Abstract
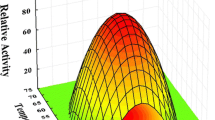
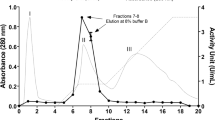
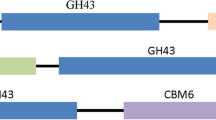
An endo-arabinanase (BLABNase) gene from Bacillus licheniformis DSM13 was cloned and expressed in Escherichia coli, and the biochemical properties of its encoded enzyme were characterized. The BLABNase gene consists of a single open reading frame of 987 nucleotides that encodes 328 amino acids with a predicted molecular mass of about 36 kDa. BLABNase exhibited the highest activity against debranched α-(1,5)-arabinan in 50 mM sodium acetate buffer (pH 6.0) at 55°C. Enzymatic characterization revealed that BLABNase hydrolyzes debranched or linear arabinans with a much higher activity than branched arabinan from sugar beet. Enzymatic hydrolysis pattern analyses demonstrated BLABNase to be a typical endo-(1,5)-α-s-arabinanase (EC 3.2.1.99) that randomly cleaves the internal α-(1,5)-linked L-arabinofuranosyl residues of a branchless arabinan backbone to release arabinotriose mainly, although a small amount of arabino-oligosaccharide intermediates is also liberated. Our results indicated that BLABNase acts preferentially along with the oligosaccharides longer than arabinopentaose, thus enabling the enzymatic production of various arabino-oligosaccharides.
Similar content being viewed by others
References
Alhassid, A., Ben-David, A., Tabachnikov, O., Libster, D., Naveh, E., Zolotnitsky, G., Shoham, Y., and Shoham, G. 2009. Crystal structure of an inverting GH 43 1,5-α-l-arabinanase from Geobacillus stearothermophilus complexed with its substrate. Biochem. J. 422, 73–82.
Bendtsen, J.D., Nielsen, H., von Heijne, G., and Brunak, S. 2004. Improved prediction of signal peptides: SignalP 3.0. J. Mol. Biol. 340, 783–795.
Grootaert, C., Delcour, J.A., Courtin, C.M., Broekaert, W.F., Verstraete, W., and Van de Wiele, T. 2007. Microbial metabolism and prebiotic potency of arabinoxylan oligosaccharides in the human intestine. Trends Food Sci. Technol. 18, 64–71.
Hizukuri, S. 1999. Nutritional and physiological functions and uses of L-arabinose. J. Appl. Glycosci. 46, 159–165.
Hong, M.R., Park, C.S., and Oh, D.K. 2009. Characterization of a thermostable endo-1,5-alpha-l-arabinanase from Caldicellulosiruptor saccharolyticus. Biotechnol. Lett. 31, 1439–1443.
Inácio, J.M. and de Sá-Nogueira, I. 2008. Characterization of abn2 (yxiA), encoding a Bacillus subtilis GH43 arabinanase, Abn2, and its role in arabino-polysaccharide degradation. J. Bacteriol. 190, 4272–4280.
Kim, T.J. 2008. Microbial exo- and endo-arabinosyl hydrolases: Structure, function, and application in l-arabinose production, pp. 229–257. In Park, K.H. (ed.) Carbohydrate-Active Enzymes. CRC Press, Woodhead Publishing Ltd., Cambridge, England.
Leal, T.F. and de Sá-Nogueira, I. 2004. Purification, characterization and functional analysis of an endo-arabinanase (AbnA) from Bacillus subtilis. FEMS Microbiol. Lett. 241, 41–48.
Lim, Y.R., Yeom, S.J., Kim, Y.S., and Oh, D.K. 2011. Synergistic production of L-arabinose from arabinan by the combined use of thermostable endo- and exo-arabinanases from Caldicellulosiruptor saccharolyticus. Bioresour. Technol. 102, 4277–4280.
McKie, V.A., Black, G.W., Millward-Sadler, S.J., Hazlewood, G.P., Laurie, J.I., and Gilbert, H.J. 1997. Arabinanase A from Pseudomonas fluorescens subsp. cellulosa exhibits both an endo- and an exo-mode of action. Biochem. J. 323, 547–555.
Miller, G.L. 1959. Use of dinitrosalicyclic acid reagent for determination of reducing sugars. Anal. Chem. 31, 426–428.
Numan, M.T. and Bhosle, N.B. 2006. α-l-Arabinofuranosidases: the potential applications in biotechnology. J. Ind. Microbiol. Biotechnol. 33, 247–260.
Nurizzo, D., Turkenburg, J.P., Charnock, S.J., Roberts, S.M., Dodson, E.J., McKie, V.A., Taylor, E.J., Gilbert, H.J., and Davies, G.J. 2002. Cellvibrio japonicus α-l-arabinanase 43A has a novel five-blade β-propeller fold. Nat. Struct. Biol. 9, 665–668.
Proctor, M.R., Taylor, E.J., Nurizzo, D., Turkenburg, J.P., Lloyd, R.M., Vardakou, M., Davies, G.J., and Gilbert, H.J. 2005. Tailored catalysis for plant cell-wall degradation: Redesigning the exo/endo preference of Cellvibrio japonicus arabinanase 43A. Proc. Natl. Acad. Sci. USA 102, 2697–2702.
Ramon, D., vd Veen, P., and Visser, J. 1993. Arabinan degrading enzymes from Aspergillus nidulans: induction and purification. FEMS Microbiol. Lett. 113, 15–22.
Seo, E.S., Lim, Y.R., Kim, Y.S., Park, C.S., and Oh, D.K. 2010. Characterization of a recombinant endo-1,5-α-l-arabinanase from the isolated bacterium Bacillus licheniformis. Biotechnol. Bioproc. Eng. 15, 590–594.
Seri, K., Sanai, K., Matsuo, N., Kawakubo, K., Xue, C., and Inoue, S. 1996. L-Arabinose selectively inhibits intestinal sucrase in an uncompetitive manner and suppresses glycemic response after sucrose ingestion in animals. Metabolism 45, 1368–1374.
Takao, M., Akiyama, K., and Sakai, T. 2002. Purification and characterization of thermostable endo-1,5-α-l-arabinase from a strain of Bacillus thermodenitrificans. Appl. Environ. Microbiol. 68, 1639–1646.
Veith, B., Herzberg, C., Steckel, S., Feesche, J., Maurer, K.H., Ehrenreich, P., B↑mer, S., Henne, A., Liesegang, H., Merkl, R., and et al. 2004. The complete genome sequence of Bacillus licheniformis DSM13, an organism with great industrial potential. J. Mol. Microbiol. Biotechnol. 7, 204–211.
Yamaguchi, A., Tada, T., Wada, K., Nakaniwa, T., Kitatani, T., Sogabe, Y., Takao, M., Sakai, T., and Nishimura, K. 2005. Structural basis for thermostability of endo-1,5-α-l-arabinanase from Bacillus thermodenitrificans TS-3. J. Biochem. 137, 587–592.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, JM., Jang, MU., Kang, JH. et al. Detailed modes of action and biochemical characterization of endo-arabinanase from Bacillus licheniformis DSM13. J Microbiol. 50, 1041–1046 (2012). https://doi.org/10.1007/s12275-012-2489-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-012-2489-3