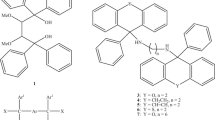
Abstract
The crystal and molecular structure is reported for the inclusion compound 2-[o-(triphenylphosphoranylidenamino)benzyliden]amino-1H-2,3-dihydroindazol-3-one/isopropylamine/water3b. The crystal structure consists of discrete dimeric salt-like aggregates joined together by strong N+−H...−O−C hydrogen bonds between pairs of centrosymmetrically-related indazolonate anions and isopropylammonium cations. Six other inclusion compounds have been prepared and characterized by NMR [with propylamine (3a), withtert-butylamine (3c), withsec-butylamine (3d), withtert-pentylamine (3e), with 1-methylbutylamine (3f) and withiso-pentylamine (3g)]. Two different arrangements are found, both with the host being in the anionic form. The guests are either: (i) one protonated amine and one water molecule (3b and3f); or (ii) one protonated amine and the corresponding neutral amine (3a, 3c, 3d, 3e and3g).
Similar content being viewed by others
References
P. Molina, A. Arques, R. Obón, A.L. Llamas-Saiz, C. Foces-Foces, R.M. Claramunt, C. López and J. Elguero:J. Phys. Org. Chem. 5, 507 (1992).
P.T. Beurskens, G. Admiraal, G. Beurskens, W.P. Bosman, S. García-Granda, R.O. Gould, J.M.M. Smits and C. Smykalla:The Dirdif Program System, Technical Report of the Crystallography Laboratory, University of Nijmegen, The Netherlands (1992).
J.M. Stewart, P.A. Machin, C.W. Dickinson, H.L. Ammon, H. Heck and H. Flack:The X-Ray System, Technical Report TR-446, Computer Science Centre, University of Maryland, USA (1976).
International Tables for X-Ray Crystallography, Kynoch Press, England, Vol. IV (1974), distr. Kluwer Academic Publishers, Dordrecht, The Netherlands.
J. Laynez, M. Menéndez, J.L. Saiz, A.L. Llamas-Saiz, C.Foces-Foces, J. Elguero, P. Molina, M. Alajarín, and A. Vidal:J. Chem. Soc., Perkin Trans. 2, 709 (1993).
C. López, R.M. Claramunt, S. Trofimenko and J. Elguero:Can. J. Chem. 71, 678 (1993).
F.H. Allen, J.E. Davies, J.J. Galloy, O. Kennard, C.F. Macrae, E.M. Mitchell, G.F. Mitchell, J.M. Smith and D.G. Watson:J. Chem. Info. Comp. Sci. 31, 187 (1991).
J.J.P. Stewart: MOPAC6.0, Frank J. Seiler Research Laboratory, United States Air Force Academy, CO 80840, USA (1990).
F.H. Cano and M. Martínez-Ripoll:J. Mol. Struct. (Theochem) 258, 139 (1992).
E. Breitmaier and W. Voelter:Carbon-13 NMR Sectroscopy, 3rd ed. VCH, New York, 1987.
J. Llinares, J. Elguero, R. Faure and E.J. Vincent:Org. Magn. Reson. 14, 20 (1980).
R. Faure, J. Llinares and J. Elguero:An. Quim. 81C, 167 (1985).
M. Czugler, J.A. Angyán, G. Náray-Szabó and E. Weber:J. Am. Chem. Soc. 108, 1275 (1986).
I. Csöregh, M. Czugler, K.W. Törnros, E. Weber and J. Ahrendt:J. Chem. Soc., Perkin Trans. 2, 1491 (1989).
E. Weber: inInclusion Compounds (eds. J.L. Atwood, J.E.D. Davies and D.D. MacNicol), Oxford University Press, Oxford, Vol. 4, p. 188 (1991).
A. Quick and D.J. Williams:Can. J. Chem. 54, 2482 (1976).
Author information
Authors and Affiliations
Additional information
Supplementary Data relating to this article (structure factors, thermal components, hydrogen parameters and bond distances and angles, and13C-NMR shifts) are deposited with the British Library at Boston Spa, Wetherby, West Yorkshire, U.K., as Supplementary Publication No. SUP 82155 (23 pages).
For Part 1, see Reference [1].
Rights and permissions
About this article
Cite this article
Foces-Foces, C., Llamas-Saiz, A.L., Claramunt, R.M. et al. Host-guest chemistry. 2. Amine inclusion compounds of 2-[o-(triphenylphosphoranylidenamino)benzyliden]amino-1H-2,3-dihydroindazol-3-one. X-ray structure of its 1∶1∶1 inclusion complex with isopropylamine and water. J Incl Phenom Macrocycl Chem 16, 155–168 (1993). https://doi.org/10.1007/BF00709149
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00709149