last two decades the molecular and cellular mechanisms underlying T cell activation, expansion, differentiation, and memory formation have been intensively investigated. These studies revealed that the generation of memory T cells is critically impacted by a number of factors, including the magnitude of the inflammatory response and cytokine production, the type of dendritic cell [DC] that presents the pathogen derived antigen, their maturation status, and the concomitant provision of costimulation. Nevertheless, the primary stimulus leading to T cell activation is generated through the T cell receptor [TCR] following its engagement with a peptide MHC ligand [pMHC]. The purpose of this review is to highlight classical and recent findings on how antigen recognition, the degree of TCR stimulation, and intracellular signal transduction pathways impact the formation of effector and memory T cells.

Similar content being viewed by others
References
Kaech SM, Wherry EJ, Ahmed R (2002) Effector and memory T-cell differentiation: implications for vaccine development. Nat Rev Immunol 2:251–262
Williams MA, Bevan MJ (2007) Effector and memory CTL differentiation. Annu Rev Immunol 25:171–192
Blattman JN, Antia R, Sourdive DJ, Wang X, Kaech SM, Murali-Krishna K, Altman JD, Ahmed R (2002) Estimating the precursor frequency of naive antigen-specific CD8 T cells. J Exp Med 195:657–664
Geiger R, Duhen T, Lanzavecchia A, Sallusto F (2009) Human naive and memory CD4+ T cell repertoires specific for naturally processed antigens analyzed using libraries of amplified T cells. J Exp Med 206:1525–1534
Moon JJ, Chu HH, Pepper M, McSorley SJ, Jameson SC, Kedl RM, Jenkins MK (2007) Naive CD4(+) T cell frequency varies for different epitopes and predicts repertoire diversity and response magnitude. Immunity 27:203–213
Obar JJ, Khanna KM, Lefrancois L (2008) Endogenous naive CD8+ T cell precursor frequency regulates primary and memory responses to infection. Immunity 28:859–869
Murali-Krishna K, Altman JD, Suresh M, Sourdive DJ, Zajac AJ, Miller JD, Slansky J, Ahmed R (1998) Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity 8:177–187
Butz EA, Bevan MJ (1998) Massive expansion of antigen-specific CD8+ T cells during an acute virus infection. Immunity 8:167–175
Miller JD, van der Most RG, Akondy RS, Glidewell JT, Albott S, Masopust D, Murali-Krishna K, Mahar PL, Edupuganti S, Lalor S, Germon S, Del Rio C, Mulligan MJ, Staprans SI, Altman JD, Feinberg MB, Ahmed R (2008) Human effector and memory CD8+ T cell responses to smallpox and yellow fever vaccines. Immunity 28:710–722
Mempel TR, Henrickson SE, Von Andrian UH (2004) T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427:154–159
Bousso P, Robey E (2003) Dynamics of CD8+ T cell priming by dendritic cells in intact lymph nodes. Nat Immunol 4:579–585
van Stipdonk MJ, Lemmens EE, Schoenberger SP (2001) Naive CTLs require a single brief period of antigenic stimulation for clonal expansion and differentiation. Nat Immunol 2:423–429
Masopust D, Kaech SM, Wherry EJ, Ahmed R (2004) The role of programming in memory T-cell development. Curr Opin Immunol 16:217–225
van Stipdonk MJ, Hardenberg G, Bijker MS, Lemmens EE, Droin NM, Green DR, Schoenberger SP (2003) Dynamic programming of CD8+ T lymphocyte responses. Nat Immunol 4:361–365
Jelley-Gibbs DM, Brown DM, Dibble JP, Haynes L, Eaton SM, Swain SL (2005) Unexpected prolonged presentation of influenza antigens promotes CD4 T cell memory generation. J Exp Med 202:697–706
Turner DL, Cauley LS, Khanna KM, Lefrancois L (2007) Persistent antigen presentation after acute vesicular stomatitis virus infection. J Virol 81:2039–2046
Henrickson SE, Mempel TR, Mazo IB, Liu B, Artyomov MN, Zheng H, Peixoto A, Flynn MP, Senman B, Junt T, Wong HC, Chakraborty AK, von Andrian UH (2008) T cell sensing of antigen dose governs interactive behavior with dendritic cells and sets a threshold for T cell activation. Nat Immunol 9:282–291
Prlic M, Hernandez-Hoyos G, Bevan MJ (2006) Duration of the initial TCR stimulus controls the magnitude but not functionality of the CD8+ T cell response. J Exp Med 203:2135–2143
Dolfi DV, Duttagupta PA, Boesteanu AC, Mueller YM, Oliai CH, Borowski AB, Katsikis PD (2011) Dendritic cells and CD28 costimulation are required to sustain virus-specific CD8+ T cell responses during the effector phase in vivo. J Immunol 186:4599–4608
Khanna KM, McNamara JT, Lefrancois L (2007) In situ imaging of the endogenous CD8 T cell response to infection. Science 318:116–120
Stock AT, Mueller SN, van Lint AL, Heath WR, Carbone FR (2004) Cutting edge: prolonged antigen presentation after herpes simplex virus-1 skin infection. J Immunol 173:2241–2244
Shaulov A, Murali-Krishna K (2008) CD8 T cell expansion and memory differentiation are facilitated by simultaneous and sustained exposure to antigenic and inflammatory milieu. J Immunol 180:1131–1138
McGill J, Van Rooijen N, Legge KL (2008) Protective influenza-specific CD8 T cell responses require interactions with dendritic cells in the lungs. J Exp Med 205:1635–1646
McGill J, Legge KL (2009) Cutting edge: contribution of lung-resident T cell proliferation to the overall magnitude of the antigen-specific CD8 T cell response in the lungs following murine influenza virus infection. J Immunol 183:4177–4181
Obst R, van Santen HM, Mathis D, Benoist C (2005) Antigen persistence is required throughout the expansion phase of a CD4(+) T cell response. J Exp Med 201:1555–1565
Williams MA, Bevan MJ (2004) Shortening the infectious period does not alter expansion of CD8 T cells but diminishes their capacity to differentiate into memory cells. J Immunol 173:6694–6702
Badovinac VP, Haring JS, Harty JT (2007) Initial T cell receptor transgenic cell precursor frequency dictates critical aspects of the CD8(+) T cell response to infection. Immunity 26:827–841
Foulds KE, Shen H (2006) Clonal competition inhibits the proliferation and differentiation of adoptively transferred TCR transgenic CD4 T cells in response to infection. Journal of immunology 176:3037–3043
Blair DA, Lefrancois L (2007) Increased competition for antigen during priming negatively impacts the generation of memory CD4 T cells. Proc Nat Acad Sci USA 104:15045–15050
Alam SM, Travers PJ, Wung JL, Nasholds W, Redpath S, Jameson SC, Gascoigne NR (1996) T-cell-receptor affinity and thymocyte positive selection. Nature 381:616–620
Naeher D, Daniels MA, Hausmann B, Guillaume P, Luescher I, Palmer E (2007) A constant affinity threshold for T cell tolerance. J Exp Med 204:2553–2559
Palmer E, Naeher D (2009) Affinity threshold for thymic selection through a T-cell receptor-co-receptor zipper. Nat Rev Immunol 9:207–213
Zehn D, Lee SY, Bevan MJ (2009) Complete but curtailed T-cell response to very low-affinity antigen. Nature 458:211–214
Goldrath AW, Bevan MJ (1999) Selecting and maintaining a diverse T-cell repertoire. Nature 402:255–262
Goldrath AW, Bevan MJ (1999) Low-affinity ligands for the TCR drive proliferation of mature CD8+ T cells in lymphopenic hosts. Immunity 11:183–190
Hamilton SE, Wolkers MC, Schoenberger SP, Jameson SC (2006) The generation of protective memory-like CD8+ T cells during homeostatic proliferation requires CD4+ T cells. Nat Immunol 7:475–481
Haluszczak C, Akue AD, Hamilton SE, Johnson LD, Pujanauski L, Teodorovic L, Jameson SC, Kedl RM (2009) The antigen-specific CD8+ T cell repertoire in unimmunized mice includes memory phenotype cells bearing markers of homeostatic expansion. J Exp Med 206:435–448
Hogquist KA, Jameson SC, Heath WR, Howard JL, Bevan MJ, Carbone FR (1994) T cell receptor antagonist peptides induce positive selection. Cell 76:17–27
Wu LC, Tuot DS, Lyons DS, Garcia KC, Davis MM (2002) Two-step binding mechanism for T-cell receptor recognition of peptide MHC. Nature 418:552–556
Stone JD, Chervin AS, Kranz DM (2009) T-cell receptor binding affinities and kinetics: impact on T-cell activity and specificity. Immunology 126:165–176
Wang XL, Altman JD (2003) Caveats in the design of MHC class I tetramer/antigen-specific T lymphocytes dissociation assays. J Immunol Methods 280:25–35
Fahmy TM, Bieler JG, Edidin M, Schneck JP (2001) Increased TCR avidity after T cell activation: a mechanism for sensing low-density antigen. Immunity 14:135–143
Slifka MK, Whitton JL (2001) Functional avidity maturation of CD8(+) T cells without selection of higher affinity TCR. Nat Immunol 2:711–717
von Herrath MG, Dockter J, Oldstone MB (1994) How virus induces a rapid or slow onset insulin-dependent diabetes mellitus in a transgenic model. Immunity 1:231–242
Zehn D, Bevan MJ (2006) T cells with low avidity for a tissue-restricted antigen routinely evade central and peripheral tolerance and cause autoimmunity. Immunity 25:261–270
Gronski MA, Boulter JM, Moskophidis D, Nguyen LT, Holmberg K, Elford AR, Deenick EK, Kim HO, Penninger JM, Odermatt B, Gallimore A, Gascoigne NR, Ohashi PS (2004) TCR affinity and negative regulation limit autoimmunity. Nat Med 10:1234–1239
Zehn D, Turner MJ, Lefrancois L, Bevan MJ (2010) Lack of original antigenic sin in recall CD8(+) T cell responses. Journal of immunology 184:6320–6326
Daniels MA, Teixeiro E, Gill J, Hausmann B, Roubaty D, Holmberg K, Werlen G, Hollander GA, Gascoigne NR, Palmer E (2006) Thymic selection threshold defined by compartmentalization of Ras/MAPK signalling. Nature 444:724–729
Yewdell JW, Reits E, Neefjes J (2003) Making sense of mass destruction: quantitating MHC class I antigen presentation. Nat Rev Immunol 3:952–961
Sykulev Y, Joo M, Vturina I, Tsomides TJ, Eisen HN (1996) Evidence that a single peptide-MHC complex on a target cell can elicit a cytolytic T cell response. Immunity 4:565–571
Purbhoo MA, Irvine DJ, Huppa JB, Davis MM (2004) T cell killing does not require the formation of a stable mature immunological synapse. Nat Immunol 5:524–530
Huppa JB, Davis MM (2003) T-cell-antigen recognition and the immunological synapse. Nat Rev Immunol 3:973–983
Irvine DJ, Purbhoo MA, Krogsgaard M, Davis MM (2002) Direct observation of ligand recognition by T cells. Nature 419:845–849
Porgador A, Yewdell JW, Deng Y, Bennink JR, Germain RN (1997) Localization, quantitation, and in situ detection of specific peptide-MHC class I complexes using a monoclonal antibody. Immunity 6:715–726
Sporri R, Reis e Sousa C. 2002. Self peptide/MHC class I complexes have a negligible effect on the response of some CD8+ T cells to foreign antigen. Euro J Immunol 32: 3161-70
Krogsgaard M, Li QJ, Sumen C, Huppa JB, Huse M, Davis MM (2005) Agonist/endogenous peptide-MHC heterodimers drive T cell activation and sensitivity. Nature 434:238–243
Wulfing C, Sumen C, Sjaastad MD, Wu LC, Dustin ML, Davis MM (2002) Costimulation and endogenous MHC ligands contribute to T cell recognition. Nat Immunol 3:42–47
Lo WL, Felix NJ, Walters JJ, Rohrs H, Gross ML, Allen PM (2009) An endogenous peptide positively selects and augments the activation and survival of peripheral CD4+ T cells. Nat Immunol 10:1155–1161
Stefanova I, Dorfman JR, Germain RN (2002) Self-recognition promotes the foreign antigen sensitivity of naive T lymphocytes. Nature 420:429–434
Tewari K, Walent J, Svaren J, Zamoyska R, Suresh M (2006) Differential requirement for Lck during primary and memory CD8+ T cell responses. Proc Natl Acad Sci USA 103:16388–16393
Au-Yeung BB, Levin SE, Zhang C, Hsu LY, Cheng DA, Killeen N, Shokat KM, Weiss A (2010) A genetically selective inhibitor demonstrates a function for the kinase Zap70 in regulatory T cells independent of its catalytic activity. Nat Immunol 11:1085–1092
Jordan MS, Singer AL, Koretzky GA (2003) Adaptors as central mediators of signal transduction in immune cells. Nat Immunol 4:110–116
Smith-Garvin JE, Burns JC, Gohil M, Zou T, Kim JS, Maltzman JS, Wherry EJ, Koretzky GA, Jordan MS (2010) T-cell receptor signals direct the composition and function of the memory CD8+ T-cell pool. Blood 116:5548–5559
Wiehagen KR, Corbo E, Schmidt M, Shin H, Wherry EJ, Maltzman JS (2010) Loss of tonic T-cell receptor signals alters the generation but not the persistence of CD8+ memory T cells. Blood 116:5560–5570
Bushar ND, Corbo E, Schmidt M, Maltzman JS, Farber DL (2010) Ablation of SLP-76 signaling after T cell priming generates memory CD4 T cells impaired in steady-state and cytokine-driven homeostasis. Proc Natl Acad Sci USA 107:827–831
D’Souza WN, Chang CF, Fischer AM, Li M, Hedrick SM (2008) The Erk2 MAPK regulates CD8 T cell proliferation and survival. J Immunol 181:7617–7629
Hedrick SM (2009) The cunning little vixen: foxo and the cycle of life and death. Nat Immunol 10:1057–1063
Barouch-Bentov R, Lemmens EE, Hu J, Janssen EM, Droin NM, Song J, Schoenberger SP, Altman A (2005) Protein kinase C-theta is an early survival factor required for differentiation of effector CD8+ T cells. J Immunol 175:5126–5134
Priatel JJ, Teh SJ, Dower NA, Stone JC, Teh HS (2002) RasGRP1 transduces low-grade TCR signals which are critical for T cell development, homeostasis, and differentiation. Immunity 17:617–627
Arbour N, Naniche D, Homann D, Davis RJ, Flavell RA, Oldstone MB (2002) c-Jun NH(2)-terminal kinase (JNK)1 and JNK2 signaling pathways have divergent roles in CD8(+) T cell-mediated antiviral immunity. J. J Exp Med 195:801–810
Kersh EN, Kaech SM, Onami TM, Moran M, Wherry EJ, Miceli MC, Ahmed R (2003) TCR signal transduction in antigen-specific memory CD8 T cells. J Immunol 170:5455–5463
Schmidt-Supprian M, Tian J, Ji H, Terhorst C, Bhan AK, Grant EP, Pasparakis M, Casola S, Coyle AJ, Rajewsky K (2004) I kappa B kinase 2 deficiency in T cells leads to defects in priming, B cell help, germinal center reactions, and homeostatic expansion. J Immunol 173:1612–1619
Schmidt-Supprian M, Courtois G, Tian J, Coyle AJ, Israel A, Rajewsky K, Pasparakis M (2003) Mature T cells depend on signaling through the IKK complex. Immunity 19:377–389
Hettmann T, Opferman JT, Leiden JM, Ashton-Rickardt PG (2003) A critical role for NF-kappaB transcription factors in the development of CD8+ memory-phenotype T cells. Immunol Lett 85:297–300
Teixeiro E, Daniels MA, Hamilton SE, Schrum AG, Bragado R, Jameson SC, Palmer E (2009) Different T cell receptor signals determine CD8+ memory versus effector development. Science 323:502–505
Pearce EL, Walsh MC, Cejas PJ, Harms GM, Shen H, Wang LS, Jones RG, Choi Y (2009) Enhancing CD8 T-cell memory by modulating fatty acid metabolism. Nature 460:103–107
Araki K, Turner AP, Shaffer VO, Gangappa S, Keller SA, Bachmann MF, Larsen CP, Ahmed R (2009) mTOR regulates memory CD8 T-cell differentiation. Nature 460:108–112
Pearce EL (2010) Metabolism in T cell activation and differentiation. Curr Opin Immunol 22:314–320
Juntilla MM, Koretzky GA (2008) Critical roles of the PI3K/Akt signaling pathway in T cell development. Immunol Lett 116:104–110
Macintyre AN, Finlay D, Preston G, Sinclair LV, Waugh CM, Tamas P, Feijoo C, Okkenhaug K, Cantrell DA (2011) Protein Kinase B Controls Transcriptional Programs that Direct Cytotoxic T Cell Fate but Is Dispensable for T Cell Metabolism. Immunity 34:224–236
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zehn, D., King, C., Bevan, M.J. et al. TCR signaling requirements for activating T cells and for generating memory. Cell. Mol. Life Sci. 69, 1565–1575 (2012). https://doi.org/10.1007/s00018-012-0965-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-0965-x