Abstract
The main aim of the present study was to compare mucoadhesion and cellular uptake efficiency of chitosan (CS) and chitosan oligosaccharide (COS) surface-modified polymer nanoparticles (NPs) for mucosal delivery of proteins. We have developed poly (d, l-lactide-co-glycolide) (PLGA) NPs, surface-modified COS-PLGA NPs and CS-PLGA NPs, by using double emulsion solvent evaporation method, for encapsulating bovine serum albumin (BSA) as a model protein. Surface modification of NPs was confirmed using physicochemical characterization methods such as particle size and zeta potential, SEM, TEM and FTIR analysis. Both surface-modified PLGA NPs displayed a slow release of protein compared to PLGA NPs. Furthermore, we have explored the mucoadhesive property of COS as a material for modifying the surface of polymeric NPs. During in vitro mucoadhesion test, positively charged COS-PLGA NPs and CS-PLGA NPs exhibited enhanced mucoadhesion, compared to negatively charged PLGA NPs. This interaction was anticipated to improve the cell interaction and uptake of NPs, which is an important requirement for mucosal delivery of proteins. All nanoformulations were found to be safe for cellular delivery when evaluated in A549 cells. Moreover, intracellular uptake behaviour of FITC-BSA loaded NPs was extensively investigated by confocal laser scanning microscopy and flow cytometry. As we hypothesized, positively charged COS-PLGA NPs and CS-PLGA NPs displayed enhanced intracellular uptake compared to negatively charged PLGA NPs. Our results demonstrated that CS- and COS-modified polymer NPs could be promising carriers for proteins, drugs and nucleic acids via nasal, oral, buccal, ocular and vaginal mucosal routes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Therapeutic macromolecules, including, proteins, peptides, antigens, monoclonal antibodies, antibody fragments, DNA, RNA, siRNA and ribozymes have been known to play a significant role in tackling numerous difficult-to-treat diseases. In particular, more than 130 active and specific protein and peptide pharmaceuticals have been approved by the USFDA for human use, or many more are currently in clinical development for treating various diseases [1, 2]. However, endosomal escape of therapeutic proteins still poses a great challenge. The major limitations include chemical and enzymatic instability, poor absorption (low permeability) through biological membranes, rapid blood clearance, and immunogenicity of these biomolecules, resulting in extremely low bioavailability [3–5]. Biomedical and translational science-related literature widely claims that translation of protein pharmaceuticals from bench to bedside is a major hurdle in their successful development and availability [6].
Over the last two decades, developments of polymeric NPs for delivery of various therapeutic macromolecules have attracted the attention of protein formulation scientists. Furthermore, encapsulation of proteins into nanoparticulate system has been found as a powerful and effective approach to (i) increase the stability of these biomolecules against enzymatic degradation, (ii) release them in a controlled fashion (iii) prolong their circulation time, (iv) minimize their non-specific biodistribution (iv) achieve site-specific delivery and (v) improve their in vivo bioactivity [5, 7].
Amongst the polymeric nanoparticles, those formulated using the FDA approved, biocompatible and biodegradable synthetic polymer, poly (d, l-lactide-co-glycolide) (PLGA) have received special attention. PLGA NPs have been extensively studied for delivering numerous therapeutic agents including a variety of drugs [8], proteins [9] and nucleic acids [10, 11]. However, the overall negative charge of these NPs has been reported to diminish their interaction with the negatively charged cell membrane [12]. More importantly, rapid opsonization of hydrophobic PLGA NPs is major limitation that hinders their employment for biomedical applications.
In fact, the size and surface charge of NPs play a crucial role in determining their electrostatic interaction with the cell membrane, mechanism of internalization and intracellular localization [13–15]. From this perspective, surface alteration of NPs is looked upon as a promising approach for favourably modifying the physico-chemical characteristics of negatively charged and hydrophobic nanocarriers. Surface modification of NPs with mucoadhesive polymer (for example, chitosan) has been observed to improvise their cellular uptake, while minimizing their toxicity and opsonization [16]. More importantly, chitosan is a natural, cationic, biocompatible, biodegradable, non-immunogenic, safe and an FDA-recognized mucoadhesive polymer [17]. It has also been categorised as a generally recognized as safe (GRAS) ingredient under sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act. Mucoadhesive- and permeation-enhancing properties of chitosan increases the electrostatic interaction between chitosan-modified NPs and negatively charged cell membrane. Polymer coating increases mucosal residence time and control the release of therapeutics from particulate systems [18]. Many pre-clinical and clinical studies have suggested potential of this mucoadhesive biopolymer for delivering biomacromolecules.
However, chitosan has several limitations such as low water solubility at physiological pH, which may be a major limitation for administration of therapeutics [19, 20]. Low molecular weight water-soluble chitosans, known as chitosan oligosaccharides (COS), are known to circumvent limitations of chitosan owing to their excellent solubility at physiological conditions and their ability to protect the proteins, loaded in nano-formulations, from enzymatic degradation [21, 22]. Recently, COS has been successfully explored as a vehicle for delivery of drugs [23], proteins [24], antigens [25] and nucleic acids [26]. Researchers have employed it as a pH-sensitive stealth coating polymer for tumour-specific drug delivery [27]. COS has also been investigated as an effective material for modifying the surface of nanoparticulate systems such as liposomes [28], solid lipid NPs [29], gold NPs [30] and iron oxide NPs [31, 32]. However, none of these investigations have employed COS for fabricating surface-modified PLGA NPs for delivering proteins, to the best of our knowledge. COS modified PLGA NPs have been hypothesized to confer an overall cationic charge to the negatively charged PLGA NPs. Coating of mucoadhesive polymers on nanoparticulate systems (PLGA NPs in our case) has been conjectured to enhance the bioavailability of therapeutic proteins by improving their cellular interaction and uptake. The main aim of the present study was to compare mucoadhesion and cellular uptake enhancement potential of COS and CS as mucoadhesive materials for modifying the surface of PLGA nanoparticles to apply them in mucosal delivery of proteins. In an attempt to verify this hypothesis, we developed PLGA NPs (negative surface) and surface-modified PLGA NPs (positive surface) containing bovine serum albumin as a model protein. The surface morphology, encapsulation efficiency, in vitro release and in vitro cytotoxicity were evaluated for all the nanoformulations. For the first time, we have explored the mucoadhesive property of COS as a material to modify the surface of protein-encapsulated PLGA NPs. Cell uptake behaviour of FITC-BSA (in-house synthesized fluorescent protein)-loaded PLGA NPs, COS-PLGA NPs and CS-PLGA NPs was extensively investigated using confocal laser scanning microscopy (CLSM) and flow cytometry.
Materials and methods
Materials
Poly (d, l-lactide-co-glycolide), (Purasorb® Lactic: Glycolic acid ratio; 50:50) was provided as a gift sample from Purac Biomaterials (Gorinchem, Netherlands). Chitosan (average molecular weight, 75–85 % degree of deacetylation, FITC (fluorescein-5-isothiocyanate), Bradford reagent, mucin from porcine stomach Type III, (bound sialic acid 0.5–1.5 %, partially purified powder) and 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide were purchased from Sigma Chemical Co. (St. Louis, MO, USA) and chitosan oligosaccharide (20 KDa), a gift sample from Amicogen Co., Ltd, Korea. Bovine serum albumin and pluronic F-68 were purchased from Himedia Pvt. Ltd (Mumbai, India). Trehalose was supplied as a gift sample from Hayashibara Co. Ltd-Gangwal chemicals Pvt. Ltd (Mumbai, Maharashtra, India). Acetone, dimethyl sulfoxide, glacial acetic acid, glucose and mannitol were purchased from SD fine chemicals (Mumbai, India). All other chemicals used in the study were of analytical grade. Potassium bromide (IR grade) was purchased from Merck, (Darmstadt, Germany). Deionized and double-distilled water (Milli-Q Plus system, Millipore, Bedford, MA, USA) were used in all the experiments.
Cell lines and culture media
A549 cells were grown at 37 °C in 5 % CO2 humidified atmosphere. A549 human lung alveolar epithelial cells from (National Centre for Cell Science (NCCS), Pune, Maharashtra, India) were cultured in 37.5-cm2 flasks and cells were seeded into wells of 96-well plates in Dulbecco’s modified eagle medium (DMEM) (Himedia) supplemented with 10 % foetal bovine serum (FBS). The cells were incubated for 24 h for adherence at 37 °C in an incubator (Galaxy® 170 S; New BrunswickTM, Eppendorf AG, Germany) at 95 % air and 5 % CO2. Before use, A549 cells were allowed to grow until confluence and were detached from the flask with trypsin phosphate versene glucose (TPVG) solution (Himedia), and then, the cell density was calculated and used at the desired density in later experiments. Dulbecco's phosphate-buffered saline (DPBS, Himedia) was used in all experiments.
Preparation of PLGA and surface-modified PLGA NPs
The protein-loaded PLGA and surface-modified NPs (COS-PLGA and CS-PLGA NPs) were prepared by double emulsion solvent evaporation by homogenization technique [33]. Briefly, a primary emulsion was formulated by emulsifying 1 mL organic phase (1 % w/v PLGA in acetone) with 10 mL aqueous phase (1 % w/v Pluronic F-68) containing of 10 mg BSA using Ultra-Turrax T25 digital homogenizer (Werke, Staufen, Germany) at 15,000 rpm for 5 min. Further, secondary emulsion was obtained by drop-wise addition of primary to the outer aqueous phase (5 mL; 1 % w/v PF-68) at 15,000 rpm for 5 min. Then, resultant secondary emulsion was placed on magnetic stirrer (IKA® RO 10P, IKA, Werke, Staufen, Germany) at 700 rpm for complete evaporation of acetone.
Surface-modified COS-PLGA NPs or CS-PLGA NPs were prepared by using an aqueous phase containing equal volume of COS or chitosan solution (0.2 % w/v) and PF-68 (1 % w/v) and following the same method as followed for PLGA NPs. The protein-loaded NPs were collected by centrifugation (Model CPR 24, REMI Instruments, Mumbai, Maharashtra, India) at 10,000 rpm for 10 min and washed with distilled water to remove excess surfactants. For cell uptake studies, in-house synthesized FITC-BSA conjugate was encapsulated into NPs instead of BSA following the same procedure as employed for BSA.
Synthesis and characterization of FITC-conjugated BSA
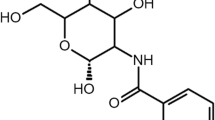
FITC-conjugated BSA was synthesized based on the reaction between isothiocyanate group of FITC and amine groups of BSA, as outlined in Fig. 1. In brief, 20 mg of FITC was taken in 100-mL amber-coloured glass bottles containing 20 mL dimethyl sulfoxide (DMSO) and placed on a magnetic stirrer for about 10 min. Subsequently, 20 mL of BSA solution in PBS pH 7.4 (2 % w/v) was added into the FITC solution and placed on a magnetic stirrer for 12 h in the dark, at ambient temperature. Thereafter, the FITC-BSA precipitate was collected by centrifugation at 10,000 rpm for 10 min. Further, unlabelled FITC was washed with deionized water and separated by centrifugation until no fluorescence was detected in the supernatant. Finally, FITC-BSA was purified by using dialysis (cellulose semipermeable membrane, MWCO = 12–14 kDa, Hi media, India) against the excess amount of deionized water for 72 h, under darkness with continuous stirring. The synthesized fluorescent protein was lyophilized and stored at 2–8 °C until further use. Lyophilized FITC-BSA conjugate was characterized by Fourier transform infrared (FTIR) and compared with the characteristics of marketed FITC-BSA (Sigma Chemical Co., St. Louis, MO, USA).
Physicochemical characterization of NPs
Particle size, size distribution and zeta potential measurement
Particle size, size distribution and zeta potential (surface charge, ζ) measurements of NPs were performed by using Zetasizer Nano ZS 90 (Malvern Instruments Ltd., Worcestershire, UK). All measurements were conducted at 25 °C and intensity of the scattered light was measured at a fixed angle of 90°. Dynamic light scattering (DLS) was used to measure the mean hydrodynamic diameter (nm) of NPs. This technique analyses the diffusion speed of the particles in a solution due to Brownian motion, which is related to their particle size. The particle size distribution was expressed in terms of polydispersity index (PDI). A low PDI value indicates narrow size distribution, indicating the formation of uniform and homogenous NPs. Similarly, laser Doppler anemometry (LDA) was used to determine zeta potential (mV) or electrophoretic mobility nanoparticulate formulation, which was used as an indicator of particle stability. A high zeta potential value (more negative or positive) is indicative of the colloidal stability of NPs. Viscosity and refraction indices were set equal to those specific of water. All samples were analysed in triplicate (n = 3, each involving 12–20 runs) and reported as mean ± S.D.
Surface morphology
The morphology and surface appearances of the PLGA NPs, COS-PLGA NPs and CS-PLGA NPs were examined by scanning electron microscopy and transmission electron microscopy.
Scanning electron microscopy
The surface morphology of the formulated NPs was visualized by Field Emission Gun-Scanning Electron Microscopy (FEG-SEM, JEOL JSM-7600 F). Briefly, 10 μL of PLGA NPs, COS-PLGA NPs and CS-PLGA NPs were placed on a brass stud and all samples were dried at ambient temperature overnight. Then, gold was sputtered on the dried brass stud, and measurements were conducted at 10 kV accelerating voltage in a FEG-SEM.
Transmission electron microscopy
The surface morphological characteristics of PLGA, COS-PLGA and CS-PLGA NPs were examined on a high-resolution transmission electron microscope (HR TEM, JEM 2100 JEOL Ltd., Tokyo, Japan). All samples were negatively stained by using 10 μL of 1 % (w/v) phosphotungstic acid for 1 mL of the formulations. Thereafter, 10 μL of sample mixtures were placed on Formvar® carbon film-coated copper grids (200 meshes, Electron Microscopy Services, Ft. Washington, PA) and air-dried at room temperature. Finally, all the samples were examined under a voltage of 200 kV.
Determination of encapsulation efficiency and loading capacity of protein in NPs
The encapsulation efficiency of BSA in NPs was determined through indirect method by separating the non-encapsulated (unloaded) BSA from the NPs. Briefly, 15 mL of the protein-encapsulated NP suspension was centrifuged at 10, 000 rpm, 4 °C for 15 min and supernatants were collected. Free amount of BSA in supernatant was quantified with Microplate reader (Synergy H1 model, BioTek Instruments, Inc., Winooski, VT, USA) at 595 nm by using Bradford protein assay reagent. Bradford reagent enables colorimetric determination of protein concentration in solution, based on the formation of a complex between the dye, Brilliant Blue G and proteins in solution [34]. All samples were analysed in triplicate (n = 3) and the results were expressed as mean ± SD. Encapsulation efficiency (EE) was calculated using Eq. (1):
For determining the loading capacity (LC%) of NPs, BSA from the NPs was extracted by dispersing a known amount loaded NPs in 0.5 mL acetone. Subsequently, an equal volume of deionized water (0.5 mL) was added and mixed for 3 min. Apart from extracting BSA, this procedure resulted in precipitation of PLGA. The amount of BSA in nanoparticle was calculated using Eq. (2):
Freeze-drying of protein-encapsulated nanoparticulate delivery systems
Optimization of cryoprotectants and process parameters
To improve long-term physicochemical stability of colloidal NPs, cryoprotectants and their concentrations were optimized during their freeze-drying (lyophilization). Various cryoprotectants or lyoprotectants like glucose, mannitol and trehalose were studied for their efficacy to protect the colloidal stability of NPs. Different concentrations of glucose, mannitol and trehalose (1, 5, 10 and 20 % w/v) were weighed into individual lyophilization glass vials containing PLGA NPs, COS-PLGA NPs and CS-PLGA NPs. All the samples were vortexed for 2–5 min and then frozen at −40.0 °C for 24 h. The freeze-drying conditions followed have been stated in Table 1. After freezing, the vials were transferred into a freeze dryer (FreeZone Plus 6 Liter Cascade Console Freeze Dry Systems, Labconco Corporation, Kansas City, MO) at shelf temperature −20 °C and condenser surface temperature of −80 °C, at a pressure of 0.100 mbar. The temperature RAMP rates were operated at 1 °C/min throughout the study. After lyophilization, all the samples were stored at temperature 4 °C until further use. Freeze-drying was performed with three independent samples of each formulation.
Reconstitution of freeze-dried samples
The freeze-dried samples were reconstituted with deionized water by gently vortexing (REMI CM 101, REMI Instruments, Mumbai, Maharashtra, India) for 3 min to yield homogenous dispersions. After reconstitution, each sample was characterized for particle size, distribution and zeta potential. All samples were analysed in triplicate (n = 3).
FTIR spectroscopy
Surface modification of the PLGA NPs was assessed using FTIR spectroscopy wherein the spectra of all three NPs (unmodified, CS and COS modified) were obtained with potassium bromide (KBr) pellet method. A small amount of NPs (~10 mg) was mixed with dried KBr and compressed into pellet under a hydraulic press at 10,000–14,000 psi. All the pellets were analysed in the wave number range of 400 and 4000 cm−1 with a resolution of 1 cm−1 by using FTIR spectrophotometer (IR affinity-1, Shimadzu, Tokyo, Japan) and the characteristic peaks for different samples were recorded.
In vitro release of protein from nanoparticles
The protein release profile from each nanoparticulate formulation was determined by incubating BSA-loaded freeze-dried PLGA NPs, COS-PLGA NPs and CS-PLGA NPs in 10 mL of PBS (pH 6.4) at 37 °C, under stirring at 100 rpm for 48 h [35]. At different time intervals (0.5, 1, 2, 4, 8, 12, 24 and 48 h), samples were withdrawn for analysis and an equal volume of fresh PBS medium was added to maintain the sink conditions. Thereafter, supernatants were collected by centrifugation (Model CPR 24, REMI Instruments, Mumbai, Maharashtra, India) at 10,000 rpm for 10 min at 4 °C. The amount of BSA released in the supernatant was analysed with Bradford protein assay as described previously using a microplate reader (Synergy H1 model, BioTek Instruments, Inc., Winooski, VT, USA). The release rate (RR %) of all nanoformulations was calculated by the given formula: RR = (W i /W total) × 100 %, where W i is the amount of BSA in release medium at the time-point, and W total is the total amount of BSA in the equal volume of NPs suspensions before performing release study [36]. Each nanoparticle formulation was analysed in triplicate (n = 3).
Mucoadhesive strength of NPs
The mucoadhesive strength of all the nanoparticulate formulations was evaluated by studying the interaction between mucin and NPs in aqueous solution [37]. In brief, equal volumes of mucin (0.5 mg/mL in PBS pH 6.4) and reconstituted NPs were mixed and incubated at 37 °C for 1 h. Subsequently, the NPs were centrifuged at 10,000 rpm for 1 h at 4 °C. The pellet was separated and the free amount of mucin in the supernatant was quantified by UV-Visible spectrophotometer (UV-1650 PC, Shimadzu) at 255 nm. All nanoformulations were analysed in triplicate (n = 3). The mucin-binding efficiency of NPs (%) or mucoadhesive strength of NPs was calculated by using equation stated as follows:
Cellular cytotoxicity study
Cytotoxicity of NPs was determined by measuring viability of NP-treated cells using MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) colorimetric assay. Briefly, A549 cells were seeded in 96-well plates at a density of 20, 000 cells/well. The cells were allowed to grow for 24 h at 37 ° C in presence of 5 % CO2 and 95 % relative humidity. The freeze dried NPs (PLGA NPs, COS-PLGA NPs and CS-PLGA NPs) were dispersed in DMEM medium containing 2 % FBS to yield various concentrations (0.25, 0.5, 0.75, 1.0, 1.25, 1.5, 1.75 and 2 mg/mL). The samples were incubated with cells only after sterilization using UV irradiation for 20 min in a biosafety cabinet (ESCO Airstream Class II type Biological Safety Cabinet; ESCO Technologies, Hatboro, PA). Further, the medium of the cells was replaced with 200 μL of NP samples, followed by incubation for 24 and 48 h at 95 % humidity and 5 % CO2. At the specified time intervals, medium was removed and the cell monolayers were washed with DPBS (pH 7.4). At each time interval, MTT solution (1.25 mg/mL in DPBS) was added to culture medium and incubated with the cells for 4 h. Subsequently, the unreacted MTT was removed and dimethyl sulfoxide (200 μL) was added to each well to dissolve formazan crystals obtained from MTT. Cell culture medium and Triton X-100 (1 %) was used as a negative control (0 % cytotoxicity) and positive control (100 % cytotoxicity), respectively. The cell viability of nanoparticulate formulation was determined by measuring the amount of formazan at 570 nm with background correction at 650 nm. The amount of formazan was considered to be directly proportional to the number of living cells in the culture. All the samples were assayed in hexaplicate.
Cell viability (%) of NPs was calculated according to the Eq. (3) stated as below:
In vitro cellular uptake study
CLSM
Cellular uptake of fluorescently labelled NPs (loaded with FITC-BSA) was visualized using confocal laser scanning microscopy (CLSM). A549 cells were seeded at density of 20,000 cells per well in cell imaging plate (24-well; Eppendorf) at 37 ° C in 95 % humidity and 5 % CO2. After 24 h, the medium was removed and the cells were washed with DPBS (pH = 7.4). For investigating the cell uptake, the cells were incubated with FITC-BSA encapsulated PLGA NPs, COS-PLGA NPs and CS-PLGA NPs for 6 h, in serum-free DMEM media. After incubation, media containing non-internalized NPs were removed and the cells were washed several times with DPBS to remove the particles merely sticking to the outer cell surface. Then the cells were harvested in DMEM containing 10 % FBS for 24 h. Thereafter the cells were rinsed and fixed with 4 % w/v paraformaldehyde (sigma aldrich) for 10 min and washed twice with cold DPBS (pH 7.4). Subsequently, cell nucleus and cell membranes were stained with blue-fluorescent DAPI nucleic acid stain and Deep Red CellMaskTM plasma membrane stain (Life Technologies, Inc., Gaithersburg, Maryland, USA), respectively. Cell monolayers were washed several times with DPBS. Finally, cell-associated fluorescence of FITC-BSA-loaded NPs was visualized using CLSM (Leica DMi8, Germany), at an excitation wavelength of 488 nm. All measurements were performed in triplicates.
Cellular internalization assay by flow cytometry
A549 cells were seeded in 6-well plates at a density of 0.5x106 cells per well and incubated for 24 h at 37 °C in 95 % air and 5 % CO2 environment. After 24 h, media was removed by aspiration and cells were washed with PBS and then treated with FITC-BSA encapsulated PLGA, COS-PLGA and CS-PLGA NPs for 6 h in serum-free media. After incubation, media was replaced with DMEM with 10 % FBS and incubated for 24 h further. Cells were then harvested using trypsin (0.25 %) and rinsed twice with DPBS. Further, live cells were stained with CellMaskTM for cell membrane (far-red plasma membrane stain; Molecular probes, Life technologies), by the method similar to that adopted for CLSM investigations. Thereafter, the cells were incubated with Trypan blue for 1 min in order to quench the FITC-fluorescent signal coming from NPs adsorbed to the cell surface. Finally, cell suspension was introduced into flow cytometry (Amnis ImageStreamx platform) to quantify fluorescent intensity of intracellularly internalized nanoparticles and results were analysed by IDEAS software (Amnis). All samples were characterized at an excitation wavelength at 488 nm (FITC) and 645 nm (CellMaskTM).
Statistical analysis
Data were performed for statistical significance by two-way ANOVA (GraphPad Prism 5.00, Instat Software, San Diego, CA, USA) followed by the Bonferroni post-test. The results were considered statistically significant at p values of < 0.05.
Results and discussion
Physicochemical characterization of nanoparticles
Particle size, distribution and surface charge analysis
BSA protein was successfully encapsulated into PLGA NPs and surface-modified PLGA NPs using double emulsion solvent evaporation method. Various physico-chemical parameters such as particle size, polydispersity index, surface charge, encapsulation efficiency and loading capacity of all formulations have been summarized in Table 2. The mean diameters of PLGA NPs increased from 143.6 ± 3.99 nm to 170.7 ± 2.34 nm and 200.2 ± 3.75 nm, upon surface coating with COS and CS, respectively. Such increase in mean particle diameter, indicating the occurrence of surface modification, has been previously reported for alternative NP systems [38]. COS-PLGA NPs were smaller in size compare to CS-PLGA NPs, which may due to the difference in the molecular weight of these polymers. These low molecular weight derivatives of CS are readily water-soluble, a property attributable to their molecular weight and degree of acetylation [24, 39], which makes them more preferable for drug delivery applications as compared to CS. Polydispersity index (PDI) of the PLGA, COS-PLGA and CS-PLGA NPs was found to be 0.085 ± 0.008, 0.134 ± 0.01 and 0.169 ± 0.01, respectively, indicating their homogenous (narrow-size) distribution. As stated in Table 2, PLGA NPs exhibited a negative surface charge of −27.2 ± 2.8 mV, whereas COS-PLGA and CS-PLGA NPs exhibited a positive surface charge of 38.4 ± 0.1 mV and 52.6 ± 1.26 mV, respectively. Negative charge of PLGA NPs was due to the presence of carboxylic groups (−COOH) on the surface of PLGA NPs. However, the cationic nature of COS-PLGA NPs and CS-PLGA NPs, that confirmed the surface modification of PLGA, may be due to the presence of free protonated amino group (−NH2) of both the polymers. Thus, it indicated that positive surface charge with small size (100–200 nm) of NPs are attractive features for enhanced cell uptake [40].
Characterization of FITC-conjugated BSA
FITC-labelled BSA was successfully synthesized as a water-soluble, macromolecular fluorophore to investigate the cellular uptake of PLGA and modified PLGA NPs. The conjugation of FITC with BSA was confirmed by recording the FTIR spectra of pure BSA, FITC and both, commercial and synthesized FITC-BSA conjugates (spectra included in supplementary information). The FTIR spectrum of pure BSA showed presence of characteristic bands at 3342, 1660 and 1537 cm−1, corresponding to the stretching vibration of –OH (and –NH2), amide-I and amide-II bands, respectively. These bands were also observed in both commercial and synthesized FITC-BSA [41]. The characteristic absorption band of FITC at 2034 cm−1, attributed to the isothiocyanate (−N = C = S) stretching, was found to be absent in the spectrum of the FITC-BSA [42]. This indicated the existence of coordination interaction between isothiocyanate group of FITC and amine group of BSA.
In addition, conjugation of FITC-BSA was further confirmed by comparing the FTIR spectra of synthesized conjugate with that of pure, commercial FITC-BSA. Both commercial and synthesized conjugates demonstrated similar spectra showing distinct characteristic peaks at around 3300, 1656 and 1544 cm−1, indicative of N-H stretching on FITC-BSA. Thus, the analysis confirmed successful conjugation of FITC with BSA.
Surface morphology
SEM and TEM were both employed to assess the size and morphology of uncoated and surface-coated PLGA NPs, as shown in Fig. 2. TEM, although an effective method for analysing size and morphology of NPs, involves a drying step that leads to removal in the hydration shell of NPs. Thus, the particle size of NPs as comprehended from TEM micrographs is not always in compliance with the results of dynamic light scattering. Thus the particle size of NPs was confirmed using FEG-SEM, a technique that allows adequate control over variations in the pressure and temperature inside the sample chamber to offer a good parallel with relative humidity of real atmospheric conditions [43].
The surface morphology results indicated that all NPs were of size <200 nm and possessed a spherical shape with a smooth surface. As shown in Fig. 2a–c, the SEM micrographs revealed the presence of homogenous NPs having a consistent size. The TEM micrographs, presented in Fig. 2d–f, exhibited a distinct presence of a surface layer in case of COS-PLGA and (Fig. 2e) CS-PLGA NPs as (Fig. 2f) compared to uncoated PLGA NPs. Thus, analysis by electron microscopy confirmed uniform coating of CS and COS on the surface of PLGA NPs. The ability of these techniques to intricately reveal the surface characteristics of NP formulations has also been reported by other researchers for PLGA NPs coated with a different grade of chitosan [44].
Encapsulation efficiency and loading capacity of protein in NPs
The BSA entrapment efficiency of PLGA NPs, COS-PLGA and CS-PLGA NPs was found to be 96.44 ± 0.68, 92.84 ± 1.58, 92.75 ± 2.63, respectively, with at drug loading level around 6 % (Table 2). The double emulsion method of PLGA hydrophobic polymer minimizes escape of the hydrophilic macromolecules (BSA) to the aqueous core of NPs, resulting in high encapsulation efficiency hydrophilic molecules. Such observations have been earlier reported by other scientists [45]. Both COS-PLGA and CS-PLGA NPs exhibited slightly lower encapsulation efficiency as compared to PLGA NPs.
Optimization of cryoprotectants and characterization of freeze-dried nanoparticles
The effect of cryoprotectants on freeze-drying of NPs was successfully investigated through characterization of reconstituted NPs for their particle size, distribution and surface charge, shown in Fig. 3. In this study, we have selected different cryoprotectants including mannitol (sugar alcohol), trehalose (non-reducing sugar) and glucose (reducing sugar) for freeze-drying of NPs [46]. Various concentrations (1, 5, 10 and 20 % w/v) of cryoprotectants and process parameters were investigated, as mentioned in Table 1.
Figure 3a reveals the effect of various concentrations of mannitol, trehalose and glucose on particle size and homogeneity of PLGA NPs. Mannitol was ineffective in protecting the NPs during lyophilization, as evident from the large particle size and non-homogenous distribution of the reconstituted NPs. However, trehalose and glucose were observed to be effective cryoprotectants and resulted in small particle size (<120 nm) and a narrow distribution (<0.08). The surface charge of PLGA NPs was more anionic upon addition of sugars, see Fig. 3d. But, formulation with glucose resulted in non-porous sticky cake.
Figure 3b shows the effect of various concentrations of mannitol, trehalose and glucose on particle size and distribution of COS-PLGA NPs. Addition of mannitol resulted in a large particle size and broad distribution. In contrast, both trehalose and glucose (10 & 20 % w/v) showed similar particle size (<200 nm) and polydispersity index (<0.3). Effect of the same cryoprotectants was also studied on CS-PLGA NPs as shown in Fig. 3c. Once again, mannitol was found to be an ineffective cryoprotectant for lyophilization of these NPs. It has been previously reported that during the freezing step of lyophilization, mannitol has a tendency to crystallise and form a eutectic phase with ice. This causes phase separation of mannitol, making it unavailable for NP stabilization. Moreover, the mechanical stress exerted by the growth of cryoprotectant and ice crystals may cause aggregation of NPs. We hypothesize occurrence of a similar mechanism upon freeze-drying of our NPs in presence of mannitol [47].
However, CS-PLGA NPs with 10 % w/v trehalose showed a mean particle size of 157 ± 9.7 nm with PDI of 0.235 ± 0.04 and zeta potential of 35.9 ± 5.0 mV (Fig. 3c, f). Overall, only trehalose at the concentration of 10 % w/v was found to be an effective cryoprotectant in terms of maintaining the particle size and homogeneity of the NPs.
This may be due to the ability of trehalose to stabilise the NPs by steric hindrance or electrostatic repulsion, at the concentration optimized in present study (10 % w/v). Higher concentration of trehalose was not found to result in additional stabilization of NPs. This may due to cases of irreversible fusion of nanoparticles at high concentration of cryoprotectants [47]. Furthermore, statistical analysis suggested that no significant difference was observed between the mean particle size and size distribution of PLGA NPs, COS-PLGA NPs and CS-PLGA NPs, before and after freeze-drying. From the results, it was observed that trehalose as a cryoprotectant resulted in a statistically significant decrease in the mean particle size and PDI of all formulations compared to mannitol and glucose (ns = not significant remarked in Fig. 3). Therefore, a minimum amount of 10 % w/v trehalose was chosen as the cryoprotectant for all three formulations during freeze-drying. This concentration was also chosen since higher cryoprotectant concentration has been known to result in particle aggregation during freeze-drying of cationic modified silica NPs and triblock poly (lactic acid)-poly (ethylene oxide) copolymer nanoparticles [48, 49]. Similar results have also been reported in previously published reports [50, 51].
FTIR spectroscopy
To confirm the surface modification of PLGA with CS or COS, all the NP formulations were analysed using FTIR to investigate any changes in the functional groups of the polymers. The FTIR spectrum (Fig SI-1 in supplementary information) of PLGA NPs exhibited characteristic peak at 3454 and 1739–1639 cm−1 attributed to –OH and carboxylic (−COOH) groups of PLGA, respectively [52]. FTIR spectra of surface modified PLGA NPs (Fig SI-2 in supplementary information) shows the broad and strong peak at 3290–3350 cm−1 corresponding to the amino (−NH2) group of coating polymers, CS and COS, Similar results were reported for surface modified PLGA NPs [53, 54]. Moreover, modification of carboxylic group of PLGA was also demonstrated by the appearance of a characteristic peak at ~1450 and ~2900 cm−1, which would be assigned to the amino group of chitosan and COS on PLGA surface. Similar observations were also reported in recent publications [38]. Thus, the results confirmed the surface modification of PLGA NPs with CS or COS.
In vitro release of protein from nanoparticles
In vitro release of BSA from PLGA NPs, COS-PLGA NPs and CS-PLGA NPs was measured at different time points, as shown in Fig. 4. PBS at pH 6.4 was used as the release medium to simulate nasal conditions since the NPs may be used for nasal vaccine delivery in future. All nanoformulations exhibited a biphasic protein release profile, typical of biodegradable PLGA polymer in physiological pH conditions. This release behaviour consisted of an initial burst release (~60 %) of BSA within 3 h, the remaining amount being release in sustained manner. Statistical analysis suggested that there was no significant difference in release rate between PLGA NPs and COS-PLGA NPs at all the time points. However, CS-PLGA NPs (66.50 ± 3.42 %) showed a significant difference in rate of release at time point 6 h compared to PLGA NPs (72.77 ± 2.10 %) (*p < 0.05), slowing the release rate of BSA. At the end of 48 h, both COS-PLGA NPs (88.15 ± 1.76 %) and CS-PLGA NPs (85.42 ± 1.44 %) displayed a slightly slow release compared to PLGA NPs (91.15 ± 4.72 %). This may be due to a control on protein release exerted by an additional polymer layer on the surface of PLGA NPs [55]. Amongst the surface-modified NPs, CS-PLGA NPs exhibited a slow release compared COS-PLGA NPs and PLGA NPs at all time intervals. This may be due to water insolubility of CS at the physiological pH. Moreover, these positively charged NPs are a suitable system for the intra-cytoplasmic delivery of drugs, proteins, or genes because they are more likely to escape into the cytoplasmic compartment for effective release [56]. This enables a longer availability of the encapsulated antigen protein for uptake by immunological B cell uptake, thereby enhancing the immune response. Similar results have been reported by other researchers in case of nanocarriers of trimethyl chitosan (TMC) and TMC-PLGA NPs [57]. In summary, the formulations were found to be suitable for protein delivery via nasal route.
Assessment of mucoadhesion strength of nanoparticles
Evaluation of mucoadhesive characteristics of PLGA NPs, COS-PLGA NPs and COS-PLGA NPs was conducted by assessing their mucin-binding efficiency at the nasal pH (pH 6.4). The results have been presented in Fig. 5. The mucoadhesive property of COS was studied for the first time, to our knowledge. This is specifically relevant due to the preferred utilization of these polymers in drug delivery due to their aqueous solubility. Possession of mucoadhesive characteristics can thus broaden the scope of application of this polymeric vehicle. In our study, COS-PLGA NPs and CS-PLGA NPs showed increased mucin-binding efficiency of 73.48 ± 0.01 and 73.58 ± 0.01, respectively, as compared to PLGA NPs (59.042 ± 0.01 %). Both chitosan and COS act as mucoadhesive polymers due to enhanced electrostatic interaction between negatively charged sialic acid residues of mucin and positively charged amino groups of COS and CS [58]. This interaction was anticipated to improve the cell interaction and uptake of NPs, which is an important requirement for mucosal delivery of therapeutics [55].
Cellular cytotoxicity study
The cytotoxicity of PLGA NPs, COS-PLGA NPs and CS-PLGA NPs as a delivery system was investigated on human lung epithelial A549 cells using MTT assay (Fig. 6). MTT is a water-soluble yellow coloured dye that is readily taken up by viable cells and reduced by mitochondrial dehydrogenases to formazan crystals (purple) [59]. After a 24-h post-incubation, all formulations resulted in around 80 % cell survival at different concentrations (0.25, 0.5, 0.75, 1, 1.25, 1.5, 1.75 and 2 mg/ml). Results indicated that the no formulation displayed cytotoxicity to the A549 cells as compared to positive control (100 % cytotoxic). Because PLGA, COS and CS polymers are well-established biodegradable and biocompatible components, these were well tolerated by the cells. Our results were in compliance with those from other investigations which have reported CS-PLGA NPs to be safe even at much higher concentrations of 20 mg/mL [55, 60, 61]. The results indicated the safety and suitability of modified NPs for further cellular investigations.
In vitro cytotoxicity of PLGA NPs, COS-PLGA NPs and CS-PLGA NPs on A549 cells. Cell viability (%) was determined by a MTT assay after 24 h incubation. The cytotoxicity of NPs with various concentrations was calculated with respect to respective controls (Triton X 100 = positive control and untreated cells = negative control). The results represent mean ± SD (n = 6)
Effect of surface modification of PLGA NPs on cell uptake
Visualization of cellular uptake by confocal laser scanning microscopy
The effect of COS and CS modified PLGA NPs on cellular uptake was evaluated by using FITC-BSA encapsulated NPs. Recently, FITC-BSA was used in PLGA NPs, chitosan-PLGA NPs and glycol chitosan-PLGA NPs and further cellular uptake of NPs was studied in nasal associated lymphoid tissue (NALT) [55]. Figure 7 shows CLSM images of A549 cells incubated for with FITC-BSA loaded NPs at 37 °C in 95 % air and 5 % CO2. Confocal image (Fig. 7a) clearly indicates that free FITC-BSA conjugate, by itself, was not taken up by the A549 cells due to electrostatic repulsion between negatively charged membrane and the conjugate. Also very poor uptake of FITC-BSA loaded PLGA NPs was observed in A549 cells, after 6 h incubation (Fig. 7b). This may again be attributed to the electrostatic repulsion between the negatively charged PLGA NPs and the negatively charged cell membranes. However, compared to the FITC-BSA control, the results suggested that the nanoparticulates could act as good vehicles for intracellular delivery of therapeutic cargos. Cellular uptake of FITC-BSA-loaded COS-PLGA NPs and CS-PLGA NPs was much greater compared to uncoated PLGA NPS, which may be due to their overall cationic nature and, hence, better attachment to the anionic cell surface. CLSM images revealed perinuclear localization of FITC-BSA-loaded COS-PLGA NPS and CS-PLGA NPs (visualized as green fluorescent spots in Fig. 7c, d). Enhanced intracellular delivery is an attractive attribute, especially for delivering protein-based vaccines, as it can lead to a more robust immune response involving cytotoxic T cells. This has already been reported in the case of alternative nanocarriers [62].
Typical confocal laser scanning microscope (CLSM) images of intracellular delivery of FITC-BSA solution (a), FITC-BSA loaded PLGA NPs (b), COS-PLGA NPs (c) and CS-PLGA NPs (d) in A549 cells. Untreated A549 Cells considered as controls for this experiment. Cell nuclei were stained with DAPI nucleic acid stain (blue part), NPs were loaded with FITC-BSA (green part), cell membranes stained with CellMaskTM plasma membrane stain (deep-red fluorescent cell labelling reagent), and overlay. The green fluorescence of FITC-BSA for all formulation indicated by arrows
Flow cytometry analysis
Figure 8a–c shows flow cytometry results of A549 cells treated with PLGA NPs, COS-PLGA NPs and C. CS-PLGA NPs, respectively. As we expected, both surface-modified COS-PLGA NPs and CS-PLGA NPs displayed a shift in fluorescence intensity due to high internalization of positively charged NPs compared to negatively charge PLGA NPs (shown in Fig. 8d). These results suggested that surface modification with cationic polymers enhanced the electrostatic interaction of PLGA NPs with negatively charged cell membrane. COS is suitable as a mucoadhesive material for surface modification of PLGA NPs for intracellular targeting because COS-PLGA NPs indicated improved mucoadhesion and cellular uptake compared to PLGA NPs.
Cellular internalization of FITC-BSA loaded NPs in A549 cells analysed by using imaging flow cytometry (Amnis Image Streamx MK II platform, Amnis corp. USA). a PLGA NPs. b COS-PLGA NPs. c CS-PLGA NPs shows representative images captured after a 6-h incubation. In all images, first column shows cells in the bright field (BF), second column shows the cell membrane, third column shows the signal of the side scatter of FITC-BSA loaded NPs and forth column shows images of the cell membrane merged with the side scatter signal of FITC-BSA loaded NPs. d. Flow cytometry histograms illustrating cellular uptake of PLGA NPs (red), COS-PLGA NPs (green) and CS-PLGA NPs (blue) analysed by Amnis IDEAS software version 6.0
Conclusion
In recent years, low molecular weight chitosan oligosaccharide has attracted great attention because of improved water solubility and reduced toxicity under physiological condition and high cellular internalization. We designed a surface modified COS-PLGA NPs and CS-PLGA NPs for mucosal delivery of proteins and further investigated for cellular uptake studies in A549 cells without any significant cytotoxicity. However, surface modification with COS as well as CS did not show any significant difference in cytotoxicity of PLGA NPs. Improved mucoadhesion results suggest that polymer NPs with the CS and COS coating represents promising candidates for efficient mucosal delivery of therapeutic macromolecules.
The positively charged amino groups on the COS-PLGA NPs and CS-PLGA NPs surface enhance both, mucoadhesion and cellular uptake via electrostatic interaction with the negatively charged mucus and cell membrane. Moreover, these positively charged NPs are suitable system for the intracytoplasmic delivery of drugs, proteins, or genes because they are more likely to escape into the cytoplasmic compartment for effective release. Both confocal laser scanning microscopy and flow cytometry study confirmed the improved internalization of surface modified COS-PLGA NPs and CS-PLGA NPs in cells as compared to PLGA NPs. These positively charged biopolymers promote the cellular internalization of negatively charged NPs by surface modification method. Thus, surface-modified PLGA NPs have proven as promising carriers for mucosal delivery of proteins and better alternative for uncoated negative charged PLGA nanoformulations. However, more research is needed to fully explore the underlying mechanisms of cellular uptake of COS surface-modified polymer nanoparticles.
References
Yadav SC, Kumari A, Yadav R. Development of peptide and protein nanotherapeutics by nanoencapsulation and nanobioconjugation. Peptides. 2011;32(1):173–87.
Gu W-X, Zhu M, Song N, Du X, Yang Y-W, Gao H. Reverse micelles based on biocompatible β-cyclodextrin conjugated polyethylene glycol block polylactide for protein delivery. J Mater Chem B. 2015;3(2):316–22.
Irie T, Uekama K. Cyclodextrins in peptide and protein delivery. Adv Drug Deliv Rev. 1999;36(1):101–23.
Park K, Kwon IC, Park K. Oral protein delivery: current status and future prospect. React Funct Polym. 2011;71(3):280–7.
Lu Y, Yang J, Sega E. Issues related to targeted delivery of proteins and peptides. AAPS J. 2006;8(3):E466–78.
Stolnik S, Shakesheff K. Formulations for delivery of therapeutic proteins. Biotechnol Lett. 2009;31(1):1–11.
Mundargi RC, Babu VR, Rangaswamy V, Patel P, Aminabhavi TM. Nano/micro technologies for delivering macromolecular therapeutics using poly (D, L-lactide-co-glycolide) and its derivatives. J Control Release. 2008;125(3):193–209.
Dong Y, Feng S-S. Poly (d, l-lactide-co-glycolide)/montmorillonite nanoparticles for oral delivery of anticancer drugs. Biomaterials. 2005;26(30):6068–76.
Blanco MD, Alonso MJ. Development and characterization of protein-loaded poly (lactide-co-glycolide) nanospheres. Eur J Pharm Biopharm. 1997;43(3):287–94.
Cun D, Foged C, Yang M, Frøkjær S, Nielsen HM. Preparation and characterization of poly (DL-lactide-co-glycolide) nanoparticles for siRNA delivery. Int J Pharm. 2010;390(1):70–5.
Tahara K, Yamamoto H, Hirashima N, Kawashima Y. Chitosan-modified poly (D, L-lactide-co-glycolide) nanospheres for improving siRNA delivery and gene-silencing effects. Eur J Pharm Biopharm. 2010;74(3):421–6.
Kumar MNVR, Bakowsky U, Lehr CM. Preparation and characterization of cationic PLGA nanospheres as DNA carriers. Biomaterials. 2004;25(10):1771–7.
Verma A, Stellacci F. Effect of surface properties on nanoparticle-cell interactions. Small. 2010;6(1):12–21.
Faraji AH, Wipf P. Nanoparticles in cellular drug delivery. Bioorg Med Chem. 2009;17(8):2950–62.
Labhasetwar V. Nanotechnology for drug and gene therapy: the importance of understanding molecular mechanisms of delivery. Curr Opin Biotechnol. 2005;16(6):674–80.
RaviKumar MNV, Mohapatra SS, Kong X, Jena PK, Bakowsky U, Lehrd CM. Cationic poly (lactide-co-glycolide) nanoparticles as efficient in vivo gene transfection agents. J Nanosci Nanotechnol. 2004;4(8):990–4.
Peniche C, Argüelles-Monal WW, Peniche H, Acosta N. Chitosan: an attractive biocompatible polymer for microencapsulation. Macromol Biosci. 2003;3(10):511–20.
Yang R, Shim W-S, Cui F-D, Cheng G, Han X, Jin Q-R, et al. Enhanced electrostatic interaction between chitosan-modified PLGA nanoparticle and tumor. Int J Pharm. 2009;371(1):142–7.
Mao S, Sun W, Kissel T. Chitosan-based formulations for delivery of DNA and siRNA. Adv Drug Deliv Rev. 2010;62(1):12–27.
Ragelle H, Vandermeulen G, Véronique P. Chitosan-based siRNA delivery systems. J Control Release. 2013;172(1):207–18.
Köping-Höggård M, Vårum KM, Issa M, Danielsen S, Christensen BE, Stokke BT, et al. Improved chitosan-mediated gene delivery based on easily dissociated chitosan polyplexes of highly defined chitosan oligomers. Gene Ther. 2004;11(19):1441–52.
Richardson SW, Kolbe HJ, Duncan R. Potential of low molecular mass chitosan as a DNA delivery system: biocompatibility, body distribution and ability to complex and protect DNA. Int J Pharm. 1999;178(2):231–43.
Hu F-Q, Liu L-N, Du Y-Z, Yuan H. Synthesis and antitumor activity of doxorubicin conjugated stearic acid-g-chitosan oligosaccharide polymeric micelles. Biomaterials. 2009;30(36):6955–63.
Huang X, Du Y-Z, Yuan H, Hu F-Q. Preparation and pharmacodynamics of low-molecular-weight chitosan nanoparticles containing insulin. Carbohydr Polym. 2009;76(3):368–73.
Biswas S, Chattopadhyay M, Sen KK, Saha MK. Development and characterization of alginate coated low molecular weight chitosan nanoparticles as new carriers for oral vaccine delivery in mice. Carbohydr Polym. 2015;121:403–10.
Hu F-Q, Zhao M-D, Yuan H, You J, Du Y-Z, Zeng S. A novel chitosan oligosaccharide-stearic acid micelles for gene delivery: Properties and in vitro transfection studies. Int J Pharm. 2006;315(1):158–66.
Amoozgar Z, Park J, Lin Q, Yeo Y. Low molecular-weight chitosan as a pH-sensitive stealth coating for tumor-specific drug delivery. Mol Pharm. 2012;9(5):1262–70.
Murata M, Nakano K, Tahara K, Tozuka Y, Takeuchi H. Pulmonary delivery of elcatonin using surface-modified liposomes to improve systemic absorption: polyvinyl alcohol with a hydrophobic anchor and chitosan oligosaccharide as effective surface modifiers. Eur J Pharm Biopharm. 2012;80(2):340–6.
Ying X-Y, Cui D, Yu L, Du Y-Z. Solid lipid nanoparticles modified with chitosan oligosaccharides for the controlled release of doxorubicin. Carbohydr Polym. 2011;84(4):1357–64.
Liu X, Huang H, Liu G, Zhou W, Chen Y, Jin Q, et al. Multidentate zwitterionic chitosan oligosaccharide modified gold nanoparticles: stability, biocompatibility and cell interactions. Nanoscale. 2013;5(9):3982–91.
Bae KH, Park M, Do MJ, Lee N, Ryu JH, Kim GW, et al. Chitosan oligosaccharide-stabilized ferrimagnetic iron oxide nanocubes for magnetically modulated cancer hyperthermia. ACS Nano. 2012;6(6):5266–73.
Shukla S, Jadaun A, Arora V, Sinha RK, Biyani N, Jain VK. In vitro toxicity assessment of chitosan oligosaccharide coated iron oxide nanoparticles. Toxic Rep. 2015;2:27–39.
Jain S, Datta M. Montmorillonite-PLGA nanocomposites as an oral extended drug delivery vehicle for venlafaxine hydrochloride. Appl Clay Sci. 2014;99:42–7.
Gan Q, Wang T. Chitosan nanoparticle as protein delivery carrier-systematic examination of fabrication conditions for efficient loading and release. Colloids Surf B: Biointerfaces. 2007;59(1):24–34.
Danhier F, Lecouturier N, Vroman B, Jérôme C, Marchand-Brynaert J, Feron O, et al. Paclitaxel-loaded PEGylated PLGA-based nanoparticles: in vitro and in vivo evaluation. J Control Release. 2009;133(1):11–7.
Wang G, Yu B, Wu Y, Huang B, Yuan Y, Liu CS. Controlled preparation and antitumor efficacy of vitamin E TPGS-functionalized PLGA nanoparticles for delivery of paclitaxel. Int J Pharm. 2013;446(1):24–33.
Md S, Khan RA, Mustafa G, Chuttani K, Baboota S, Sahni JK, et al. Bromocriptine loaded chitosan nanoparticles intended for direct nose to brain delivery: pharmacodynamic, pharmacokinetic and scintigraphy study in mice model. Eur J Pharm Sci. 2013;48(3):393–405.
Parveen S, Sahoo SK. Long circulating chitosan/PEG blended PLGA nanoparticle for tumor drug delivery. Eur J Pharmacol. 2011;670(2):372–83.
Philippova OE, Korchagina EV, Volkov EV, Smirnov VA, Khokhlov AR, Rinaudo M. Aggregation of some water-soluble derivatives of chitin in aqueous solutions: role of the degree of acetylation and effect of hydrogen bond breaker. Carbohydr Polym. 2012;87(1):687–94.
He C, Hu Y, Yin L, Tang C, Yin C. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials. 2010;31(13):3657–66.
Huang P, Li Z, Hu H, Cui D. Synthesis and characterization of bovine serum albumin-conjugated copper sulfide nanocomposites. J Nanomater. 2010;2010:33.
Kaewsaneha C, Opaprakasit P, Polpanich D, Smanmoo S, Tangboriboonrat P. Immobilization of fluorescein isothiocyanate on magnetic polymeric nanoparticle using chitosan as spacer. J Colloid Interface Sci. 2012;377(1):145–52.
Dandekar P, Jain R, Stauner T, Loretz B, Koch M, Wenz G, et al. A hydrophobic starch polymer for nanoparticle-mediated delivery of docetaxel. Macromol Biosci. 2012;12(2):184–94.
Paolicelli P, Prego C, Sanchez A, Alonso MJ. Surface-modified PLGA-based nanoparticles that can efficiently associate and deliver virus-like particles. Nanomedicine. 2010;5(6):843–53.
Cohen-Sela E, Chorny M, Koroukhov N, Danenberg HD, Golomb G. A new double emulsion solvent diffusion technique for encapsulating hydrophilic molecules in PLGA nanoparticles. J Control Release. 2009;133(2):90–5.
Fonte P, Soares S, Sousa F, Costa A, Seabra V, Reis S, et al. Stability study perspective of the effect of freeze-drying using cryoprotectants on the structure of insulin loaded into PLGA nanoparticles. Biomacromolecules. 2014;15(10):3753–65.
Abdelwahed W, Degobert G, Stainmesse S, Fessi H. Freeze-drying of nanoparticles: formulation, process and storage considerations. Adv Drug Deliv Rev. 2006;58(15):1688–713.
Sameti M, Bohr G, Kumar MNVR, Kneuer C, Bakowsky U, Nacken M, et al. Stabilisation by freeze-drying of cationically modified silica nanoparticles for gene delivery. Int J Pharm. 2003;266(1):51–60.
De Jaeghere F, Allémann E, Feijen J, Kissel T, Doelker E, Gurny R. Freeze-drying and lyopreservation of diblock and triblock poly (lactic acid)-poly (ethylene oxide)(PLA-PEO) copolymer nanoparticles. Pharm Dev Technol. 2000;5(4):473–83.
Holzer M, Vogel V, Mäntele W, Schwartz D, Haase W, Langer K. Physico-chemical characterisation of PLGA nanoparticles after freeze-drying and storage. Eur J Pharm Biopharm. 2009;72(2):428–37.
Prajakta D, Ratnesh J, Chandan K, Suresh S, Grace S, Meera V, et al. Curcumin loaded pH-sensitive nanoparticles for the treatment of colon cancer. J Biomed Nanotechnol. 2009;5(5):445–55.
Thasneem YM, Rekha MR, Sajeesh S, Sharma CP. Biomimetic mucin modified PLGA nanoparticles for enhanced blood compatibility. J Colloid Interface Sci. 2013;409:237–44.
Ma FK, Li J, Kong M, Liu Y, An Y, Chen XG. Preparation and hydrolytic erosion of differently structured PLGA nanoparticles with chitosan modification. Int J Biol Macromol. 2013;54:174–9.
Guo M, Rong W-T, Hou J, Wang D-F, Lu Y, Wang Y, et al. Mechanisms of chitosan-coated poly (lactic-co-glycolic acid) nanoparticles for improving oral absorption of 7-ethyl-10-hydroxycamptothecin. Nanotechnology. 2013;24(24):245101.
Pawar D, Mangal S, Goswami R, Jaganathan KS. Development and characterization of surface modified PLGA nanoparticles for nasal vaccine delivery: effect of mucoadhesive coating on antigen uptake and immune adjuvant activity. Eur J Pharm Biopharm. 2013;85(3):550–9.
Jagani HV, Josyula VR, Palanimuthu VR, Hariharapura RC, Gang SS. Improvement of therapeutic efficacy of PLGA nanoformulation of siRNA targeting anti-apoptotic Bcl-2 through chitosan coating. Eur J Pharm Sci. 2013;48(4):611–8.
Slütter B, Bal S, Keijzer C, Mallants R, Hagenaars N, Que I, et al. Nasal vaccination with N-trimethyl chitosan and PLGA based nanoparticles: nanoparticle characteristics determine quality and strength of the antibody response in mice against the encapsulated antigen. Vaccine. 2010;28(38):6282–91.
He P, Davis SS, Illum L. In vitro evaluation of the mucoadhesive properties of chitosan microspheres. Int J Pharm. 1998;166(1):75–88.
Burton JD. The MTT assay to evaluate chemosensitivity. Chemosensitivity. Springer. 2005. p. 69–78.
Grabowski N, Hillaireau H, Vergnaud J, Santiago LA, Kerdine-Romer S, Pallardy M, et al. Toxicity of surface-modified PLGA nanoparticles toward lung alveolar epithelial cells. Int J Pharm. 2013;454(2):686–94.
Tahara K, Sakai T, Yamamoto H, Takeuchi H, Hirashima N, Kawashima Y. Improved cellular uptake of chitosan-modified PLGA nanospheres by A549 cells. Int J Pharm. 2009;382(1):198–204.
Foster S, Duvall CL, Crownover EF, Hoffman AS, Stayton PS. Intracellular delivery of a protein antigen with an endosomal-releasing polymer enhances CD8 T-cell production and prophylactic vaccine efficacy. Bioconjug Chem. 2010;21(12):2205–12.
Acknowledgments
Mr. Sathish Dyawanapelly would like to thank Department of Biotechnology (BT/PR5372/MED/29/489/2012), Govt. of India for the fellowship. Dr. Prajakta Dandekar is thankful to Ramanujan Fellowship Grant (SR/S2/RJN-139/2011), DST, Govt. of India. Dr. Ratnesh Jain is thankful to Ramalingaswami Fellowship (BT/RLF/RE-ENTRY/51/2011), DBT, Govt. of India. The authors are grateful for the technical assistance of Maruthi Prasanna, Nishant Jain and Kireeti Kumar Kota. PURAC Biopolymers, Netherlands and Evonik, India is acknowledged for the generous gift of PLGA. Authors are thankful to DST Nanomission (SR/NM/NS 1145/2012) for Imaging Flow Cytometer facility. Dr. Ratnesh Jain is thankful to DST-FIST (SR/FST/ETII-058/2013) for Confocal Microscopy facility.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Statement of human and animal rights
‘No animal or human studies were carried out by the authors for this article.’
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 280 kb)
Rights and permissions
About this article
Cite this article
Dyawanapelly, S., Koli, U., Dharamdasani, V. et al. Improved mucoadhesion and cell uptake of chitosan and chitosan oligosaccharide surface-modified polymer nanoparticles for mucosal delivery of proteins. Drug Deliv. and Transl. Res. 6, 365–379 (2016). https://doi.org/10.1007/s13346-016-0295-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-016-0295-x