Abstract
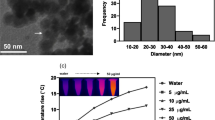
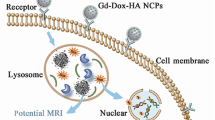
Although chemotherapy is mostly performed by direct injection of the chemotherapeutic agents systemically, this approach can cause undesirable side effects in normal tissues and lacks targeting efficiency. In this study, we have developed a micron-sized, bead-type multifunctional anticancer-drug carrier that can be injected in the vicinity of a lesion using a syringe. The multifunctional anticancer-drug carrier bead was fabricated by incorporating functional nanomaterials, such as near-infrared (NIR)-responsive gold nanorod (GNR) and superparamagnetic iron oxide nanoparticles (IONP) that work as a magnetic resonance imaging (MRI) contrast agent, into an alginate hydrogel bead. The carrier bead containing GNR and IONP was spherical, with an average size of 362.2 ± 22.7 µm. These multifunctional anticancer-drug carrier beads could successfully release doxorubicin (Dox) into the external environment upon irradiation with an NIR laser. The laser responsive on-demand release profile demonstrated well-controlled and sustained release of Dox, and the effectiveness of this drug delivery system can be confirmed in vitro by the 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide assay. Additionally, the control experiments evidenced that the thermal effect of GNR was insignificant and the cytotoxic action of Dox was expressed only after irradiation with the NIR laser. The multifunctional anticancer-drug carrier beads can be precisely monitored with the MRI T2 imaging mode.



Similar content being viewed by others
References
Zhang, L., Abdullah, R., Hu, X., Bai, H., Fan, H., He, L., Liang, H., Zou, J., Liu, Y., Sun, Y., Zhang, X. & Tan, W. Engineering of Bioinspired, Size-Controllable, Self-Degradable Cancer-Targeting DNA Nanoflowers via the Incorporation of an Artificial Sandwich Base. J. Am. Chem. Soc. 141, 4282–4290 (2019).
Park, H., Kim, J., Jung, S. & Kim, W.J. DNA-Au Nanomachine Equipped with I-Motif and G-Quadruplex for Triple Combinatorial Anti-Tumor Therapy. Adv. Funct. Mater. 28, 1705416 (2017).
Cheng, W., Liang, C., Xu, L., Liu, G., Gao, N., Tao, W., Luo, L., Zuo, Y., Wang, X., Zhang, X., Zeng, X. & Mei, L. TPGS-Functionalized Polydopamine-Modified Mesoporous Silica as Drug Nanocarriers for Enhanced Lung Cancer Chemotherapy Against Multidrug Resistance. Small 13, 1700623 (2017).
Lin, L.S., Yang, X., Zhou, Z., Yang, Z., Jacobson, O., Liu, Y., Yang, A., Niu, G., Song, J., Yang, H.H. & Chen, X. Yolk-Shell Nanostructure: an Ideal Architecture to Achieve Harmonious Integration of Magnetic-Plasmonic Hybrid Theranostic Platform. Adv. Mater. 29, 1606681 (2017).
Kearney, C.J. & Mooney, D.J. Macroscale Delivery Systems for Molecular and Cellular Payloads. Nat. Mater. 12, 1004–1017 (2013).
Bencherif, S.A., Sands, R.W., Bhatta, D., Arany, P., Verbeke, C.S., Edwards, D.A. & Mooney, D.J. Injectable Preformed Scaffolds with Shape-Memory Properties. Proc. Natl. Acad. Sci. U.S.A 109, 19590–19595 (2012).
Shin, D.S., You, J., Rahimian, A., Vu, T., Siltanen, C., Ehsanipour, A., Stybayeva, G., Sutcliffe, J. & Revzin, A. Photodegradable Hydrogels for Capture, Detection, and Release of Live Cells. Angew. Chem. Int. Ed. 53, 8221–8224 (2014).
Kim, S.H., Lee, B., Heo, J.H., Lee, K.E., Shankar, P., Han, K.H. & Lee, J.H. The Effect of ζ-Potential and Hydrodynamic Size on Nanoparticle Interactions in Hydrogels. Part. Part. Syst. Charact. 36, 1800292 (2019).
Delplace, V., Obermeyer, J. & Shoichet, M. S. Local Affinity Release. ACS Nano 10, 6433–6436 (2016).
Mura, S., Nicolas, J. & Couvreur, P. Stimuli-Responsive Nanocarriers for Drug Delivery. Nat. Mater. 12, 991–1003 (2013).
Ling, D., Xia, H., Park, W., Hackett, M.J., Song, C., Na, K., Hui, K.M. & Hyeon, T. pH-Sensitive Nanoformulated Triptolide as a Targeted Therapeutic Strategy for Hepatocellular Carcinoma. ACS Nano 8, 8027–8039 (2014).
Zhao, X., Kim, J., Cezar, C.A., Huebsch, N., Lee, K., Bouhadir, K. & Mooney, D.J. Active Scaffolds for on-Demand Drug and Cell Delivery. Proc. Natl. Acad. Sci. U.S.A. 108, 67–72 (2011).
Dam, D.H. M., Lee, J.H., Sisco, P.N., Co, D.T., Zhang, M., Wasielewski, M.R. & Odom, T.W. Direct Observation of Nanoparticle-Cancer Cell Nucleus Interactions. ACS Nano 6, 3318–3326 (2012).
Noh, J.-Y., Kim, J.-I., Chang, Y. W., Park, J.-M., Song, H.-W., Kang, M.-J. & Pyun, J.-C. Gold Nanoislands Chip for Laser Desorption/Ionization (LDI) Mass Spectrometry. BioChip J. 11, 246–254 (2017).
Jung, H., Jung, J., Kim, Y.-H., Kwon, D., Kim, B. -G., Na, H.B. & Lee, H.H. Surface Plasmon Resonance Characteristics of Au Nanoparticles Layered Sensor Chip for Direct Detection of Stress Hormone Conjugated by Nanoparticles. BioChip J. 12, 249–256 (2018).
Yoon, S., Lee, B., Kim, C. & Lee, J.H. Controlled Heterogeneous Nucleation for Synthesis of Uniform Mesoporous Silica-Coated Gold Nanorods with Tailorable Rotational Diffusion and 1 nm-Scale Size Tunability. Cryst. Growth Des. 18, 4731–4736 (2018).
Lee, J.W., Jung, H., Cho, H.H., Lee, J.H. & Nam, Y. Gold Nanostar-Mediated Neural Activity Control Using Plasmonic Photothermal Effects. Biomaterials 153, 59–69 (2018).
Wei, H., Bruns, O.T., Kaul, M.G., Hansen, E.C., Barch, M., Wiśniowska, A., Chen, O., Chen, Y., Li, N., Okada, S., Cordero, J.M., Heine, M., Farrar, C.T., Montana, D.M., Adam, G., Ittrich, H., Jasanoff, A., Nielsen, P. & Bawendi, M.G. Exceedingly Small Iron Oxide Nanoparticles as Positive MRI Contrast Agents. Proc. Natl. Acad. Sci. U.S.A. 114, 2325–2330 (2017).
Yigit, M.V., Mazumdar, D., Kim, H.K., Lee, J.H., Odintsov, B. & Lu, Y. Smart “Turn-on” Magnetic Resonance Contrast Agents Based on Aptamer-Functionalized Superparamagnetic Iron Oxide Nanoparticles. ChemBioChem 8, 1675–1678 (2007).
Yoon, S., Lee, B., Yun, J., Han, J.G., Lee, J.S. & Lee, J.H. Systematic Study of Interdependent Relationship on Gold Nanorod Synthesis Assisted by Electron Microscopy Image Analysis. Nanoscale 9, 7114–7123 (2017).
Zhang, Z. & Lin, M. Fast Loading of PEG-SH on CTAB-Protected Gold Nanorods. RSC Adv. 34, 17760–17767 (2014).
Domenech, M., Marrero-Berrios, I., Torres-Lugo, M. & Rinaldi, C. Lysosomal Membrane Permeabilization by Targeted Magnetic Nanoparticles in Alternating Magnetic Fields. ACS Nano 7, 5091–5101 (2013).
Li, J. & Mooney, D.J. Designing Hydrogels for Controlled Drug Delivery. Nat. Rev. Mater. 1, 16071 (2016).
Park, C., Kong, M., Lee, J.-H., Ryu, S. & Park, S. Detection of Bacillus Cereus Using Bioluminescence Assay with Cell Wall-Binding Domain Conjugated Magnetic Nanoparticles. BioChip J. 12, 287–293 (2018).
Acknowledgements
This research was supported by GRRC program of Gyeonggi Province (Grant No. GRRC Sungkyunkwan 2017-A01). S. Moon and Y Park are supported by Gyeonggi Science High School R&E fund. K.H.H. appreciate the support from the Research Fellowship Program (NRF-2018R1A6A3A 11047269). The manuscript was written through the contributions of all authors.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of Interests
The authors declare no competing financial interests.
These authors contrilbuted equally.
Rights and permissions
About this article
Cite this article
Yoon, S., Chang, J., Kwon, N. et al. Multifunctional Nanomaterial-alginate Drug Delivery and Imaging System for Cancer Therapy. BioChip J 13, 236–242 (2019). https://doi.org/10.1007/s13206-019-3309-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-019-3309-1