Abstract
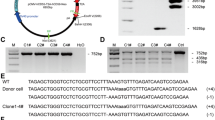
Massachusetts General Hospital miniature pigs (MGH minipigs) have been established for organ transplantation studies across the homozygous major histocompatibility complex, but cloning efficiency of MGH minipigs is extremely low. This study was designed to increase the productivity of MGH minipigs by nuclear transfer of post-thaw donor cells after 1 h co-incubation with roscovitine. The MGH minipig cells were genetically modified with GT KO (alpha1,3-galactosyltransferase knock-out) and hCD46 KI (human CD46 knock-in) and used as donor cells. The GT KO/hCD46 KI donor cells were cultured for either 3 days (control group) or 1 h after thawing with 15 μM roscovitine (experimental group) prior to the nuclear transfer. The relative percentage of the transgenic donor cells that entered into G0/G1 was 93.7 % (±2.54). This was different from the donor cells cultured for 1 h with the roscovitine-treated group (84.6 % ±4.6) (P < 0.05) and without roscovitine (78.6 % ±5.5) (P < 0.01), respectively. The pregnancy rate and delivery rate in the roscovitine group (8/12 and 6/8, respectively) were significantly higher (P < 0.01) than those in the control group (6/19 and 3/6, respectively). In the experimental group, 12 GT KO/hCD46 KI transgenic minipigs were successfully generated, and five minipigs among them survived for more than 6 months so far. The recipient-based individual cloning efficiency ranged from 0.74 to 2.54 %. In conclusion, gene-modified donor cells can be used for cloning of MGH minipigs if the cells are post-thawed and treated with roscovitine for 1 h prior to nuclear transfer.

Similar content being viewed by others
References
Griesemer, A. D., Hirakata, A., Shimizu, A., Moran, S., Tena, A., Iwaki, H., et al. (2009). Results of Gal-Knockout porcine thymokidney xenografts. American Journal of Transplantation, 9, 2669–2678.
Yamada, K., Yazawa, K., Shimizu, A., Iwanaga, T., Hisashi, Y., Nuhn, M., et al. (2005). Marked prolongation of porcine renal xenograft survival in baboons through the use of alpha1, 3-galactosyltransferase gene-knockout donors and the cotransplantation of vascularized thymic tissue. Nature Medicine, 11, 32–34.
Vodicka, P., Smetana, K, Jr, Dvorankova, B., Emerick, T., Xu, Y. Z., Ourednik, J., et al. (2005). The miniature pig as an animal model in biomedical research. Annals of the New York Academy of Sciences, 1049, 161–171.
Hanekamp, J. S., Duran-Struuck, R., & Sachs, D. H. (2011). Allotransplantation in miniature swine in genetics and immunology. In P. A. McAnulty, A. D. Dayan, N. C. Ganderup, & K. L. Hastings (Eds.), The minipig in biological research (pp. 357–372). Boca Raton: CRC.
Sachs, D. H., Leight, G., Cone, J., Schwarz, S., Stuart, L., & Rosenberg, S. (1976). Transplantation in miniature swine, I: fixation of the major histocompatibility complex. Transplantation, 22, 559–567.
Lai, L., Kolber-Simonds, D., Park, K. W., Cheong, H. T., Greenstein, L. J., et al. (2002). Production of alpha-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science, 295, 1089–1092.
Zhao, J., Ross, J. W., Hao, Y., Spate, L. D., Walters, E. M., Samuel, M. S., et al. (2009). Significant improvement in cloning efficiency of an inbred miniature pig by histone deacetylase inhibitor treatment after somatic cell nuclear transfer. Biology of Reproduction, 81, 525–530.
Alessi, F., Quarta, S., Savio, M., Riva, F., Rossi, L., Stivala, L. A., et al. (1998). The cyclin dependent kinase inhibitors olomoucine and roscovitine arrest human fibroblasts in G1 phase by specific inhibition of CDK2 kinase activity. Experimental Cell Research, 245, 8–18.
Park, H. J., Koo, O. J., Kwon, D. K., Kang, J. T., Jang, G., & Lee, B. C. (2010). Effect of roscovitine-treated donor cells on development of porcine cloned embryos. Reproduction in Domestic Animals, 45, 1082–1088.
Ko, N., Lee, J. W., Hwang, S., Kim, B., Park, J. K., Ock, S. A., et al. (2013) Nucleofection-mediated alpha1, 3-galactosyltransferase gene inactivation and membrane cofactor protein expression for pig-to-primate xenotransplantation. Animal Biotechnology (in press).
Ahn, K. S., Kim, Y. J., Kim, M., Lee, B. H., Heo, S. Y., Kang, M. J., et al. (2011). Resurrection of an alpha-1,3-galactosyltransferase gene-targeted miniature pig by recloning using postmortem ear skin fibroblasts. Theriogenology, 75, 933–939.
Mezrich, J. D., Haller, G. W., Arn, J. S., Houser, S. L., Madsen, J. C., & Sachs, D. H. (2003). Histocompatible miniature swine: an inbred large-animal model. Transplantation, 27, 904–907.
Conley, A. J., Jung, Y. C., Schwartz, N. K., Warner, C. M., Rothschild, M. F., & Ford, S. P. (1988). Influence of SLA haplotype on ovulation rate and litter size in miniature pigs. Journal of Reproduction and Fertility, 82, 595–601.
Miyamoto, K., Hoshino, Y., Minami, N., Yamada, M., & Imai, H. (2007). Effects of synchronization of donor cell cycle on embryonic development and DNA synthesis in porcine nuclear transfer embryos. Journal of Reproduction and Development, 53, 237–246.
Shufaro, Y., & Reubinoff, B. E. (2011). Cell cycle synchronization for the purpose of somatic cell nuclear transfer (SCNT). Methods in Molecular Biology, 761, 239–247.
Nagashima, H., Fujimura, T., Takahagi, Y., Kurome, M., Wako, N., Ochiai, T., et al. (2003). Development of efficient strategies for the production of genetically modified pigs. Theriogenology, 59, 95–106.
Hayes, O., Ramos, B., Rodriguez, L. L., Aguilar, A., Badia, T., & Castro, F. O. (2005). Cell confluency is as efficient as serum starvation for inducing arrest in the G0/G1 phase of the cell cycle in granulosa and fibroblast cells of cattle. Animal Reproduction Science, 87, 181–192.
Cho, S. R., Ock, J. G., Yoo, B., Kumar, M., Choe, S. Y., & Rho, G. J. (2005). Effects of confluent, roscovitine treatment and serum starvation on the cell-cycle synchronization of bovine foetal fibroblasts. Reproduction in Domestic Animals, 45(40), 171–176.
Kues, W. A., Carnwath, J. W., Paul, D., & Niemann, H. (2002). Cell cycle synchronization of porcine fetal fibroblasts by serum deprivation initiates a nonconventional form of apoptosis. Cloning and Stem Cells, 4, 231–243.
Acknowledgments
This study received Grant support from the Agenda Program (No. PJ008587) and (PJ009095), Rural Development Administration (http://www.rda.go.kr), Republic of Korea. The authors appreciate Professor Hochi Shinichi (Faculty of Textile Science and Technology, Shinshu University, Nagano, Japan) for his helpful discussions to improve this article.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hwang, S., Oh, K.B., Kwon, DJ. et al. Improvement of Cloning Efficiency in Minipigs Using Post-thawed Donor Cells Treated with Roscovitine. Mol Biotechnol 55, 212–216 (2013). https://doi.org/10.1007/s12033-013-9671-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-013-9671-7