Abstract
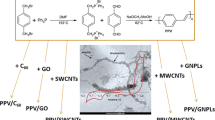
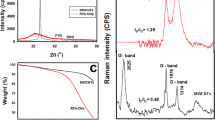
Covalent crosslinking of carbon nanostructures of different dimensionalities such as nanodiamond, single walled carbon nanotubes (SWNTs) and graphene can yield useful homo- and hetero-binary conjugates. Binary conjugation of the nanocarbons has been achieved by introducing symmetrical amide-linkages between acid (-COOH) functionalized nanocarbons and a diamine-linker. The binary conjugates have been characterized by using transmission electron microscopy as well as infrared, Raman and photoluminescence spectroscopies. Dispersions of covalently crosslinked binary conjugates of nanocarbons could be obtained in dimethyl formamide (DMF). Composites of the binary conjugates with polymer can be readily prepared by using the DMF suspensions.

Covalent crosslinking of carbon nanostructures of different dimensionalities such as nanodiamond, single-walled carbon nanotubes (SWNTs) and graphene yield useful homo- and hetero-binary conjugates. Dispersions of covalently crosslinked binary conjugates of nanocarbons could be obtained in dimethyl formamide (DMF).
Similar content being viewed by others
References
D’Evelyn M P 1998 Surface Properties of diamond. In Handbook of industrial diamonds and diamond films (eds.) M A Prelas, G Popovici and L K Bigelow (New York: Marcel Dekker Inc.) p 89
Dolmatov V Y 2001 Russ. Chem. Rev. 70 607
Zhu W, Kochanski G P and Jin S 1998 Science 282 1471
Yu S J, Kang M W, Chang H C, Chen K M and Yu Y C 2005 J. Am. Chem. Soc. 127 17604
Fu C C, Lee H Y, Chen K, Lim T S, Wu H Y, Lin P K, Wei P K, Tsao P H, Chang H C and Fann W 2007 Proc. Natl. Acad. Sci. USA 104 727
Zhao W, Qiu J X Q and Chen H Y 2006 Biosens. Bioelectron. 22 649
Rao C N R and Govindaraj A 2005 Nanotubes and Nanowires; Royal Society of Chemistry: Cambridge, UK
Saito R, Dresselhaus G and Dresselhaus M S 1998 Physical Properties of Carbon Nanotubes, Imperial College Press: London, UK
(a) Dai H 2002 Acc. Chem. Res. 35 1035; (b) Banerjee S, Hemraj-Benny T and Wong S-S 2005 Adv. Mater. 17 17
Rao C N R, Sood A K, Subrahmanyam K S and Govindaraj A 2009 Angew. Chem. Int. Ed. 48 7752
Prasad K E, Das B, Maitra U, Ramamurty U and Rao C N R 2009 Proc. Natl. Acad. Sci. USA 106 13186
Guglielmotti V, Chieppa S, Orlanducci S, Tamburri E, Toschi F, Terranova M L and Rossi M 2009 Appl. Phys. Lett. 95 222113
Chiu P W, Duesburg G S, Wegiikowska U D and Roth S 2002 Appl. Phys. Lett. 80 3811
Frehill F, Vos J G, Benrezzak S, Koós A A, Kónya Z, Rüther M G, Blau W J, Fonseca A, Nagy J B, Biró L P, Minett A I and Panhuis M 2002 J. Am. Chem. Soc. 124 13694
Holzinger M, Steinmetz J, Samaille D, Glerup M, Paillet M, Bernier P, Ley L and Graupner R 2004 Carbon 42 941
Avinash M B, Subrahmanyam K S, Sundarayya Y and Govindaraju T 2010 Nanoscale 2 1762
Dresselhaus M S, Jorio A and Saito R 2010 Annu. Rev. Condens. Matter Phys. 1 89
Dresselhaus M S and Eklund P C 2000 Adv. Phys. 49 705
Das B, Voggu R, Rout C S and Rao C N R 2008 Chem. Commun. 41 5155
Weglikowska U D, Benoit J M, Chiu P W, Graupner R, Lebedkin S and Roth S 2002 Curr. Appl. Phys. 2 497
Rao C N R, Biswas K, Subrahmanyam K S and Govindaraj A 2009 J. Mater. Chem. 19 2457
Jaramillo D, Wheate N J, Ralph S F, Howard W A, Tor Y, Aldrich-Wright J R 2006 Inorg. Chem. 45 6004
Maitra U, Gomathi A and Rao C N R 2008 J. Exp. Nanosci. 3 271
Subrahmanyam K S, Ghosh A, Gomathi A, Govindaraj A and Rao C N R 2009 Nanosci. Nanotech. Lett. 1 28
Rao C N R, Sood A K, Voggu R, Subrahmanyam K S 2010 J. Phys. Chem. Lett. 1 572
Chung P-H, Perevedentseva E and Cheng C-L 2007 Surf. Sci. 601 3866
Vlasov I I, Shenderova O, Turner S, Lebedev O I, Basov A A, Sildos I, Rähn M, Shiryaev A A and Tendeloo G V 2010 Small 6 687
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
MAITRA, U., PANDEESWAR, M. & GOVINDARAJU, T. Covalent crosslinking of carbon nanostructures. J Chem Sci 124, 551–556 (2012). https://doi.org/10.1007/s12039-012-0255-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-012-0255-z