Summary
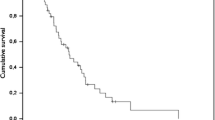
Six children (ages 2 to 14 years) with recurrent or poor risk primary brain tumors were treated with the `8 drugs in 1 day' chemotherapy regimen. As renal function deteriorated (median after five cycles), carboplatin was substituted in place of cisplatin at a median dose of 300 mg/m2 as a one hour infusion at hour three. Four patients also had their lomustine deleted and replaced by etoposide 100 mg/m2 at hour four. Compared to earlier cycles of chemotherapy, there was a moderate increase in transfusion requirement (especially platelets) but the incidences of fever and neutropenia were unchanged. Glomerular function as measured by nuclear glomerular filtration rate (GFR) remained unchanged or improved at the end of therapy. The severity of nausea and vomiting after substitution was markedly improved. Four of the six patients are still in complete remission 19 to 41 months after diagnosis or recurrence. It is feasible to substitute carboplatin and etoposide into the ‘8 drugs in 1 day’ regimen when extramedullary toxicity is found.
Similar content being viewed by others
References
Pendergrass TW, Milstein JM, Geyer JR, Mulne AF, Kosnik EJ, Morris JD, Heideman RL, Ruymann FB, Stuntz T, Bleyer WA: Eight drugs in one day chemotherapy for brain tumours, experience in 107 children and rationale for preradiation chemotherapy. J Clin Oncol 5: 1221–1231, 1987
Goldie JH: Rationale for ‘eight in one’ chemotherapy. J Clin Oncol 6: 395, 1988
Geyer JR, Pendergrass TW, Milstein JM, Bleyer WA: Eight drugs in one day, chemotherapy in children with brain tumours, a critical toxicity appraisal. J Clin Oncol 6: 996–1000, 1988
Allen JC, Walker R, Luks E, Jennings M, Barfoot S, Tan C: Carboplatin and recurrent childhood brain tumors. J Clin Oncol 5: 459–463, 1987
Tirelli U, D'Incalci M, Canetta R, Tumolo S, Franchin G, Vernesi A, Galligioni E, Trovo MG, Rossi C, Grigoletto E: Etoposide (VP-16-213) in malignant brain tumors, A phase II study. J Clin Oncol 2: 432–437, 1984
O'Dwyer PJ, Leyland-Jones B, Alonso MT, Marsoni S, Wittes RE: Etoposide (VP-16-213), current status of an active anticancer drug. N Engl J Med 312: 692–700, 1985
Gaynon PS, Ettinger LJ, Moel, Siegel SE, Baum ES, Krivit WA, Hammond GD: Pediatric phase I trial of Carboplatin: A Children's Cancer Study Group Report. Cancer Treat Rep 71: 1039–1042, 1987
Ross W, Rowe T, Glisson B, Yalowich J, Lin L: Tole of topoisomerase II in mediating epidophyllotoxin induced DNA cleavage. Cancer Res 44: 5857–5860, 1984
Horwich A, Duchesne G, Dearnaley D, Peckham M: Single agents carboplatin (SAC) as initial therapy for advanced seminoma. Proc Am Soc Clin Oncol 7: 117, 1988
Edmonson JH, McCormack GW, Krook JE, Long HJ, Jefferies JA, Richardson RL: Pilot study of cyclophosphamide plus carboplatin in advanced ovarian carcinoma. Cancer Treat Rep 71: 199–200, 1987
Egorin MJ, Van Echo DA, Tipping SJ, Olman EA, Whitecare MY, Thompson BW, Aisner J: Pharmacokinetics and dosage reduction of cis-diammine (1-1, cyclobutanedicarboxylate) platinum in patients with impaired renal function. Cancer Res 44: 5432–5438, 1984
Stewart DJ, Richard MT, Hugenholtz H, Dennery J, Nundy D, Prior J, Montpetit V, Hopkins HS: Penetration of teniposide (VM-26) into human intracerebral tumors: preliminary observations on the effect of tumor type, rate of drug infusion and prior treatment with amphotericin B or oral glycerol. J Neuro-Oncol 2: 315–324, 1984
Durand RE, Goldie JH: Interaction of etoposide and cisplatin in an in vitro tumor model. Cancer Treat Rep 71: 673–9, 1987
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chan, K.W., Fryer, C.J.H. & Steinbok, P. Modification of the 8 drugs in 1 day regimen: feasibility of substitution of other agents. J Neuro-Oncol 9, 153–157 (1990). https://doi.org/10.1007/BF02427836
Issue Date:
DOI: https://doi.org/10.1007/BF02427836