Abstract
Background
p19arf, primarily known as a tumor suppressor, has also been reported to play an essential role in normal development of mouse eyes. Consistently, lack of p19arf has been associated with ocular defects, but the mixed background of the knockout (KO) mouse strain used raised a concern on the accuracy of the phenotypes observed in association with the targeted gene due to genetic heterogeneity.
Object
We carried out a study to investigate into the effect of genetic background on the manifestation of p19arf KO associated phenotypes.
Methods
We characterized the phenotypes of novel p19arf KO mouse lines generated in FVB/N and C57BL/6J using a transcription activator-like effector nuclease (TALEN) system in comparison to the reported phenotypes of three other p19arf-deficient mouse lines generated using homologous recombination.
Results
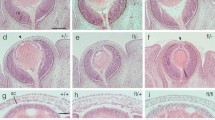
Ninety-five percent of FVB/N-p19arf KO mice showed ocular opacity from week 4 after birth which worsened rapidly until week 6, while such abnormality was absent in C57BL/6J-p19arf KO mice up to the age of 26 weeks. Histopathological analysis revealed retrolental masses and dysplasia in the retinal layer in FVB/N-p19arf KO mice from week 4. Besides these, both strains developed normally from birth to week 26 without increased tumorigenesis except for a subcutaneous tumor found in a C57BL/6J-p19arf KO mouse.
Conclusion
Our findings demonstrated surprisingly variable manifestation of p19arf-linked phenotypes between FVB/N and C57BL/6J mice, and furthermore between our mouse lines and the established lines, indicating a critical impact of genetic background on functional study of genes using gene targeting strategies in mice.






Similar content being viewed by others
References
Cheong C, Sung YH, Lee J, Choi YS, Song J, Kee C, Lee H-W (2006) Role of INK4a locus in normal eye development and cataract genesis. Mech Ageing Dev 127:633–638
Dalke C et al (2004) Electroretinography as a screening method for mutations causing retinal dysfunction in mice. Investig Ophthalmol Vis Sci 45:601–609
Doetschman T (2009) Influence of genetic background on genetically engineered mouse phenotypes. Methods Mol Biol (Clifton, NJ) 530:423–433
Gaj T, Gersbach CA, Barbas CF III (2013) ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 31:397–405
Hendrich HJ (2012) The laboratory mouse, 2nd edn. Elsevier, Italy
Holzenberger M, Leneuve P, Hamard G, Ducos B, Perin L, Binoux M, Le Bouc Y (2000) A targeted partial invalidation of the insulin-like growth factor I receptor gene in mice causes a postnatal growth deficit. Endocrinology 141:2557–2566
Iqbal NS, Devitt CC, Sung CY, Skapek SX (2016) p19Arf limits primary vitreous cell proliferation driven by PDGF-B. Exp Eye Res 145:224–229
Kamijo T et al (1997) Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19 ARF. Cell 91:649–659
Kamijo T, Weber JD, Zambetti G, Zindy F, Roussel MF, Sherr CJ (1998) Functional and physical interactions of the ARF tumor suppressor with p53 and Mdm2. Proc Natl Acad Sci 95:8292–8297
Keane TM et al (2011) Mouse genomic variation and its effect on phenotypes and gene regulation. Nature 477:289–294
Kim J-I et al (2018) CRISPR/Cas9-mediated knockout of Rag-2 causes systemic lymphopenia with hypoplastic lymphoid organs in FVB mice. Lab Anim Res 34:166–175
Kim T-H, Son T, Le D, Yao X (2019) Longitudinal OCT and OCTA monitoring reveals accelerated regression of hyaloid vessels in retinal degeneration 10 (rd10) mice. Sci Rep 9:1–9
Lee JH et al (2020) The position of the target site for engineered nucleases improves the aberrant mRNA clearance in in vivo genome editing. Sci Rep 10:4173
Lilue J et al (2018) Sixteen diverse laboratory mouse reference genomes define strain-specific haplotypes and novel functional loci. Nat Genet 50:1574–1583
Martin AC et al (2004) Pathogenesis of persistent hyperplastic primary vitreous in mice lacking the arf tumor suppressor gene. Invest Ophthalmol Vis Sci 45:3387–3396
McKeller RN, Fowler JL, Cunningham JJ, Warner N, Smeyne RJ, Zindy F, Skapek SX (2002) The Arf tumor suppressor gene promotes hyaloid vascular regression during mouse eye development. Proc Natl Acad Sci 99:3848–3853
Montana CL, Kolesnikov AV, Shen SQ, Myers CA, Kefalov VJ, Corbo JC (2013) Reprogramming of adult rod photoreceptors prevents retinal degeneration. Proc Natl Acad Sci 110:1732–1737
NRC (2010) Guide for the care and use of laboratory animals, 8th edn. National Academies Press, New York
Park BM, Roh J-I, Lee J, Lee H-W (2018) Generation of knockout mouse models of cyclin-dependent kinase inhibitors by engineered nuclease-mediated genome editing. Lab Anim Res 34:264–269
Scacheri P et al (2001) Bidirectional transcriptional activity of PGK-neomycin and unexpected embryonic lethality in heterozygote chimeric knockout mice. Genesis 30:259–263
Serrano M, Lee H-W, Chin L, Cordon-Cardo C, Beach D, DePinho RA (1996) Role of the INK4a locus in tumor suppression and cell mortality. Cell 85:27–37
Tong C, Huang G, Ashton C, Li P, Ying Q-L (2011) Generating gene knockout rats by homologous recombination in embryonic stem cells. Nat Protoc 6:827
Valera A, Perales JC, Hatzoglou M, Bosch F (1994) Expression of the neomycin-resistance (neo) gene induces alterations in gene expression and metabolism. Hum Gene Ther 5:449–456
Weber JD et al (2000) p53-independent functions of the p19ARF tumor suppressor. Genes Dev 14:2358–2365
Wong AA, Brown RE (2006) Visual detection, pattern discrimination and visual acuity in 14 strains of mice. Genes Brain Behav 5:389–403
Yang J, Nan C, Ripps H, Shen W (2015) Destructive changes in the neuronal structure of the FVB/N mouse retina. PLoS One 10:e0129719
Yeo JH et al (2019) Development of a Pde6b gene knockout rat model for studies of degenerative retinal diseases. Investig Ophthalmol Vis Sci 60:1519–1526
Acknowledgements
This research was supported by a grant (14182MFDS978) from Ministry of Food and Drug Safety in 2015.
Author information
Authors and Affiliations
Contributions
BCK, HWL and KT contributed to the study conception and design. Material preparation, data collection and analysis were performed by JSP, JIK, HJL, SR, EK and KMH. The first draft of the manuscript was written by JSP and JIK and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All experiments were approved by the Institutional Animal Care and Use Committee (IACUC) in Seoul National University Hospital (Protocol number: 15-0031-C2A0(1)) and animals were maintained and used in accordance with Guide for the Care and Use of Laboratory Animals (NR 2010).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, JS., Kim, JI., Lim, HJ. et al. Differential manifestation of ocular phenotypes in TALEN-mediated p19arf knockout FVB/N and C57BL/6J mouse lines. Genes Genom 42, 1023–1033 (2020). https://doi.org/10.1007/s13258-020-00959-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-020-00959-z