Abstract
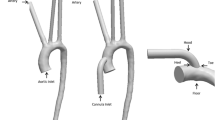
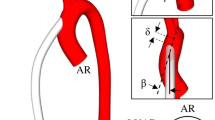
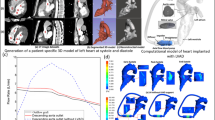
Although there are many studies that focus on understanding the consequence of pumping mode (continuous vs. pulsatile) associated with ventricular assist devices (VADs) on pediatric vascular pulsatility, the impact on local hemodynamics has been largely ignored. Hence, we compare not only the hemodynamic parameters indicative of pulsatility but also the local flow fields in the aorta and the great vessels originating from the aortic arch. A physiologic graft anastomotic model is constructed based on a pediatric, patient specific, aorta with a graft attached on the ascending aorta. The flow is simulated using a previously validated second-order accurate Navier–Stokes flow solver based upon a finite volume approach. The major findings are: (1) pulsatile support provides a greater degree of vascular pulsatility when compared to continuous support, which, however, is still 20% less than pulsatility in the healthy aorta; (2) pulsatile support increases the flow in the great vessels, while continuous support decreases it; (3) complete VAD support results in turbulence in the aorta, with maximum principal Reynolds stresses for pulsatile support and continuous support of 7081 and 249 dyn/cm2, respectively; (4) complete pulsatile support results in a significant increase in predicted hemolysis in the aorta; and (5) pulsatile support causes both higher time-averaged wall shear stresses (WSS) and oscillatory shear indices (OSI) in the aorta than does continuous support. These findings will help to identify the risk of graft failure for pediatric patients with pulsatile and continuous VADs.











Similar content being viewed by others
References
Moller, J. H., ed. Prevalence and incidence of cardiac malformations. In: Surgery of Congenital Heart Disease: Pediatric Cardiac Care Consortium 1984–1995, vol. 6. Armonk, NY: Futura Publishing Company, 1998, p. 20.
Baldwin, J. T., H. S. Borovetz, B. W. Duncan, M. J. Gartner, R. K. Jarvik, W. J. Weiss, and T. R. Hoke. The National Heart, Lung, and Blood Institute pediatric circulatory support program. Circulation 113:147–155, 2006.
Connell, J. M., T. Khalapyan, J. L. Myers, G. Rosenberg, and W. J. Weiss. Anatomic fit assessment for the Penn state pediatric ventricular assist device. ASAIO J. 53:687–691, 2007.
Dellgren, G., and J. G. Coles. Pediatric heart transplantation: improving results in high-risk patients. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. Annu. 4:103–114, 2001.
Driscoll, R. J., and L. A. Kennedy. A model for the turbulent energy spectrum. Phys. Fluids 26:1228–1233, 1983.
Farinas, M. I., A. Garon, D. Lacasse, and D. N’dri. Asymptotically consistent numerical approximation of hemolysis. J. Biomech. Eng. 128:688–696, 2006.
Figueroa, C. A., I. E. Vignon-Clementel, K. E. Jansen, T. J. R. Hughes, and C. A. Taylor. A coupled momentum method for modeling blood flow in three-dimensional deformable arteries. Comput. Methods Appl. Mech. Eng. 195:5685–5706, 2006.
Fogel, M. A., P. M. Weinberg, J. Rychik, A. Hubbard, M. Jacobs, T. L. Spray, and J. Haselgrove. Caval contribution to flow in the branch pulmonary arteries of Fontan patients with a novel application of magnetic resonance presaturation pulse. Circulation 99:1215–1221, 1999.
Fureby, C., and F. F. Grinstein. Large eddy simulation of high-Reynolds-number free and wall-bounded flows. J. Comp. Phys. 181:68–97, 2002.
Fureby, C., G. Tabor, H. G. Weller, and A. D. Gosman. A comparative study of subgrid scale models in homogeneous isotropic turbulence. Phys. Fluids 9:1416–1429, 1997.
Garon, A., and M. I. Farinas. Fast three-dimensional numerical hemolysis approximation. Artif. Organs 28:1016–1025, 2004.
Giersiepen, M., L. J. Wurzinger, R. Opitz, and H. Reul. Estimation of shear stress-related blood damage in heart valve prostheses—in vitro comparison of 25 aortic valves. Int. J. Artif. Organs 13:300–306, 1990.
Grinstein, F. F., and C. Fureby. On flux-limiting-based implicit large eddy simulation. J. Fluid Eng. 129:1483–1492, 2007.
Grinstein, F. F., C. Fureby, and C. R. DeVore. On MILES based on flux-limiting algorithm. Int. J. Numer. Methods Fluids 47:1043–1051, 2005.
Healy, T. M., J. T. Ellis, A. A. Fontaine, C. A. Jarrett, and A. P. Yoganathan. An automated method for analysis and visualization of laser Doppler velocimetry data. Ann. Biomed. Eng. 25:335–343, 1997.
Heiss, K. F., R. H. Clark, J. D. Cornish, M. Stovroff, R. Ricketts, K. Kesser, and M. Stonecash. Preferential use of venovenous extracorporeal membrane oxygenation for congenital diaphragmatic hernia. J. Pediatr. Surg. 30:416–419, 1995.
Higdon, A., E. Ohlsen, and W. B. Stiles. Mechanics of Material. New York: John Wiley & Sons, 1988.
Huo, Y., X. Guo, and G. S. Kassab. The flow field along the entire length of mouse aorta and primary branches. Ann. Biomed. Eng. 36:685–699, 2008.
Issa, R. I. Solution of the implicitly discretised fluid flow equations by operator-splitting. J. Comput. Phys. 62:40–65, 1985.
Kent, A. L., Z. Kecskes, B. Shadbolt, and M. C. Falk. Blood pressure in the first year of life in healthy infants born at term. Pediatr. Nephrol. 22:1743–1749, 2007.
Lacasse, D., A. Garon, and D. Pelletier. Development of an adaptive discontinuous-Galerkin finite element method for advection–reaction equations. Comput. Methods Appl. Mech. Eng. 196:2071–2083, 2007.
Loth, F., P. F. Fischer, and H. S. Bassiouny. Blood flow in end-to-side anastomosis. Annu. Rev. Fluid Mech. 40:367–393, 2008.
Machii, M., and A. E. Becker. Morphologic features of the normal aortic arch in neonates, infants, and children pertinent to growth. Ann. Thorac. Surg. 64:511–515, 1997.
May-Newman, K., B. Hillen, and W. Dembitsky. Effect of left ventricular assist device outflow conduit anastomosis location on flow patterns in the native aorta. ASAIO J. 52:132–139, 2006.
Minakawa, M., I. Fukuda, J. Yamazaki, K. Fukui, H. Yanaoka, and T. Inamura. Effect of cannula shape on aortic wall and flow turbulence: hydrodynamic study during extracorporeal circulation in mock thoracic aorta. Artif. Organs 31:880–886, 2007.
Nose, Y. Nonpulsatile mode of blood flow required for cardiopulmonary bypass and total body perfusion. Artif. Organs 17:92–102, 1993.
Ojha, M. Spatial and temporal variations of wall shear stress within an end-to-side arterial anastomosis model. J. Biomech. 26:1377–1388, 1993.
OpenFOAM 1.5 User Guide. UK: OpenCFD Ltd, 2008.
Pantalos, G. M., G. Giridharan, J. Colyer, M. Mitchell, J. Speakman, C. Lucci, G. Johnson, M. Gartner, and S. C. Koenig. Effect of continuous and pulsatile flow left ventricular assist on pulsatility in a pediatric animal model of left ventricular dysfunction: pilot observations. ASAIO J. 53:385–391, 2007.
Pekkan, K., O. Dur, K. Sundareswaran, K. Kanter, M. Fogel, A. Yoganathan, and A. Undar. Neonatal aortic arch hemodynamics and perfusion during cardiopulmonary bypass. J. Biomech. Eng. 130:061012, 2008.
Sallam, A. M., and N. H. Hwang. Human red blood cell hemolysis in a turbulent shear flow: contribution of Reynolds shear stresses. Biorheology 21:783–797, 1984.
Taylor, K. M., W. H. Bain, K. J. Maxted, M. M. Hutton, W. Y. McNab, and P. K. Caves. Comparative studies of pulsatile and nonpulsatile flow during cardiopulmonary bypass I. Pulsatile system employed and its hematologic effects. J. Thorac. Cardiovasc. Surg. 75:569–573, 1978.
Taylor, G. A., G. R. Martin, and B. L. Short. Cardiac determinants of cerebral blood flow during extracorporeal membrane oxygenation. Invest. Radiol. 24:511–516, 1989.
Taylor, K. M., G. S. Wright, W. H. Bain, P. K. Caves, and G. S. Beastall. Comparative studies of pulsatile and nonpulsatile flow during cardiopulmonary bypass III. Response of anterior pituitary gland to thyrotropin-releasing hormone. J. Thorac. Cardiovasc. Surg. 75:579–584, 1978.
Taylor, K. M., G. S. Wright, J. M. Reid, W. H. Bain, P. K. Caves, M. S. Walker, and J. K. Grant. Comparative studies of pulsatile and nonpulsatile flow during cardiopulmonary bypass II. The effects on adrenal secretion of cortisol. J. Thorac. Cardiovasc. Surg. 75:574–578, 1978.
Thom, T., N. Haase, W. Rosamond, V. J. Howard, J. Rumsfeld, T. Manolio, Z. J. Zheng, K. Flegal, C. O’Donnell, S. Kittner, D. Lloyd-Jones, D. C. Goff, Jr, Y. Hong, R. Adams, G. Friday, K. Furie, P. Gorelick, B. Kissela, J. Marler, J. Meigs, V. Roger, S. Sidney, P. Sorlie, J. Steinberger, S. Wasserthiel-Smoller, M. Wilson, P. Wolf, and American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics–2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 113:e85–e151, 2006.
Travis, A. R., G. A. Giridharan, G. M. Pantalos, R. D. Dowling, S. D. Prabhu, M. S. Slaughter, M. Sobieski, A. Undar, D. J. Farrar, and S. C. Koenig. Vascular pulsatility in patients with a pulsatile- or continuous-flow ventricular assist device. J. Thorac. Cardiovasc. Surg. 133:517–524, 2007.
Undar, A., T. Masai, O. H. Frazier, and C. D. Fraser, Jr. Pulsatile and nonpulsatile flows can be quantified in terms of energy equivalent pressure during cardiopulmonary bypass for direct comparisons. ASAIO J. 45:610–614, 1999.
Undar, A., T. Masai, S. Q. Yang, J. Goddard-Finegold, O. H. Frazier, and C. D. Fraser, Jr. Effects of perfusion mode on regional and global organ blood flow in a neonatal piglet model. Ann. Thorac. Surg. 68:1336–1342, 1999.
Vignon-Clementel, I. E., C. A. Figueroa, K. E. Jansen, and C. A. Taylor. Outflow boundary conditions for three-dimensional finite element modeling of blood flow and pressure in arteries. Comput. Methods Appl. Mech. Eng. 195:3776–3796, 2006.
Yang, N., S. Deutsch, E. G. Paterson, and K. B. Manning. Hemodynamics of a end-to-side anastomotic graft for a pulsatile pediatric ventricular assist device. J. Biomech. Eng., in press.
Yang, N., S. Deutsch, E. G. Paterson, and K. B. Manning. Numerical study of blood flow at the end-to-side anastomosis of a left ventricular assist device for adult patient. J. Biomech. Eng., in press.
Yano, T., K. Sekine, A. Mitoh, Y. Mitamura, E. Okamoto, D. W. Kim, I. Nishimura, S. Murabayashi, and R. Yozu. An estimation method of hemolysis within an axial flow blood pump by computational fluid dynamics analysis. Artif. Organs 27:920–925, 2003.
Acknowledgments
This work was supported by NIH NHLBI HV 48191. The authors would like to thank Jason Holmes and Jeff Nucciarone of the High Performance Computing Group at the Pennsylvania State University for their technical support with PC clusters. The authors would also like to thank Dr. Ajit Yoganathan and Mr. Chris Haggerty from the Department of Biomedical Engineering at the Georgia Institute of Technology for providing the pediatric aortic model.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, N., Deutsch, S., Paterson, E.G. et al. Comparative Study of Continuous and Pulsatile Left Ventricular Assist Devices on Hemodynamics of a Pediatric End-to-Side Anastomotic Graft. Cardiovasc Eng Tech 1, 88–103 (2010). https://doi.org/10.1007/s13239-010-0006-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-010-0006-6