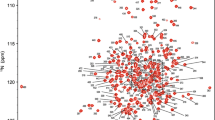
Abstract
Almost complete assignment of backbone 1H, 13C, 15N and side chain 13Cβ resonances for the immune-regulatory cytokine IL-10 is reported. The protein was overexpressed in Escherichia coli and was refolded from inclusion bodies. The point mutation C149Y was introduced to suppress incorrect disulfide bond formation and to improve protein refolding.


Similar content being viewed by others
References
Angulo J, Rademacher C, Biet T, Benie AJ, Blume A, Peters H, Palcic M, Parra F, Peters T (2006) NMR analysis of carbohydrate–protein interactions. Meth Enzymol 416:12–30
Asadullah K, Sterry W, Volk HD (2003) Interleukin-10 therapy—review of a new approach. Pharmacol Rev 55:241–269
Ball C, Vigues S, Gee CK, Poole S, Bristow AF (2001) Rat interleukin-10: production and characterisation of biologically active protein in a recombinant bacterial expression system. Eur Cytokine Netw 12:187–193
Bortesi L, Rossato M, Schuster F, Raven N, Stadlmann J, Avesani L, Falorni A, Bazzoni F, Bock R, Schillberg S, Pezzotti M (2009) Viral and murine interleukin-10 are correctly processed and retain their biological activity when produced in tobacco. BMC Biotechnol 9:22–35
Coombe DR (2008) Biological implications of glycosaminoglycan interactions with haemopoietic cytokines. Immunol Cell Biol 86:598–607
de Waal MR, Yssel H, Roncarolo MG, Spits H, de Vries JE (1992) Interleukin-10. Curr Opin Immunol 4:314–320
Gandhi NS, Mancera RL (2008) The structure of glycosaminoglycans and their interactions with proteins. Chem Biol Drug Des 72:455–482
Grzesiek S, Dobeli H, Gentz R, Garotta G, Labhardt AM, Bax A (1992) 1H, 13C, and 15N NMR backbone assignments and secondary structure of human interferon-gamma. Biochemistry 31:8180–8190
Kim JM, Brannan CI, Copeland NG, Jenkins NA, Khan TA, Moore KW (1992) Structure of the mouse IL-10 gene and chromosomal localization of the mouse and human genes. J Immunol 148:3618–3623
Moore KW, Vieira P, Fiorentino DF, Trounstine ML, Khan TA, Mosmann TR (1990) Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein–Barr virus gene BCRFI. Science 248:1230–1234
Moore KW, O’Garra A, de Waal MR, Vieira P, Mosmann TR (1993) Interleukin-10. Annu Rev Immunol 11:165–190
Moore KW, de Waal MR, Coffman RL, O’Garra A (2001) Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol 19:683–765
Peattie RA (2012) Release of growth factors, cytokines and therapeutic molecules by hyaluronan-based hydrogels. Curr Pharm Biotechnol 13:1299–1305
Roers A, Müller W (2008) Distinct functions of interleukin-10 derived from different cellular sources. Curr Immunol Rev 4:37–42
Sabat R, Grutz G, Warszawska K, Kirsch S, Witte E, Wolk K, Geginat J (2010) Biology of interleukin-10. Cytokine Growth Factor Rev 21:331–344
Salek-Ardakani S, Arrand JR, Shaw D, Mackett M (2000) Heparin and heparan sulfate bind interleukin-10 and modulate its activity. Blood 96:1879–1888
Schiller J, Huster D (2012) New methods to study the composition and structure of the extracellular matrix in natural and bioengineered tissues. Biomatter 2:115–131
Schultz GS, Wysocki A (2009) Interactions between extracellular matrix and growth factors in wound healing. Wound Repair Regen 17:153–162
Shen Y, Delaglio F, Cornilescu G, Bax A (2009) TALOS+: a hybrid method for predicting protein backbone torsion angles from NMR chemical shifts. J Biomol NMR 44:213–223
Spera S, Bax A (1991) Empirical correlation between protein backbone conformation and Cα and Cβ 13C nuclear magnetic resonance chemical shifts. J Am Chem Soc 113:5490–5492
Windsor WT, Syto R, Tsarbopoulos A, Zhang R, Durkin J, Baldwin S, Paliwal S, Mui PW, Pramanik B, Trotta PP (1993) Disulfide bond assignments and secondary structure analysis of human and murine interleukin 10. Biochemistry 32:8807–8815
Wishart DS, Bigam CG, Holm A, Hodges RS, Sykes BD (1995a) 1H, 13C and 15N random coil NMR chemical shifts of the common amino acids. I. Investigations of nearest-neighbor effects. J Biomol NMR 5:67–81
Wishart DS, Bigam CG, Yao J, Abildgaard F, Dyson HJ, Oldfield E, Markley JL, Sykes BD (1995b) 1H, 13C and 15N chemical shift referencing in biomolecular NMR. J Biomol NMR 6:135–140
Zdanov A (2004) Structural features of the interleukin-10 family of cytokines. Curr Pharm Des 10:3873–3884
Zdanov A, Schalk-Hihi C, Gustchina A, Tsang M, Weatherbee J, Wlodawer A (1995) Crystal structure of interleukin-10 reveals the functional dimer with an unexpected topological similarity to interferon gamma. Structure 3:591–601
Acknowledgments
We thank Sebastian Lanvermann (Technical University Dresden) who provided the IL-10 cDNA and Professors Stefan Berger (University of Leipzig) and Jochen Balbach (Martin Luther University Halle/Wittenberg) who kindly provided access to their NMR spectrometers. The study was supported by a grant from the Deutsche Forschungsgemeinschaft (Transregio 67, A6) and a grant from the Stipendienfonds der Chemischen Industrie e.V.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Künze, G., Theisgen, S. & Huster, D. Backbone 1H, 15N, 13C and side chain 13Cβ NMR chemical shift assignment of murine interleukin-10. Biomol NMR Assign 8, 375–378 (2014). https://doi.org/10.1007/s12104-013-9521-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-013-9521-3