Abstract
Background
Calprotectin is calcium-binding protein which can be found in the cytosol of neutrophils. Several studies have studied its levels in preeclamptic women; however, to date there is no consensus regarding its effectiveness in the field.
Purpose
To investigate whether serum calprotectin levels are elevated among preeclamptic women compared to healthy controls.
Materials and methods
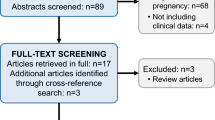
We used Medline (1966–2015), Scopus (2004–2015), ClinicalTrials.gov (2008–2015), Cochrane Central Register of Controlled Trials CENTRAL (1999–2015) and Google Scholar (2004–2015) search engines in our primary search, together with reference lists from included studies.
Results
Seven studies were finally included in our systematic review which recruited 439 women (245 with preeclampsia and 194 healthy controls). Their methodological quality was relatively high as they reached a score that ranged between 6 and 7 according to the Ottawa–Newcastle classification. All included studies reported that the serum calprotectin levels were significantly elevated among preeclamptic patients (p < 0.05). One study suggested that patients with severe preeclampsia have significantly higher levels of calprotectin than patients with mild preeclampsia (p = 0.01). However, to date there is no evidence regarding specific cut-off values which would help screen women for preeclampsia, or even follow the course of the disease.
Conclusion
Current evidence suggests that serum calprotectin is significantly raised among women with preeclampsia during the third trimester. Future research is needed to reach firm conclusions regarding its use as a potential screening and surveillance marker during the pregnancy course of women at risk of developing preeclampsia.

Similar content being viewed by others
References
Ronsmans C, Graham WJ. Maternal mortality: who, when, where, and why. Lancet. 2006;368:1189–200.
Thornton C, Dahlen H, Korda A, Hennessy A. The incidence of preeclampsia and eclampsia and associated maternal mortality in Australia from population-linked datasets: 2000–2008. Am J Obstet Gynecol. 2013;208(476):e1–5.
Lisonkova S, Sabr Y, Mayer C, Young C, Skoll A, Joseph KS. Maternal morbidity associated with early-onset and late-onset preeclampsia. Obstet Gynecol. 2014;124:771–81.
Lisonkova S, Joseph KS. Incidence of preeclampsia: risk factors and outcomes associated with early- versus late-onset disease. Am J Obstet Gynecol. 2013;209:544 e1–12.
Jelin AC, Cheng YW, Shaffer BL, Kaimal AJ, Little SE, Caughey AB. Early-onset preeclampsia and neonatal outcomes. J Matern Fetal Neonatal Med. 2010;23:389–92.
Raymond D, Peterson E. A critical review of early-onset and late-onset preeclampsia. Obstet Gynecol Surv. 2011;66:497–506.
Naljayan MV, Karumanchi SA. New developments in the pathogenesis of preeclampsia. Adv Chronic Kidney Dis. 2013;20:265–70.
Chaiworapongsa T, Chaemsaithong P, Yeo L, Romero R. Pre-eclampsia part 1: current understanding of its pathophysiology. Nat Rev Nephrol. 2014;10:466–80.
Wang A, Rana S, Karumanchi SA. Preeclampsia: the role of angiogenic factors in its pathogenesis. Physiology (Bethesda). 2009;24:147–58.
Verdonk K, Visser W, Steegers EA, Kappers M, Danser AH, van den Meiracker AH. New insights into the pathogenesis of pre-eclampsia: the role of angiogenesis-inhibiting factors. Ned Tijdschr Geneeskd. 2011;155:A2946.
Zhou Y, McMaster M, Woo K, Janatpour M, Perry J, Karpanen T, et al. Vascular endothelial growth factor ligands and receptors that regulate human cytotrophoblast survival are dysregulated in severe preeclampsia and hemolysis, elevated liver enzymes, and low platelets syndrome. Am J Pathol. 2002;160:1405–23.
Venkatesha S, Toporsian M, Lam C, Hanai J, Mammoto T, Kim YM, et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat Med. 2006;12:642–9.
Sahay AS, Sundrani DP, Joshi SR. Regional changes of placental vascularization in preeclampsia: a review. IUBMB Life 2015.
Laresgoiti-Servitje E. A leading role for the immune system in the pathophysiology of preeclampsia. J Leukoc Biol. 2013;94:247–57.
Lok CA, Jebbink J, Nieuwland R, Faas MM, Boer K, Sturk A, et al. Leukocyte activation and circulating leukocyte-derived microparticles in preeclampsia. Am J Reprod Immunol. 2009;61:346–59.
Mellembakken JR, Aukrust P, Olafsen MK, Ueland T, Hestdal K, Videm V. Activation of leukocytes during the uteroplacental passage in preeclampsia. Hypertension. 2002;39:155–60.
Striz I, Trebichavs.ky I. Calprotectin—a pleiotropic molecule in acute and chronic inflammation. Physiol Res. 2004;53:245–53.
Nilsen T, Sunde K, Larsson A. A new turbidimetric immunoassay for serum calprotectin for fully automatized clinical analysers. J Inflamm (Lond). 2015;12:45.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700.
Wells GA, Shea B, O’Connel D, Peterson J, V W, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analysis. www.ohri.ca/programs/clinical_epidemiology/oxford.asp, 2011.
Liosi S, Briana DD, Gourgiotis D, Boutsikou M, Baka S, Marmarinos A, et al. Calprotectin in human cord blood: relation to perinatal parameters and restricted fetal growth. J Perinat Med. 2010;38:523–6.
Staff AC, Braekke K, Johnsen GM, Karumanchi SA, Harsem NK. Circulating concentrations of soluble endoglin (CD105) in fetal and maternal serum and in amniotic fluid in preeclampsia. Am J Obstet Gynecol. 2007;197(176):e1–6.
Akcum S, Zulfikaroglu E, Eserdag S, Ozcan U. Plasma Calprotectin Levels in Preeclamptic Normotensive Pregnant and Nonpregnant Women. 2014.
Braekke K, Holthe MR, Harsem NK, Fagerhol MK, Staff AC. Calprotectin, a marker of inflammation, is elevated in the maternal but not in the fetal circulation in preeclampsia. Am J Obstet Gynecol. 2005;193:227–33.
Sugulle M, Kvehaugen AS, Braekke K, Harsem NK, Staff AC. Plasma calprotectin as inflammation marker in pregnancies complicated by diabetes mellitus and superimposed preeclampsia. Pregnancy Hypertens. 2011;1:137–42.
Börekçi B, Aksoy H, Öztürk N, Kadanalı S. Correlation between calprotectin and oxidized LDL in preeclampsia. Turk J Med Sci. 2009;39:191–5.
Feng C, Tao Y, Shang T, Yu M. Calprotectin, RAGE and TNF-alpha in hypertensive disorders in pregnancy: expression and significance. Arch Gynecol Obstet. 2011;283:161–6.
Ramma W, Buhimschi IA, Zhao G, Dulay AT, Nayeri UA, Buhimschi CS, et al. The elevation in circulating anti-angiogenic factors is independent of markers of neutrophil activation in preeclampsia. Angiogenesis. 2012;15:333–40.
Zaid O, Abdel-Maksoud H, Elmagid A, Omnia M. Biochemical effect of pre eclampsia on serum calprotectin, ascorbic acid, Uric Acid and Calcium. Benha Veterinary Med J. 2014;27(2):217–24.
Holthe MR, Staff AC, Berge LN, Fagerhol MK, Lyberg T. Calprotectin plasma level is elevated in preeclampsia. Acta Obstet Gynecol Scand. 2005;84:151–4.
Ramma W, Buhimschi IA, Zhao G, Dulay AT, Nayeri UA, Buhimschi CS, et al. The elevation in circulating anti-angiogenic factors is independent of markers of neutrophil activation in preeclampsia. Angiogenesis. 2012;15:333–40.
Dale I, Brandtzaeg P, Fagerhol MK, Scott H. Distribution of a new myelomonocytic antigen (L1) in human peripheral blood leukocytes. Immunofluorescence and immunoperoxidase staining features in comparison with lysozyme and lactoferrin. Am J Clin Pathol. 1985;84:24–34.
Brandtzaeg P, Dale I, Fagerhol MK. Distribution of a formalin-resistant myelomonocytic antigen (L1) in human tissues. I. Comparison with other leukocyte markers by paired immunofluorescence and immunoenzyme staining. Am J Clin Pathol. 1987;87:681–99.
Nakashige TG, Zhang B, Krebs C, Nolan EM. Human calprotectin is an iron-sequestering host-defense protein. Nat Chem Biol 2015.
Kerkhoff C, Klempt M, Sorg C. Novel insights into structure and function of MRP8 (S100A8) and MRP14 (S100A9). Biochim Biophys Acta. 1998;1448:200–11.
Yui S, Nakatani Y, Mikami M. Calprotectin (S100A8/S100A9), an inflammatory protein complex from neutrophils with a broad apoptosis-inducing activity. Biol Pharm Bull. 2003;26:753–60.
Espinoza J, Chaiworapongsa T, Romero R, Edwin S, Rathnasabapathy C, Gomez R, et al. Antimicrobial peptides in amniotic fluid: defensins, calprotectin and bacterial/permeability-increasing protein in patients with microbial invasion of the amniotic cavity, intra-amniotic inflammation, preterm labor and premature rupture of membranes. J Matern Fetal Neonatal Med. 2003;13:2–21.
Buimer M, Keijser R, Jebbink JM, Wehkamp D, van Kampen AH, Boer K, et al. Seven placental transcripts characterize HELLP-syndrome. Placenta. 2008;29:444–53.
Gallery ED, Campbell S, Arkell J, Nguyen M, Jackson CJ. Preeclamptic decidual microvascular endothelial cells express lower levels of matrix metalloproteinase-1 than normals. Microvasc Res. 1999;57:340–6.
Isaksen B, Fagerhol MK. Calprotectin inhibits matrix metalloproteinases by sequestration of zinc. Mol Pathol. 2001;54:289–92.
Bian Z, Shixia C, Duan T. First-trimester maternal serum levels of sFLT1, PGF and ADMA predict preeclampsia. PLoS One. 2015;10:e0124684.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors report they did not receive funding for the present paper.
Conflict of interest
None for all authors.
Additional information
Responsible Editor: John Di Battista.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pergialiotis, V., Prodromidou, A., Pappa, E. et al. An evaluation of calprotectin as serum marker of preeclampsia: a systematic review of observational studies. Inflamm. Res. 65, 95–102 (2016). https://doi.org/10.1007/s00011-015-0903-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-015-0903-0