Summary
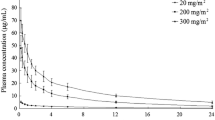
Flavone acetic acid pharmacokinetics were studied in 31 patients in a phase I clinical trial. The drug was given by i.v. infusions over 1, 1.5, 3, and 6 h at doses ranging from 0.5 to 6.4 g/m2. The pharmacokinetic parameters were determined according to a nonlinear model including Michaelis-Menten-type kinetics. The mean elimination half-life is 4.8 h and the mean volume of distribution of the central compartment, 7.61. Our model predicted a maximal tolerated dose (MTD) of 11.1 g/m2 on the basis of the “therapeutic window” concept, very close to the clinically observed MTD of 10 g/m2. This model is also operational when different protocols of inoculation are considered, such as a divided-dose schedule vs a unique infusion, and indicates that, at the MTD, injections should be made every 72 h to avoid drug accumulation.
Similar content being viewed by others
References
Bissery MC, Chabot GG, Corbett TH, Rutkowski K (1986) Flavone acetic acid (FAA, NSC-347512): non linearity of area under the concentration x time curves with changing dosage. Proc Am Assoc Cancer Res 27:282
Boston RC, Phillips DR (1983) Evidence of possible dose-dependent doxorubicin plasma kinetics in man. Cancer Treat Rep 67:63
Corbett TH, Bissery MC, Wozniak A, Plowman J, Polin L, Tapazoglou E, Dieckman J, Valeriote F (1986) Activity of flavone acetic acid (NSC-347512) against solid tumors of mice. Invest New Drugs 4:207
D'Argenio DZ, Schumitzky A (1979) A program package for simulation and parameter estimation in pharmacokinetic systems. Comput Progr Biomed 9:115
Garrett ER, Bres J, Schnelle K, Rolf LL (1974) Pharmacokinetics of saturably metabolized amobarbital. J Pharmacokinet Biopharm 2:43
Gouyette A (1983) Pharmacokinetics: statistical moment calculations. Arzneim-Forsch/Drug Res 33:173
Gouyette A, Huertas D, Droz JP, Rouesse J, Amiel JL (1982) Pharmacokinetics of 2-methyl-9-hydroxyellipticinium acetate (NSC-264137) in cancer patients (phase I study). Eur J Cancer Clin Oncol 18:1285
Gouyette A, Apchin A, Foka M, Richard JM (1986a) Pharmacokinetics of intra-arterial and intravenous cisplatin in head and neck cancer patients. Eur J Cancer Clin Oncol 22: 257
Gouyette A, Ducret JP, Caille P, Amiel JL, Rouesse J, Foka M, Carde P, Hayat M, Sancho-Garnier H (1986b) Preliminary phase I clinical study and pharmacokinetics of (1,2-diaminocyclohexane) (isocitrato) platinum(II) or PHIC. Anticancer Res 6:1127
Kerr DJ, Kaye SB, Graham J, Cassidy J, Harding M, Setanoians A, McGrawth JC, Vezin WR, Cunningham D, Forrest G, Soukop M (1986) Phase I and pharmacokinetic study of LM985 (flavone acetic ester). Cancer Res 46:3142
Kerr DJ, Kaye SB, Cassidy J, Bradley C, Rankin EM, Adams L, Setanoians A, Young T, Forrest G, Soukop M, Clavel M (1987) Phase I and pharmacokinetic study of flavone acetic acid. Cancer Res 47:6776
McAinsh J, Gay MA (1985) The critical Michaelis-Menten elimination model for propanolol. Eur J Drug Metab Pharmacokinet 10:241
Plowman J, Narayanan VL, Dykes D, Szarvasi E, Briet P, Yoder OC, Paull KD (1986) Flavone acetic acid: a novel agent with preclinical antitumor activity against colon adenocarcinoma 38 in mice. Cancer Treat Rep 70:631
Powis G (1983) Dose-dependent metabolism, therapeutic effect, and toxicity of anticancer drugs in man. Drug Metab Rev 14:1145
Redmond JW, Batley M, Djordjevic MA, Innes RW, Kuempel PL, Rolfe BG (1986) Flavones induce expression of nodulation genes in Rhizobium. Nature 323:632
Wagner JG, Gyves JN, Stetson PL, Walker-Andrews SC, Wollner IS, Cochran MK, Ensminger WD (1986) Steady-state nonlinear pharmacokinetics of 5-fluorouracil during hepatic arterial and intravenous infusion in cancer patients. Cancer Res 46:1499
Zaharko DS, Grieshaber CK, Plowman J, Cradock JC (1986) Therapeutic and pharmacokinetic relationships of flavone acetic acid: an agent with activity against solid tumors. Cancer Treat Rep 70:1415
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gouyette, A., Kerr, D.J., Kaye, S.B. et al. Flavone acetic acid: a nonlinear pharmacokinetic model. Cancer Chemother. Pharmacol. 22, 114–119 (1988). https://doi.org/10.1007/BF00257307
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00257307