Summary
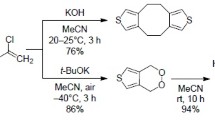
4:5-Benzothionaphthene (II) was obtained by theα-cyclization ofβ-naphthylω-dimethoxyethyl sulphide (I), and the hitherto unknown naphtho-(1′:8′-bc)-thiapyran (IV) by theperi-cyclization ofα-naphthylω-dimethoxyethyl sulphide (III).
Naphthalene-1:5-dithiol was obtained in 85% yield by an improved method. Condensation of naphthalene-1:5 and 2:6-dithiols with two molecules of bromoacetaldehyde dimethyl acetal gave (VIII) and (XI). The former cyclized in theperi-positions to give “1:6-thiapyrene” (IX) which was orange in colour, and the latter cyclized in theα-positions to give 4:5, 5′:4′-dithionaphthene (XII), an isoster of chrysene which was colourless. Chrysene and (XII) gave similar absorption spectra, which were markedly different from the absorption spectrum of (IX).
Similar content being viewed by others
References
Tilak..Proc. Ind. Acad. Sci., 1950,32A, 390.
Sunthankar and Tilak.., 396.
Mayer,et al...Annalen, 1931,488, 259.
Szmuszkovicz and Modest..J. A. C. S., 1950,72, 571.
.. See following paper.
Renard..Bull. Soc. Chim., 1889,3 (3), 958; 1891,5, 278.
Anschutz and Rodins..Ber., 1914,47, 2733.
Horton..J. Org. Chem., 1949,14, 761.
Albenga and Corbellini..Gazz. chim. Ital., 1931,61, 111.
Leuckart..J. pr. Chem., 1890,41, 189.
Author information
Authors and Affiliations
Additional information
Communicated by Dr. K. Venkataraman,f.a.sc.
Rights and permissions
About this article
Cite this article
Tilak, B.D. A new synthesis of thiophenes and thiapyrans. Proc. Indian Acad. Sci. (Math. Sci.) 33, 71 (1951). https://doi.org/10.1007/BF03172185
Received:
DOI: https://doi.org/10.1007/BF03172185