Abstract
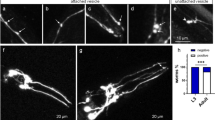
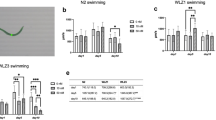
In a previous study, we have shown that methylmercury (MeHg) exposure causes focal aggregation of intracellular transgenic mCherry protein in dendrites of cephalic (CEP) neurons in Caenorhabditis elegans (C. elegans). However, the underlying mechanism is unknown. We hypothesized that reduced cellular release of mCherry via extracellular vesicles by MeHg contributes to its accumulation and intracellular aggregation. Thus, we characterized vesicular structures in CEP dendrites, which were 1–3 μm in diameter and could readily bud off from the plasma membrane of the dendrites. Chronic treatment of C. elegans with MeHg (5 μM, 4–10 days) reduced the number of vesicles attached to CEP dendrites (attached vesicles) and vesicles unattached to CEP dendrites (unattached vesicles), as well as the presence of extracellular mCherry, supporting the hypothesis that release of mCherry by microvesicle formation is inhibited by MeHg. Leucine-rich repeat kinase 2 (LRRK2) has an important function in membrane biology. Further investigation showed that the effects of MeHg were modified by human LRRK2. In worms with the wild-type LRRK2, the vesicle numbers were significantly reduced by MeHg (0.5 and 5 μM). The effects of MeHg on the presence of extracellular mCherry and attached vesicles were modified by the human wild-type LRRK2. Independent of MeHg treatment, the G2019S mutant LRRK2 showed reduced number of unattached vesicles; however, the levels of extracellular mCherry were increased. Knockdown of C. elegans irk-1, the homolog of human LRRK2, reduced the number of attached vesicles, corroborating that LRRK2 plays an important role in the formation of microvesicles.










Similar content being viewed by others
References
Albanese F, Novello S, Morari M (2019) Autophagy and LRRK2 in the aging brain. Front Neurosci 13:1352. https://doi.org/10.3389/fnins.2019.01352
Al-Nedawi K, Meehan B, Rak J (2009) Microvesicles: messengers and mediators of tumor progression. Cell Cycle 8:2014–2018. https://doi.org/10.4161/cc.8.13.8988
Apfeld J, Kenyon C (1998) Cell nonautonomy of C. elegans daf-2 function in the regulation of diapause and life span. Cell 95:199–210. https://doi.org/10.1016/s0092-8674(00)81751-1
Berwick DC, Heaton GR, Azeggagh S, Harvey K (2019) LRRK2 biology from structure to dysfunction: research progresses, but the themes remain the same. Mol Neurodegener 14:49. https://doi.org/10.1186/s13024-019-0344-2
Bodega G, Alique M, Puebla L, Carracedo J, Ramirez RM (2019) Microvesicles: ROS scavengers and ROS producers. J Extracellular Vesicles 8:1626654. https://doi.org/10.1080/20013078.2019.1626654
Boulin T, Etchberger JF, Hobert O (2006) Reporter gene fusions. WormBook:1–23. https://doi.org/10.1895/wormbook.1.106.1
Calixto A, Chelur D, Topalidou I, Chen X, Chalfie M (2010) Enhanced neuronal RNAi in C. elegans using SID-1. Nat Methods 7:554–559. https://doi.org/10.1038/nmeth.1463
di Domenico A, Carola G, Calatayud C, Pons-Espinal M, Munoz JP et al (2019) Patient-specific iPSC-derived astrocytes contribute to non-cell-autonomous neurodegeneration in Parkinson's disease. Stem Cell Rep 12:213–229. https://doi.org/10.1016/j.stemcr.2018.12.011
Doroquez DB, Berciu C, Anderson JR, Sengupta P, Nicastro D (2014) A high-resolution morphological and ultrastructural map of anterior sensory cilia and glia in Caenorhabditis elegans. Elife 3:e01948. https://doi.org/10.7554/eLife.01948
Emmanouilidou E, Melachroinou K, Roumeliotis T, Garbis SD, Ntzouni M et al (2010) Cell-produced alpha-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J Neurosci 30:6838–6851. https://doi.org/10.1523/jneurosci.5699-09.2010
Farina M, Aschner M, Rocha JB (2011) Oxidative stress in MeHg-induced neurotoxicity. Toxicol Appl Pharmacol 256:405–417. https://doi.org/10.1016/j.taap.2011.05.001
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE et al (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391:806–811. https://doi.org/10.1038/35888
Greggio E, Jain S, Kingsbury A, Bandopadhyay R, Lewis P et al (2006) Kinase activity is required for the toxic effects of mutant LRRK2/dardarin. Neurobiol Dis 23:329–341. https://doi.org/10.1016/j.nbd.2006.04.001
Hall DH, Hedgecock EM (1991) Kinesin-related gene unc-104 is required for axonal transport of synaptic vesicles in C. elegans. Cell 65:837–847. https://doi.org/10.1016/0092-8674(91)90391-b
Harischandra DS, Rokad D, Neal ML, Ghaisas S, Manne S, Sarkar S, Panicker N, Zenitsky G, Jin H, Lewis M, Huang X, Anantharam V, Kanthasamy A, Kanthasamy AG (2019) Manganese promotes the aggregation and prion-like cell-to-cell exosomal transmission of alpha-synuclein. Sci Signal 12. https://doi.org/10.1126/scisignal.aau4543
Howlett EH, Jensen N, Belmonte F, Zafar F, Hu X et al (2017) LRRK2 G2019S-induced mitochondrial DNA damage is LRRK2 kinase dependent and inhibition restores mtDNA integrity in Parkinson's disease. Hum Mol Genet 26:4340–4351. https://doi.org/10.1093/hmg/ddx320
Kastelowitz N, Yin H (2014) Exosomes and microvesicles: identification and targeting by particle size and lipid chemical probes. Chembiochem 15:923–928. https://doi.org/10.1002/cbic.201400043
Ke T, Tsatsakis A, Santamaria A, Antunes Soare FA, Tinkov AA et al (2020) Chronic exposure to methylmercury induces puncta formation in cephalic dopaminergic neurons in Caenorhabditis elegans. Neurotoxicology. https://doi.org/10.1016/j.neuro.2020.01.003
Kim J, Pajarillo E, Rizor A, Son DS, Lee J et al (2019) LRRK2 kinase plays a critical role in manganese-induced inflammation and apoptosis in microglia. PLoS One 14:e0210248. https://doi.org/10.1371/journal.pone.0210248
Kosinski M, McDonald K, Schwartz J, Yamamoto I, Greenstein D (2005) C. elegans sperm bud vesicles to deliver a meiotic maturation signal to distant oocytes. Development 132:3357–3369. https://doi.org/10.1242/dev.01916
Lee SS, Lee RY, Fraser AG, Kamath RS, Ahringer J et al (2003) A systematic RNAi screen identifies a critical role for mitochondria in C. elegans longevity. Nat Genet 33:40–48. https://doi.org/10.1038/ng1056
Liu Z, Mobley JA, DeLucas LJ, Kahn RA, West AB (2016) LRRK2 autophosphorylation enhances its GTPase activity. FASEB J 30:336–347. https://doi.org/10.1096/fj.15-277095
Luzon-Toro B, Rubio de la Torre E, Delgado A, Perez-Tur J, Hilfiker S (2007) Mechanistic insight into the dominant mode of the Parkinson's disease-associated G2019S LRRK2 mutation. Hum Mol Genet 16:2031–2039. https://doi.org/10.1093/hmg/ddm151
Maas JW, Yang J, Edwards RH (2017) Endogenous Leucine-rich repeat kinase 2 slows synaptic vesicle recycling in striatal neurons. Front Synaptic Neurosci 9:5. https://doi.org/10.3389/fnsyn.2017.00005
Melentijevic I, Toth ML, Arnold ML, Guasp RJ, Harinath G et al (2017) C. elegans neurons jettison protein aggregates and mitochondria under neurotoxic stress. Nature 542:367–371. https://doi.org/10.1038/nature21362
Miklavc P, Ehinger K, Thompson KE, Hobi N, Shimshek DR et al (2014) Surfactant secretion in LRRK2 knock-out rats: changes in lamellar body morphology and rate of exocytosis. PLoS One. 9:e84926. https://doi.org/10.1371/journal.pone.0084926
Naegeli KM, Hastie E, Garde A, Wang Z, Keeley DP et al (2017) Cell invasion in vivo via rapid exocytosis of a transient lysosome-derived membrane domain. Dev Cell. 43(403-417):e410. https://doi.org/10.1016/j.devcel.2017.10.024
Orenstein SJ, Kuo SH, Tasset I, Arias E, Koga H et al (2013) Interplay of LRRK2 with chaperone-mediated autophagy. Nat Neurosci 16:394–406. https://doi.org/10.1038/nn.3350
Proia P, Schiera G, Mineo M, Ingrassia AM, Santoro G et al (2008) Astrocytes shed extracellular vesicles that contain fibroblast growth factor-2 and vascular endothelial growth factor. Int J Mol Med. 21:63–67
Reddy A, Caler EV, Andrews NW (2001) Plasma membrane repair is mediated by Ca(2+)-regulated exocytosis of lysosomes. Cell 106:157–169. https://doi.org/10.1016/s0092-8674(01)00421-4
Richmond JE, Davis WS, Jorgensen EM (1999) UNC-13 is required for synaptic vesicle fusion in C. elegans. Nat Neurosci 2:959–964. https://doi.org/10.1038/14755
Roosen DA, Cookson MR (2016) LRRK2 at the interface of autophagosomes, endosomes and lysosomes. Mol Neurodegener 11:73. https://doi.org/10.1186/s13024-016-0140-1
Russell JC, Merrihew GE, Robbins JE, Postupna N, Kim T-K et al (2018) Isolation and characterization of extracellular vesicles from Caenorhabditis elegans for multi-omic analysis. J bioRxiv:476226. https://doi.org/10.1101/476226
Saha S, Guillily MD, Ferree A, Lanceta J, Chan D et al (2009) LRRK2 modulates vulnerability to mitochondrial dysfunction in Caenorhabditis elegans. J Neurosci 29:9210–9218. https://doi.org/10.1523/JNEUROSCI.2281-09.2009
Sakaguchi-Nakashima A, Meir JY, Jin Y, Matsumoto K, Hisamoto N (2007) LRK-1, a C. elegans PARK8-related kinase, regulates axonal-dendritic polarity of SV proteins. Curr Biol 17:592–598. https://doi.org/10.1016/j.cub.2007.01.074
Samann J, Hegermann J, von Gromoff E, Eimer S, Baumeister R et al (2009) Caenorhabditits elegans LRK-1 and PINK-1 act antagonistically in stress response and neurite outgrowth. J Biol Chem 284:16482–16491. https://doi.org/10.1074/jbc.M808255200
Sontag EM, Samant RS, Frydman J (2017) Mechanisms and functions of spatial protein quality control. Annu Rev Biochem 86:97–122. https://doi.org/10.1146/annurev-biochem-060815-014616
Tolosa E, Vila M, Klein C, Rascol O (2020) LRRK2 in Parkinson disease: challenges of clinical trials. Nat Rev Neurol 16:97–107. https://doi.org/10.1038/s41582-019-0301-2
Tsunemi T, Perez-Rosello T, Ishiguro Y, Yoroisaka A, Jeon S et al (2019) Increased Lysosomal exocytosis induced by Lysosomal Ca(2+) channel agonists protects human dopaminergic neurons from alpha-Synuclein Toxicity. J Neurosci 39:5760–5772. https://doi.org/10.1523/jneurosci.3085-18.2019
Wang J, Silva M, Haas LA, Morsci NS, Nguyen KC et al (2014) C. elegans ciliated sensory neurons release extracellular vesicles that function in animal communication. Curr Biol. 24:519–525. https://doi.org/10.1016/j.cub.2014.01.002
Ward S, Thomson N, White JG, Brenner S (1975) Electron microscopical reconstruction of the anterior sensory anatomy of the nematode Caenorhabditis elegans.?2UU. J Comp Neurol. 160:313–337. https://doi.org/10.1002/cne.901600305
Wehman AM, Poggioli C, Schweinsberg P, Grant BD, Nance J (2011) The P4-ATPase TAT-5 inhibits the budding of extracellular vesicles in C. elegans embryos. Curr Biol. 21:1951–1959. https://doi.org/10.1016/j.cub.2011.10.040
West AB (2017) Achieving neuroprotection with LRRK2 kinase inhibitors in Parkinson disease. Exp Neurol 298:236–245. https://doi.org/10.1016/j.expneurol.2017.07.019
Zimprich A, Biskup S, Leitner P, Lichtner P, Farrer M et al (2004) Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron 44:601–607. https://doi.org/10.1016/j.neuron.2004.11.005
Funding
This work was supported by the National Institutes of Health to MA and ABB (NIEHS R01ES007331 and R01ES010563). The authors thank the Analytical Imaging Facility (AIF) at Albert Einstein College of Medicine, which is sponsored by NCI cancer center support grant P30CA013330 and Shared Instrumentation Grant (SIG) 1S10OD023591-01. Some strains were provided by the CGC, which is funded by the NIH Office of Research Infrastructure Programs (P40 OD010440).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ke, T., Santamaria, A., Rocha, J.B.T. et al. The Role of Human LRRK2 in Methylmercury-Induced Inhibition of Microvesicle Formation of Cephalic Neurons in Caenorhabditis elegans. Neurotox Res 38, 751–764 (2020). https://doi.org/10.1007/s12640-020-00262-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00262-5