Abstract
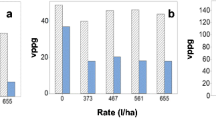
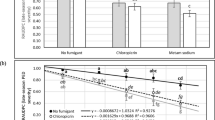
Verticillium wilt, caused by Verticillium dahliae Kleb., is a primary component of the early dying complex of potato (Solanum tuberosum L.) in the United States. Although genetic resistance to V. dahliae exists and has been incorporated into several potato cultivars, the commercial potato industry is still dominated by cultivars susceptible to the pathogen. As a result, soil fumigation with metam sodium remains an important means by which Verticillium wilt is controlled, despite its expense and potentially negative environmental impact. Recent restrictions on metam sodium use by the Environmental Protection Agency directed at reducing exposure to vapor emissions have increased the need to improve shank injection of the soil fumigant. In studies reported here, the application of metam sodium reduced the severity of Verticillium wilt, however, soil temperature at the time of injection, metam sodium injection depth, and application rate had little overall effect. In 2011, temperature at the time of metam sodium injection did not result in significant differences in any parameter evaluated. However, in 2012, soil populations of V. dahliae, wilt severity and host colonization were significantly reduced when metam sodium was applied at 4 °C compared to 13 or 15 °C. No significant differences were observed between a single or two metam sodium injection depths in any parameter evaluated across the 2 years the study was conducted. While all rates of metam sodium significantly reduced soil populations of V. dahliae compared to the non-treated control, significant differences across rates were rarely observed. Improved control of Verticillium wilt and increased yield can be achieved as a result of these studies. The effective control of Verticillium wilt can be obtained by using metam sodium at a comparatively low rate of 373 l/ha, particularly when applied at a relatively cold soil temperature of 4 °C using a single injection depth of 25 cm. The potential impact of these application modifications of metam sodium in reducing emissions and non-target exposure is discussed.
Resumen
La marchitez por Verticillium, causada por Verticillium dahliae Kleb., es un componente primario del complejo de muerte temprana de papa (Solanum tuberosum L.) en los Estados Unidos. Aun cuando existe resistencia genética a V. dahliae y se ha incorporado a varias variedades de papa, la industria de la papa comercial aún está dominada por variedades susceptibles al patógeno. Como resultado, la fumigación del suelo con metam sodio permanece como un medio importante por el cual se controla la marchitez por Verticillium, a pesar de que es caro y de su impacto ambiental potencialmente negativo. Restricciones recientes en el uso de metam sodio por la Agencia de Protección al Ambiente dirigidas a la reducción de la exposición a las emisiones de vapor, han aumentado la necesidad de mejorar las inyecciones del fumigante del suelo. En estudios aquí reportados, la aplicación del metam sodio redujo la severidad de la marchitez por Verticillium, no obstante, la temperatura del suelo al momento de la inyección, la profundidad de ésta, y la dosis de aplicación, tuvieron poco efecto en general. En 2011, la temperatura al momento de la inyección del metam sodio no resultó en diferencias significativas en cualquier parámetro. No obstante, en 2012, las poblaciones del suelo de V. dahliae, la severidad del marchitamiento y la colonización del hospedante se redujeron significativamente cuando el metam sodio se aplicó a 4 °C comparado a 13º o 15 °C. No se observaron diferencias significativas entre una o dos profundidades de inyección de metam sodio en cualquier parámetro evaluado a lo largo de los dos años de la conducción del estudio. Mientras todas las dosis de metam sodio redujeron significativamente las poblaciones en el suelo de V. dahliae comparadas con el testigo sin tratar, raramente se observaron diferencias significativas entre los niveles. El mejoramiento en el control del marchitamiento por Verticillium y el incremento en el rendimiento se pueden lograr como resultado de estos estudios. El control efectivo de la marchitez por Verticillium se puede obtener mediante el uso de metam sodio a una dosis comparativamente baja de 373 l/ha, particularmente cuando se aplica a una temperatura relativamente fría del suelo de 4 °C usando una sola profundidad de inyección de 25 cm. Se discute el impacto potencial de estas modificaciones de aplicación de metam sodio en la reducción de emisiones y de exposición a no-objetivos.

Similar content being viewed by others
References
Anonymous. 1998. Estimating soil moisture by feel and appearance. USDA, Natural Resources Conservation Service. Program Aid Number 1619. 6pp.
Bae, J., Z.K. Atallah, S.H. Jansky, D.I. Rouse, and W.R. Stevenson. 2007. Colonization dynamics and spatial progression of Verticillium dahliae in individual stems of two potato cultivars with differing responses to potato early dying. Plant Disease 91: 1137–1141.
Ben-Yephet, Y., and Z.R. Frank. 1985. Effect of soil structure on penetration by metham-sodium and of temperature on concentrations required to kill soilborne pathogens. Phytopathology 75: 403–406.
Ben-Yephet, Y., and Y. Szmulewich. 1985. Inoculum levels of Verticillium dahliae in the soils of the hot semi-arid Negev region of Israel. Phytoparasitica 13: 193–200.
Ben-Yephet, Y., E. Siti, and Z. Frank. 1983. Control of Verticillium dahliae by metam sodium in loessial soil and effect on potato tuber yields. Plant Disease 67: 1223–1225.
Bockus, W.W., and J.P. Shroyer. 1998. The impact of reduced tillage on soilborne plant pathogens. Annual Review of Phytopathology 36: 485–500.
Burpee, L.L., and J.R. Bloom. 1978. The influence of Pratylenchus penetrans on the incidence and severity of Verticillium wilt of potato. Journal of Nematology 10: 95–99.
Cappeart, M.R., M.L. Powelson, N.W. Christensen, and F.J. Crowe. 1992. Influence of irrigation on severity of potato early dying and tuber yield. Phytopathology 82: 1448–1453.
Collins, H.P., A. Alva, R.A. Boydston, R.L. Cochran, P.B. Hamm, A. McGuire, and E. Riga. 2006. Soil microbial, fungal, and nematode responses to soil fumigation and cover crops under potato production. Biology and Fertility of Soils 42: 247–257.
Corsini, D.L., J.R. Davis, and J.J. Pavek. 1985. Stability of resistance of potato to strains of Verticillium dahliae from different vegetative compatibility groups. Plant Disease 69: 980–982.
Corsini, D.L., J.J. Pavek, and J.R. Davis. 1990. Verticillium wilt resistant potato germplasm: A66107-51 and A68113-4. American Potato Journal 67: 517–525.
Cox, C. 2006. Metam sodium: fumigant factsheet. Journal of Pesticide Reform 26: 12–16.
Davis, J.R. 1985. Approaches to control of potato early dying caused by Verticillium dahliae. American Potato Journal 62: 177–185.
Davis, J.R., and D.O. Everson. 1986. Relation of Verticillium dahliae in soil and potato tissue, irrigation method, and N-fertility to Verticillium wilt of potato. Phytopathology 76: 730–736.
Davis, J.R., and O.C. Huisman. 2001. Verticillium wilt. In Compendium of potato diseases, 45–46. Minneapolis: APS Press.
Davis, J.R., and L.H. Sorenson. 1986. Influence of soil solarization at moderate temperatures on potato genotypes with differing resistance to Verticillium dahliae. Phytopathology 76: 1021–1026.
Davis, J.R., W.H. Loescher, M.W. Hammond, and R.E. Thornton. 1983a. Response of Russet Burbank potatoes to soil fumigation and nitrogen fertilizers. American Potato Journal 63: 71–79.
Davis, J.R., J.J. Pavek, and D.L. Corsini. 1983b. A sensitive method for quantifying Verticillium dahliae colonization in plant tissue and evaluating resistance among potato genotypes. Phytopathology 73: 1009–1014.
Davis, J.R., J.J. Pavek, D.L. Corsini, L.H. Sorenson, A.T. Schneider, D.O. Everson, D.T. Westermann, and O.C. Huisman. 1994. Influence of continuous cropping of several potato clones on the epidemiology of Verticillum wilt of potato. Phytopathology 84: 207–214.
Davis, J.R., O.C. Huisman, D.T. Westermann, S.L. Hafez, D.O. Everson, L.H. Sorenson, and A.T. Schneider. 1996. Effects of green manures on Verticillium wilt of potato. Phytopathology 86: 444–453.
Davis, J.R., O.C. Huisman, D.O. Everson, and A.T. Schneider. 2001. Verticillium wilt of potato: a model of key factors related to disease severity and tuber yield in southeastern Idaho. American Journal of Potato Research 78: 291–300.
DeRoo, H.C., and P.E. Waggoner. 1961. Root development of potatoes. Agronomy Journal 53: 15–17.
Dung, J.K.S., and D.K. Johnson. 2012. Roles of infected seed tubers and soilborne inoculum on Verticillium wilt of ‘Russet Burbank’ potato. Plant Disease 96: 379–383.
Farley, J.D. 1972. A selective medium for assay of Colletotrichum coccodes in soil. Phytopathology 62: 1288–1293.
Frost, K.E., D.I. Rouse, and S.H. Jansky. 2007. Considerations for Verticillium wilt resistance in potato. Plant Disease 91: 360–367.
Fry, W.E. 1978. Quantification of general resistance of potato cultivars and fungicide effects for integrated control of potato late blight. Phytopathology 68: 1650–1655.
Hamm, P.B., R.E. Ingham, J.R. Jaeger, W.H. Swanson, and K.C. Volker. 2003. Soil fumigant effects on three genera of potential soilborne pathogenic fungi and their effect on potato yield in the Columbia Basin of Oregon. Plant Disease 87: 1449–1456.
Hoyos, G.P., P.J. Zambino, and N.A. Anderson. 1991. An assay to quantify vascular colonization of potato by Verticillium dahliae. American Potato Journal 68: 727–743.
Hu, X., R.N. Nazar, and J. Robb. 1993. Quantification of Verticillium biomass in wilt disease development. Physiological and Molecular Plant Pathology 42: 23–36.
Jansky, S.H. 2009. Identification of Verticillium wilt resistance in U.S. potato breeding programs. American Journal of Potato Research 86: 504–512.
Klose, S., J.A. Ajwa, G.T. Browne, K.V. Subbarao, F.N. Martin, S.A. Fennimore, and B.B. Westerdahl. 2008. Dose response of weed seeds, plant-parasitic nematodes, and pathogens to twelve rates of metam sodium in a California soil. Plant Disease 92: 1537–1546.
Larkin, R.P., and T.S. Griffin. 2007. Control of soilborne potato diseases using Brassica green manures. Crop Protection 26: 1067–1077.
Larkin, R.P., C.W. Honeycutt, and O.M. Olanya. 2011. Management of Verticillium wilt of potato with disease-suppressive green manures and as affected by previous cropping history. Plant Disease 95: 568–576.
Leistra, M., and J.H. Smelt. 1974. Optimum soil conditions for fumigation with metam-sodium. Agro-Ecosystems 1: 169–174.
Loria, R. 2001. Development and anatomy of the potato plant. In Compendium of potato diseases, 2–3. Minneapolis: APS Press.
Macalady, J.L., M.E. Fuller, and K.M. Scow. 1998. Effects of metam sodium fumigation on soil microbial activity and community structure. Journal of Environmental Quality 27: 54–63.
MacRae, A., and J. Noling. 2010. Overview of new EPA regulations affecting use of metam sodium and metam potassium. University of Florida Extension HS1167.
Martin, M.J., R.M. Riedel, and R.C. Rowe. 1982. Verticillium dahliae and Pratylenchus penetrans: interactions in the early dying complex of potato in Ohio. Phytopathology 72: 640–644.
McGuire, A.M. 2003. Mustard green manures replace fumigant and improve infiltration in potato cropping system. Crop Management. doi:10.1094/CM-2003-0822-01-RS. Online.
Nicot, P.C., and D.I. Rouse. 1987a. Precision and bias of three quantitative soil assays for Verticillium dahliae. Phytopathology 77: 875–881.
Nicot, P.C., and D.I. Rouse. 1987b. Relationship between soil inoculum density of Verticillium dahliae and systemic colonization of potato stems in commercial fields over time. Phytopathology 77: 1346–1355.
Nnodu, E.C., and M.D. Harrison. 1979. The relationship between Verticillium albo-atrum inoculum density and potato yield. American Journal of Potato Research 56: 11–25.
Pasche, J.S., I. Mallik, N.R. Anderson, and N.C. Gudmestad. 2013a. Development and validation of a real-time PCR assay for the quantification of Verticillium dahliae in potato. Plant Disease 97: 608–618.
Pasche, J.S., A.L. Thompson, and N.C. Gudmestad. 2013b. Quantification of field resistance to Verticillium dahliae in eight russet-skinned potato cultivars using real-time PCR. American Journal of Potato Research 90: 158–170.
Powelson, M.L., and R.C. Rowe. 1993. Biology and management of early dying of potatoes. Annual Review of Phytopathology 31: 111–126.
Powelson, M.L., K.B. Johnson, and R.C. Rowe. 1993. Management of diseases caused by soilborne pathogens. In Potato health management, ed. R.C. Rowe, 149–151. St. Paul: American Phytopathological Society.
Rich, A.E. 1983. Verticillium wilt. In Potato diseases, 71–78. New York: Academic Press
Rothrock, C.S. 1992. Tillage systems and plant disease. Soil Science 154: 308–315.
Rowe, R.C. 1985. Potato early dying—a serious threat to the potato industry. American Potato Journal 62: 157–161.
Rowe, R.C., and M.L. Powelson. 2002. Potato early dying: management challenges in a changing production environment. Plant Disease 86: 1184–1193.
Rowe, R.C., R.M. Riedel, and M.J. Martin. 1985. Synergistic interactions between Verticillium dahliae and Pratylenchus penetrans in potato early dying disease. Phytopathology 75: 412–418.
Rowe, R.C., J.R. Davis, M.L. Powelson, and D.I. Rouse. 1987. Potato early dying: causal agents and management strategies. Plant Disease 71: 482–489.
Saeed, I.A.M., D.I. Rouse, J.M. Harkin, and K.P. Smith. 1997. Effects of soil water content and soil temperature on erfficacy of metham-sodium against Verticillium dahliae. Plant Disease 81: 773–776.
Saeed, I.A.M., D.I. Rouse, and J.M. Harkin. 2000. Methyl isothiocyanate volatilization from fields treated with metam-sodium. Pest Management Science 56: 813–817.
Shaner, G., and R.E. Finney. 1977. The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology 67: 1051–1056.
Smelt, J.H., and M. Leistra. 1974. Conversion of metham-sodium to methyl isothiocyanate and basic data on the behaviour of methyl isothiocyante in soil. Pesticide Science 5: 401–407.
Sullivan, D.A., M.T. Holdsworth, and D.J. Hlinka. 2004. Control of off-gassing rates of methyl isothiocyanate from the application of metam-sodium by chemigation and shank injection. Atmospheric Environment 38: 2457–2470.
Taylor, R.J., J.S. Pasche, and N.C. Gudmestad. 2005. Influence of tillage and method of metam sodium application on distribution and survival of Verticillium dahliae in the soil and the development of Verticillium wilt of potato. American Journal of Potato Research 82: 451–461.
Tsror (Lahkim), L., E. Shlevin, and I. Peretz-Alon. 2005. Efficacy of metam sodium for controlling Verticillium dahliae prior to potato production in sandy soils. American Journal of Potato Research 82: 419–424.
Vanachter, A., and C. Van Assche. 1970. The influence of soil temperature and moisture content on the effect of soil fumigants. Netherlands Journal of Plant Pathology 76: 240–248.
Wilhelm, S. 1955. Longevity of Verticillium wilt fungus in the laboratory and field. Phytopathology 45: 180–181.
Yates, S.R., J. Gan, S.K. Papiernik, R. Dungan, and D. Wang. 2002. Reducing fumigant emissions after soil application. Phytopathology 92: 1344–1348.
Zhang, Y., and D. Wang. 2007. Emission, distribution and leaching of methyl isothiocyanate and chloropicrin under different surface containments. Chemosphere 68: 445–454.
Zheng, W., S.R. Yates, S.K. Papiernik, and J. Nunez. 2006. Conversion of metam sodium and emission of fumigant from soil columns. Atmospheric Environment 40: 7046–7056.
Acknowledgments
The authors gratefully acknowledge the financial support of the Northern Plains Potato Growers Association, Minnesota Area II Potato Growers, AmVac, Inc., RD Offutt, Co., CSS Farms and Walther Farms. The authors also gratefully acknowledge the technical assistance of Dean Peterson, Russell Benz and Ipsita Mallik.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pasche, J.S., Taylor, R.J., David, N.L. et al. Effect of Soil Temperature, Injection Depth, and Metam Sodium Rate on the Management of Verticillium Wilt of Potato. Am. J. Potato Res. 91, 277–290 (2014). https://doi.org/10.1007/s12230-013-9348-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-013-9348-6