Abstract
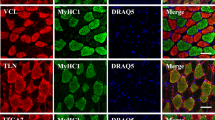
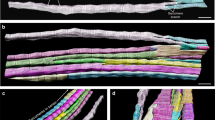
A rat muscle spindle typically contains four intrafusal fibers — one nuclear bag2, one nuclear bag1 and two nuclear chain fibers. We compared the sequence of formation of the three intrafusal fiber types among the tibialis anterior (TA), soleus (SOL) and medial gastrocnemius (MG) muscles using immunocytochemistry of spindle-specific myosin heavy chain isoforms. Spindles of the TA began to differentiate earlier and acquired the full complement of intrafusal fibers sooner than spindles of the SOL or MG muscles. At the onset of spindle assembly, the intrafusal myotubes expressed myosin heavy chains similar to those expressed by extrafusal myotubes. The first intrafusal myotube then differentiated into the bag2 fiber regardless of the muscle. However, the fate of the second-forming intrafusal myotube varied among the muscles studied. It usually differentiated into a chain fiber in the TA, into a bag1 fiber in the SOL, and into either a bag1 or a chain in the MG. The fate of the third-forming intrafusal myotube was reciprocal to that of the second; i.e. in those spindles in which the bag1 fiber was second to form, a chain was third, and vice versa. The fourth and last intrafusal myotube gave rise to a chain fiber. The inter- and intramuscular variability in the fate of intrafusal myotubes of the second and third generation argues against the existence of a program intrinsic to the myotubes that would mandate their differentiation along specific paths. Rather, an extrinsic regulatory factor, probably associated with the primary afferent neuron, may govern differentiation of pluripotential myotubes into particular types of intrafusal fiber. The fate of the intrafusal myotubes might then depend on the timing of the regulatory effect of afferents relative to the stage of development of the intrafusal bundle.
Similar content being viewed by others
References
Bakker GJ, Richmond FJR (1981) Two types of muscle spindles in cat neck muscles: a histochemical study of intrafusal fiber composition. J Neurophysiol 45:973–986
Banks RW, Harker DW, Stacey MJ (1977) Correlation between ultrastructure and histochemistry of mammalian intrafusal muscle fibres using a combined histochemical and ultrastructural technique. J Anat 123:783–796
Barker D, Banks RW (1986) The muscle spindle. In: Engel AG, Banker BQ (eds) Myology. McGraw-Hill, New York, pp 309–341
Barker D, Banks RW, Harker DW, Milburn A, Stacey MJ (1976) Studies of the histochemistry, ultrastructure, motor innervation and regeneration of mammalian intrafusal muscle fibres. Prog Brain Res 44:67–88
Cho M, Webster SG, Blau HM (1993) Evidence for myoblast-extrinsic regulation of slow myosin heavy chain expression during muscle fiber formation in embryonic development. J Cell Biol 121:795–810
Condon K, Silberstein L, Blau HM, Thompson WJ (1990) Development of muscle fiber types in the prenatal rat hindlimb. Dev Biol 138:256–274
Draeger A, Weeds AG, Fitzsimons RB (1987) Primary, secondary and tertiary myotubes in developing skeletal muscle: a new approach to the analysis of human myogenesis. J Neurol Sci 81:19–43
Duxson MJ, Ross JJ, Harris AJ (1986) Transfer of differentiated synaptic terminals from primary myotubes to new-formed muscle cells during embryonic development in the rat. Neurosci Lett 71:147–152
Duxson AJ, Usson Y, Harris AJ (1989) The origin of secondary myotubes in mammalian skeletal muscles: ultrastructural studies. Development 107:743–750
Harris AJ, Fitzsimons RB, McEwan JC (1989) Neural control of the sequence of expression of myosin heavy chain isoforms in foetal mammalian muscles. Development 107:751–769
Hoh JFY, Hughes S (1989) Immunocytochemical analysis of the perinatal development of cat masseter muscle using anti-myosin antibodies. J Muscle Res Cell Motil 10:312–325
Jacoby J, Ko K, Weiss C, Rushbrook JI (1989) Systematic variation in myosin expression along extraocular muscle fibres of the adult rat. J Muscle Res Cell Motil 11:25–40
Kucera J (1982a) One-bag-fiber muscle spindles in tenuissimus muscles of the cat. Histochemistry 76:315–328
Kucera J (1982b) The topography of long nuclear chain intrafusal fibers in the cat muscle spindle. Histochemistry 74:183–197
Kucera J (1982c) A study of sensory innervation to long nuclear chain intrafusal fibers in the cat muscle spindle. Histochemistry 75:113–121
Kucera J (1983) Multiple-bag-fiber muscle spindles in tenuissimus muscles of the cat. Histochemistry 79:457–476
Kucera J, Walro JM (1989) Postnatal expression of myosin heavy chains in muscle spindles of the rat. Anat Embryol 179:369–376
Kucera J, Walro JM (1990a) Origin of intrafusal muscle fibers in the rat. Histochemistry 93:567–580
Kucera J, Walro JM (1990b) Treatment with β-bungarotoxin blocks muscle spindle formation in fetal rats. Development 110:483–489
Kucera J, Walro JM (1991) Aggregation of myonuclei and the spread of slow-tonic myosin immunoreactivity in developing muscle spindles. Histochemistry 96:381–390
Kucera J, Walro JM (1992) Superfluousness of motor innervation for the formation of muscle spindles in neonatal rats. Anat Embryol 186:301–309
Kucera J, Walro JM (1993) Transient expression of a slow-tonic MHC isoform by extrafusal fibers in the developing rat. Anat Embryol 186:409–418
Kucera J, Dorovini-Zis K, Engel WK (1978) Histochemistry of rat intrafusal muscle fibers and their motor innervation. J Histochem Cytochem 26:973–988
Kucera J, Walro JM, Reichler J (1989) Role of nerve and muscle factors in the development of rat muscle spindles. Am J Anat 186:144–160
Kucera J, Walro JM, Reichler J (1991) Neural organization of spindles in three hindlimb muscles of the rat. Am J Anat 190:74–88
Kucera J, Walro JM, Gorza L (1992) Expression of type-specific MHC isoforms in rat intrafusal muscle fibers. J Histochem Cytochem 40:293–307
Kucera J, Walro JM, Gao Y (1993) Influence of muscle cell substrates on differentiation of intrafusal fiber types in neonatal rats. Neuroscience 52:1001–1008
Landon DN (1972) The fine structure of the equatorial regions of developing muscle spindles in the rat. J Neurocytol 1:189–210
Landon DN (1985) Skeletal muscle-normal morphology, development and innervation. In: Mastaglia FL, Walton J (eds) Skeletal muscle pathology. Churchill Livingstone, New York, pp 1–87
Milburn A (1973) The early development of muscle spindles in the rat. J Cell Sci 12:175–195
Milburn A (1984) Stages in the development of cat muscle spindles. J Embryol Exp Morphol 82:177–216
Narusawa M, Fitzsimons RB, Izumo S, Nadal-Ginard B, Rubinstein NA, Kelly AM (1987) Slow myosin in developing rat skeletal muscle. J Cell Biol 104:447–459
Pedrosa F, Thornell L-E (1990) Expression of myosin heavy chain isoforms in developing rat muscle spindles. Histochemistry 94:231–244
Pedrosa F, Butler-Browne GS, Dhoot GK, Fischman DA, Thornell L-E (1989) Diversity in expression of myosin heavy chain isoforms and M-band proteins in rat muscle spindles. Histochemistry 92:185–194
Pedrosa F, Soukup T, Thornell L-E (1990) Expression of an alpha cardiac-like myosin heavy chain in muscle spindle fibres. Histochemistry 95: 105–113
Pedrosa-Domellöf F (1991) Expression of myosin heavy chain isoforms in rat muscle spindles. Medical dissertations, University of Umea, Umea, Sweden, New Series No. 304, pp 1–51
Pedrosa-Domellöf F, Soukup T, Thornell L-E (1991) Rat muscle spindle immunocytochemistry revisited. Histochemistry 96:327–338
Rudnicki MA, Jackowski G, Saggin L, McBurney MW (1990) Actin and myosin expression during development of cardiac muscle from cultured embryonal carcinoma cells. Dev Biol 138:348–358
Zelená J (1957) The morphogenetic influence of innervation on the ontogenic development of muscle spindles. J Embryol Exp Morphol 5:283–292
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kucera, J., Walro, J.M. Sequences of intrafusal fiber formation are muscle-dependent in rat hindlimbs. Anat Embryol 190, 273–286 (1994). https://doi.org/10.1007/BF00234305
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00234305