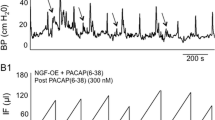
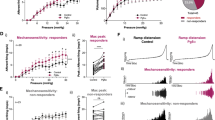
Abstract
Neural injury, inflammation, or diseases commonly and adversely affect micturition reflex function that is organized by neural circuits in the CNS and PNS. One neuropeptide receptor system, pituitary adenylate cyclase-activating polypeptide (PACAP; Adcyap1), and its cognate receptor, PAC1 (Adcyap1r1), have tissue-specific distributions in the lower urinary tract. PACAP and associated receptors are expressed in the LUT and exhibit changes in expression, distribution, and function in preclinical animal models of bladder pain syndrome (BPS)/interstitial cystitis (IC), a chronic, visceral pain syndrome characterized by pain, and LUT dysfunction. Blockade of the PACAP/PAC1 receptor system reduces voiding frequency and somatic (e.g., hindpaw, pelvic) sensitivity in preclinical animal models and a transgenic mouse model that mirrors some clinical symptoms of BPS/IC. The PACAP/receptor system in micturition pathways may represent a potential target for therapeutic intervention to reduce LUT dysfunction following urinary bladder inflammation.


Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- BOO:
-
Bladder outlet obstruction
- BPS:
-
Bladder pain syndrome
- CGRP:
-
Calcitonin-gene related peptide
- CNS:
-
Central nervous system
- CYP:
-
Cyclophosphamide
- DO:
-
Detrusor overactivity
- DRG:
-
Dorsal root ganglion
- GPCR:
-
G protein-coupled receptor
- HPA:
-
Hypothalamic-pituitary-adrenal
- IC:
-
Interstitial cystitis
- LUT:
-
Lower urinary tract
- NGF:
-
Nerve growth factor
- NGF-OE:
-
Nerve growth factor overexpression
- NVC:
-
Non-voiding bladder contraction
- PAC1:
-
PACAP type I receptor
- PACAP:
-
Pituitary adenylate cyclase-activating polypeptide
- PAG:
-
Periaqueductal gray
- PC:
-
Pheochromocytoma
- PMC:
-
Pontine micturition center
- PNS:
-
Peripheral nervous system
- Sub-P:
-
Substance P
- TrkA:
-
Receptor tyrosine kinase A, tropomyosin-related kinase A
- TrkB:
-
Receptor tyrosine kinase B, tropomyosin-related kinase B
- TrkC:
-
Receptor tyrosine kinase C, tropomyosin-related kinase C
- VIP:
-
Vasoactive intestinal polypeptide
- VPAC1:
-
VIP receptor 1
- VPAC2:
-
VIP receptor 2
References
Aloe L, Tuveri MA, Carcassi U, Levi-Montalcini R (1992) Nerve growth factor in the synovial fluid of patients with chronic arthritis. Arthritis Rheum 35(3):351–355
Andersson KE (2002) Bladder activation: afferent mechanisms. Urology 59(5 Suppl 1):43–50
Andersson KE, Arner A (2004) Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiol Rev 84(3):935–986. https://doi.org/10.1152/physrev.00038.2003
Apfel SC (2001) Neurotrophic factor therapy—prospects and problems. Clin Chem Lab Med 39(4):351–355. https://doi.org/10.1515/CCLM.2001.055
Arimura A (1998) Perspectives on pituitary adenylate cyclase activating polypeptide (PACAP) in the neuroendocrine, endocrine, and nervous systems. Jpn J Physiol 48(5):301–331
Arms L, Girard BM, Vizzard MA (2010) Expression and function of CXCL12/CXCR4 in rat urinary bladder with cyclophosphamide-induced cystitis. Am J Physiol Renal Physiol 298(3):F589–F600. https://doi.org/10.1152/ajprenal.00628.2009
Batista CK, Brito GA, Souza ML, Leitao BT, Cunha FQ, Ribeiro RA (2006) A model of hemorrhagic cystitis induced with acrolein in mice. Braz J Med Biol Res 39(11):1475–1481
Beaudet MM, Braas KM, May V (1998) Pituitary adenylate cyclase activating polypeptide (PACAP) expression in sympathetic preganglionic projection neurons to the superior cervical ganglion. J Neurobiol 36(3):325–336
Beckel JM, Holstege G (2011) Neuroanatomy of the lower urinary tract. Handb Exp Pharmacol 202:99–116. https://doi.org/10.1007/978-3-642-16499-6_6
Botz B, Imreh A, Sandor K, Elekes K, Szolcsanyi J, Reglodi D et al (2013) Role of pituitary adenylate-cyclase activating polypeptide and Tac1 gene derived tachykinins in sensory, motor and vascular functions under normal and neuropathic conditions. Peptides 43:105–112. https://doi.org/10.1016/j.peptides.2013.03.003
Boucher W, el-Mansoury M, Pang X, Sant GR, Theoharides TC (1995) Elevated mast cell tryptase in the urine of patients with interstitial cystitis. Br J Urol 76(1):94–100
Braas KM, May V (1996) Pituitary adenylate cyclase-activating polypeptides, PACAP-38 and PACAP-27, regulation of sympathetic neuron catecholamine, and neuropeptide Y expression through activation of type I PACAP/VIP receptor isoforms. Ann N Y Acad Sci 805:204–216 discussion 217–208
Braas KM, May V (1999) Pituitary adenylate cyclase-activating polypeptides directly stimulate sympathetic neuron neuropeptide Y release through PAC(1) receptor isoform activation of specific intracellular signaling pathways. J Biol Chem 274(39):27702–27710
Braas KM, May V, Harakall SA, Hardwick JC, Parsons RL (1998) Pituitary adenylate cyclase-activating polypeptide expression and modulation of neuronal excitability in Guinea pig cardiac ganglia. J Neurosci 18(23):9766–9779
Braas KM, May V, Zvara P, Nausch B, Kliment J, Dunleavy JD, Nelson MT, Vizzard MA (2006) Role for pituitary adenylate cyclase activating polypeptide in cystitis-induced plasticity of micturition reflexes. Am J Physiol Regul Integr Comp Physiol 290(4):R951–R962. https://doi.org/10.1152/ajpregu.00734.2005
Butrick CW (2003) Interstitial cystitis and chronic pelvic pain: new insights in neuropathology, diagnosis, and treatment. Clin Obstet Gynecol 46(4):811–823
Cattaneo A (2010) Tanezumab, a recombinant humanized mAb against nerve growth factor for the treatment of acute and chronic pain. Curr Opin Mol Ther 12(1):94–106
Cheppudira BP, Girard BM, Malley SE, Dattilio A, Schutz KC, May V, Vizzard MA (2009) Involvement of JAK-STAT signaling/function after cyclophosphamide-induced bladder inflammation in female rats. Am J Physiol Renal Physiol 297(4):F1038–F1044. https://doi.org/10.1152/ajprenal.00110.2009
Chuang YC, Fraser MO, Yu Y, Chancellor MB, de Groat WC, Yoshimura N (2001) The role of bladder afferent pathways in bladder hyperactivity induced by the intravesical administration of nerve growth factor. J Urol 165(3):975–979
Clemow DB, Steers WD, McCarty R, Tuttle JB (1998) Altered regulation of bladder nerve growth factor and neurally mediated hyperactive voiding. Am J Phys 275(4 Pt 2):R1279–R1286
Cox PJ (1979) Cyclophosphamide cystitis--identification of acrolein as the causative agent. Biochem Pharmacol 28:2045–2049
de Groat WC (1986) Spinal cord projections and neuropeptides in visceral afferent neurons. Prog Brain Res 67:165–187
de Groat WC (1990) Central neural control of the lower urinary tract. CIBA Found Symp 151:27–44 discussion 44–56
de Groat WC (1993) Anatomy and physiology of the lower urinary tract. Urol Clin North Am 20(3):383–401
DiCicco-Bloom E, Deutsch PJ, Maltzman J, Zhang J, Pintar JE, Zheng J, Friedman WF, Zhou X, Zaremba T (2000) Autocrine expression and ontogenetic functions of the PACAP ligand/receptor system during sympathetic development. Dev Biol 219(2):197–213. https://doi.org/10.1006/dbio.2000.9604
Dinarello CA (1997) Role of pro- and anti-inflammatory cytokines during inflammation: experimental and clinical findings. J Biol Regul Homeost Agents 11(3):91–103
Dmitrieva N, McMahon SB (1996) Sensitisation of visceral afferents by nerve growth factor in the adult rat. Pain 66(1):87–97
Dmitrieva N, Shelton D, Rice AS, McMahon SB (1997) The role of nerve growth factor in a model of visceral inflammation. Neuroscience 78(2):449–459
Donnerer J, Schuligoi R, Stein C (1992) Increased content and transport of substance P and calcitonin gene-related peptide in sensory nerves innervating inflamed tissue: evidence for a regulatory function of nerve growth factor in vivo. Neuroscience 49(3):693–698
Donovan MK, Winternitz SR, Wyss JM (1983) An analysis of the sensory innervation of the urinary system in the rat. Brain Res Bull 11(3):321–324
Dray A (1995) Inflammatory mediators of pain. Br J Anaesth 75(2):125–131
Driscoll A, Teichman JM (2001) How do patients with interstitial cystitis present? J Urol 166(6):2118–2120
Enerback L, Fall M, Aldenborg F (1989) Histamine and mucosal mast cells in interstitial cystitis. Agents Actions 27(1–2):113–116
Erickson DR, Belchis DA, Dabbs DJ (1997) Inflammatory cell types and clinical features of interstitial cystitis. J Urol 158(3 Pt 1):790–793
Evans RJ, Moldwin RM, Cossons N, Darekar A, Mills IW, Scholfield D (2011) Proof of concept trial of tanezumab for the treatment of symptoms associated with interstitial cystitis. J Urol 185(5):1716–1721. https://doi.org/10.1016/j.juro.2010.12.088
Fahrenkrug J, Hannibal J (1998) PACAP in visceral afferent nerves supplying the rat digestive and urinary tracts. Ann N Y Acad Sci 865:542–546
Fitzgerald MP, Koch D, Senka J (2005) Visceral and cutaneous sensory testing in patients with painful bladder syndrome. Neurourol Urodyn 24(7):627–632. https://doi.org/10.1002/nau.20178
Girard BA, Lelievre V, Braas KM, Razinia T, Vizzard MA, Ioffe Y, Meskini RE, Ronnett GV, Waschek JA, May V (2006) Noncompensation in peptide/receptor gene expression and distinct behavioral phenotypes in VIP- and PACAP-deficient mice. J Neurochem 99(2):499–513. https://doi.org/10.1111/j.1471-4159.2006.04112.x
Girard BM, Wolf-Johnston A, Braas KM, Birder LA, May V, Vizzard MA (2008) PACAP-mediated ATP release from rat urothelium and regulation of PACAP/VIP and receptor mRNA in micturition pathways after cyclophosphamide (CYP)-induced cystitis. J Mol Neurosci 36(1–3):310–320. https://doi.org/10.1007/s12031-008-9104-4
Girard BM, Malley SE, Braas KM, May V, Vizzard MA (2010) PACAP/VIP and receptor characterization in micturition pathways in mice with overexpression of NGF in urothelium. J Mol Neurosci 42(3):378–389. https://doi.org/10.1007/s12031-010-9384-3
Girard BM, Tooke K, Vizzard MA (2017) PACAP/receptor system in urinary bladder dysfunction and pelvic pain following urinary bladder inflammation or stress. Front Syst Neurosci 11:90. https://doi.org/10.3389/fnsys.2017.00090
Girard BM, Tompkins JD, Parsons RL, May V, Vizzard MA (2012) Effects of CYP-induced cystitis on PACAP/VIP and receptor expression in micturition pathways and bladder function in mice with overexpression of NGF in urothelium. J Mol Neurosci 48(3):730-43
Gonzalez EJ, Arms L, Vizzard MA (2014) The role(s) of cytokines/chemokines in urinary bladder inflammation and dysfunction. Biomed Res Int 2014:120525–120517. https://doi.org/10.1155/2014/120525
Grover S, Srivastava A, Lee R, Tewari AK, Te AE (2011) Role of inflammation in bladder function and interstitial cystitis. Ther Adv Urol 3(1):19–33. https://doi.org/10.1177/1756287211398255
Grumolato L, Louiset E, Alexandre D, Ait-Ali D, Turquier V, Fournier A et al (2003) PACAP and NGF regulate common and distinct traits of the sympathoadrenal lineage: effects on electrical properties, gene markers and transcription factors in differentiating PC12 cells. Eur J Neurosci 17(1):71–82
Guerios SD, Wang ZY, Bjorling DE (2006) Nerve growth factor mediates peripheral mechanical hypersensitivity that accompanies experimental cystitis in mice. Neurosci Lett 392(3):193–197. https://doi.org/10.1016/j.neulet.2005.09.026
Guerios SD, Wang ZY, Boldon K, Bushman W, Bjorling DE (2008) Blockade of NGF and trk receptors inhibits increased peripheral mechanical sensitivity accompanying cystitis in rats. Am J Physiol Regul Integr Comp Physiol 295(1):R111–R122. https://doi.org/10.1152/ajpregu.00728.2007
Hammack SE, May V (2015) Pituitary adenylate cyclase activating polypeptide in stress-related disorders: data convergence from animal and human studies. Biol Psychiatry 78(3):167–177. https://doi.org/10.1016/j.biopsych.2014.12.003
Hanno PM, Sant GR (2001) Clinical highlights of the National Institute of Diabetes and Digestive and Kidney Diseases/Interstitial Cystitis Association scientific conference on interstitial cystitis. Urology 57(6 Suppl 1):2–6
Hashimoto H, Ishihara T, Shigemoto R, Mori K, Nagata S (1993) Molecular cloning and tissue distribution of a receptor for pituitary adenylate cyclase-activating polypeptide. Neuron 11(2):333–342
Hashimoto H, Shintani N, Tanaka K, Mori W, Hirose M, Matsuda T, Sakaue M, Miyazaki JI, Niwa H, Tashiro F, Yamamoto K, Koga K, Tomimoto S, Kunugi A, Suetake S, Baba A (2001) Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP). Proc Natl Acad Sci U S A 98(23):13355–13360. https://doi.org/10.1073/pnas.231094498
Hattori S, Takao K, Tanda K, Toyama K, Shintani N, Baba A, Hashimoto H, Miyakawa T (2012) Comprehensive behavioral analysis of pituitary adenylate cyclase-activating polypeptide (PACAP) knockout mice. Front Behav Neurosci 6:58. https://doi.org/10.3389/fnbeh.2012.00058
Herrera GM, Braas KM, May V, Vizzard MA (2006) PACAP enhances mouse urinary bladder contractility and is upregulated in micturition reflex pathways after cystitis. Ann N Y Acad Sci 1070:330–336. https://doi.org/10.1196/annals.1317.040
Hosoya M, Onda H, Ogi K, Masuda Y, Miyamoto Y, Ohtaki T, Okazaki H, Arimura A, Fujino M (1993) Molecular cloning and functional expression of rat cDNAs encoding the receptor for pituitary adenylate cyclase activating polypeptide (PACAP). Biochem Biophys Res Commun 194(1):133–143. https://doi.org/10.1006/bbrc.1993.1795
Hu VY, Zvara P, Dattilio A, Redman TL, Allen SJ, Dawbarn D et al (2005) Decrease in bladder overactivity with REN1820 in rats with cyclophosphamide induced cystitis. J Urol 173(3):1016–1021. https://doi.org/10.1097/01.ju.0000155170.15023.e5
Huang EJ, Reichardt LF (2001) Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 24:677–736. https://doi.org/10.1146/annurev.neuro.24.1.677
Iannone F, De Bari C, Dell’Accio F, Covelli M, Patella V, Lo Bianco G et al (2002) Increased expression of nerve growth factor (NGF) and high affinity NGF receptor (p140 TrkA) in human osteoarthritic chondrocytes. Rheumatology (Oxford) 41(12):1413–1418
Inagaki N, Yoshida H, Mizuta M, Mizuno N, Fujii Y, Gonoi T, Miyazaki J, Seino S (1994) Cloning and functional characterization of a third pituitary adenylate cyclase-activating polypeptide receptor subtype expressed in insulin-secreting cells. Proc Natl Acad Sci U S A 91(7):2679–2683
Ishihara T, Shigemoto R, Mori K, Takahashi K, Nagata S (1992) Functional expression and tissue distribution of a novel receptor for vasoactive intestinal polypeptide. Neuron 8(4):811–819
Jaggar SI, Scott HC, Rice AS (1999) Inflammation of the rat urinary bladder is associated with a referred thermal hyperalgesia which is nerve growth factor dependent. Br J Anaesth 83(3):442–448
Jamen F, Bouschet T, Laden JC, Bockaert J, Brabet P (2002) Up-regulation of the PACAP type-1 receptor (PAC1) promoter by neurotrophins in rat PC12 cells and mouse cerebellar granule cells via the Ras/mitogen-activated protein kinase cascade. J Neurochem 82(5):1199–1207
Jongsma Wallin H, Danielsen N, Johnston JM, Gratto KA, Karchewski LA, Verge VM (2001) Exogenous NT-3 and NGF differentially modulate PACAP expression in adult sensory neurons, suggesting distinct roles in injury and inflammation. Eur J Neurosci 14(2):267–282
Kastrup J, Hald T, Larsen S, Nielsen VG (1983) Histamine content and mast cell count of detrusor muscle in patients with interstitial cystitis and other types of chronic cystitis. Br J Urol 55(5):495–500
Keast JR, De Groat WC (1992) Segmental distribution and peptide content of primary afferent neurons innervating the urogenital organs and colon of male rats. J Comp Neurol 319(4):615–623. https://doi.org/10.1002/cne.903190411
Kim JC, Kim DB, Seo SI, Park YH, Hwang TK (2004) Nerve growth factor and vanilloid receptor expression, and detrusor instability, after relieving bladder outlet obstruction in rats. BJU Int 94(6):915–918. https://doi.org/10.1111/j.1464-4096.2003.05059.x
Kim JC, Park EY, Hong SH, Seo SI, Park YH, Hwang TK (2005) Changes of urinary nerve growth factor and prostaglandins in male patients with overactive bladder symptom. Int J Urol 12(10):875–880. https://doi.org/10.1111/j.1442-2042.2005.01140.x
Kim JC, Park EY, Seo SI, Park YH, Hwang TK (2006) Nerve growth factor and prostaglandins in the urine of female patients with overactive bladder. J Urol 175(5):1773–1776; discussion 1776. https://doi.org/10.1016/S0022-5347(05)00992-4
Kimura C, Ohkubo S, Ogi K, Hosoya M, Itoh Y, Onda H, Miyata A, Jiang L, Dahl RR, Stibbs HH, Arimura A, Fujino M (1990) A novel peptide which stimulates adenylate cyclase: molecular cloning and characterization of the ovine and human cDNAs. Biochem Biophys Res Commun 166(1):81–89
Lamb K, Gebhart GF, Bielefeldt K (2004) Increased nerve growth factor expression triggers bladder overactivity. J Pain 5(3):150–156
Larsen JO, Hannibal J, Knudsen SM, Fahrenkrug J (1997) Expression of pituitary adenylate cyclase-activating polypeptide (PACAP) in the mesencephalic trigeminal nucleus of the rat after transsection of the masseteric nerve. Brain Res Mol Brain Res 46(1–2):109–117
Lee FS, Rajagopal R, Kim AH, Chang PC, Chao MV (2002) Activation of Trk neurotrophin receptor signaling by pituitary adenylate cyclase-activating polypeptides. J Biol Chem 277(11):9096–9102. https://doi.org/10.1074/jbc.M107421200
Levi-Montalcini R, Skaper SD, Dal Toso R, Petrelli L, Leon A (1996) Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci 19(11):514–520. https://doi.org/10.1016/S0166-2236(96)10058-8
Lindsay RM, Harmar AJ (1989) Nerve growth factor regulates expression of neuropeptide genes in adult sensory neurons. Nature 337(6205):362–364. https://doi.org/10.1038/337362a0
Liu HT, Kuo HC (2007) Intravesical botulinum toxin a injections plus hydrodistension can reduce nerve growth factor production and control bladder pain in interstitial cystitis. Urology 70(3):463–468. https://doi.org/10.1016/j.urology.2007.04.038
Liu HT, Kuo HC (2008) Urinary nerve growth factor levels are increased in patients with bladder outlet obstruction with overactive bladder symptoms and reduced after successful medical treatment. Urology 72(1):104–108; discussion 108. https://doi.org/10.1016/j.urology.2008.01.069
Liu HT, Kuo HC (2009) Urinary nerve growth factor levels are elevated in patients with overactive bladder and do not significantly increase with bladder distention. Neurourol Urodyn 28(1):78–81. https://doi.org/10.1002/nau.20599
Lowe EM, Anand P, Terenghi G, Williams-Chestnut RE, Sinicropi DV, Osborne JL (1997) Increased nerve growth factor levels in the urinary bladder of women with idiopathic sensory urgency and interstitial cystitis. Br J Urol 79(4):572–577
Lutz EM, Sheward WJ, West KM, Morrow JA, Fink G, Harmar AJ (1993) The VIP2 receptor: molecular characterisation of a cDNA encoding a novel receptor for vasoactive intestinal peptide. FEBS Lett 334(1):3–8
Maggi CA, Lecci A, Santicioli P, Del Bianco E, Giuliani S (1992) Cyclophosphamide cystitis in rats: involvement of capsaicin-sensitive primary afferents. J Auton Nerv Syst 38(3):201–208
May V, Braas KM (1995) Pituitary adenylate cyclase-activating polypeptide (PACAP) regulation of sympathetic neuron neuropeptide Y and catecholamine expression. J Neurochem 65(3):978–987
May V, Parsons RL (2017) G protein-coupled receptor endosomal signaling and regulation of neuronal excitability and stress responses: signaling options and lessons from the PAC1 receptor. J Cell Physiol 232(4):698–706. https://doi.org/10.1002/jcp.25615
May V, Vizzard MA (2010) Bladder dysfunction and altered somatic sensitivity in PACAP−/− mice. J Urol 183(2):772–779. https://doi.org/10.1016/j.juro.2009.09.077
May V, Beaudet MM, Parsons RL, Hardwick JC, Gauthier EA, Durda JP et al (1998) Mechanisms of pituitary adenylate cyclase activating polypeptide (PACAP)-induced depolarization of sympathetic superior cervical ganglion (SCG) neurons. Ann N Y Acad Sci 865:164–175
Merrill L, Gonzalez EJ, Girard BM, Vizzard MA (2016) Receptors, channels, and signalling in the urothelial sensory system in the bladder. Nat Rev Urol 13(4):193–204. https://doi.org/10.1038/nrurol.2016.13
Missig G, Roman CW, Vizzard MA, Braas KM, Hammack SE, May V (2014) Parabrachial nucleus (PBn) pituitary adenylate cyclase activating polypeptide (PACAP) signaling in the amygdala: implication for the sensory and behavioral effects of pain. Neuropharmacology 86:38–48. https://doi.org/10.1016/j.neuropharm.2014.06.022
Missig G, Mei L, Vizzard MA, Braas KM, Waschek JA, Ressler KJ, Hammack SE, May V (2017) Parabrachial pituitary adenylate cyclase-activating polypeptide activation of amygdala endosomal extracellular signal-regulated kinase signaling regulates the emotional component of pain. Biol Psychiatry 81(8):671–682. https://doi.org/10.1016/j.biopsych.2016.08.025
Mizumura K, Murase S (2015) Role of nerve growth factor in pain. Handb Exp Pharmacol 227:57–77. https://doi.org/10.1007/978-3-662-46450-2_4
Moller K, Reimer M, Hannibal J, Fahrenkrug J, Sundler F, Kanje M (1997) Pituitary adenylate cyclase-activating peptide (PACAP) and PACAP type 1 receptor expression in regenerating adult mouse and rat superior cervical ganglia in vitro. Brain Res 775(1–2):156–165
Mullins C, Bavendam T, Kirkali Z, Kusek JW (2015) Novel research approaches for interstitial cystitis/bladder pain syndrome: thinking beyond the bladder. Transl Androl Urol 4(5):524–533. https://doi.org/10.3978/j.issn.2223-4683.2015.08.01
Nazif O, Teichman JM, Gebhart GF (2007) Neural upregulation in interstitial cystitis. Urology 69(4 Suppl):24–33. https://doi.org/10.1016/j.urology.2006.08.1108
Ogi K, Kimura C, Onda H, Arimura A, Fujino M (1990) Molecular cloning and characterization of cDNA for the precursor of rat pituitary adenylate cyclase activating polypeptide (PACAP). Biochem Biophys Res Commun 173(3):1271–1279
Ohkubo S, Kimura C, Ogi K, Okazaki K, Hosoya M, Onda H et al (1992) Primary structure and characterization of the precursor to human pituitary adenylate cyclase activating polypeptide. DNA Cell Biol 11(1):21–30. https://doi.org/10.1089/dna.1992.11.21
Okazaki K, Itoh Y, Ogi K, Ohkubo S, Onda H (1995) Characterization of murine PACAP mRNA. Peptides 16(7):1295–1299
Okragly AJ, Niles AL, Saban R, Schmidt D, Hoffman RL, Warner TF et al (1999) Elevated tryptase, nerve growth factor, neurotrophin-3 and glial cell line-derived neurotrophic factor levels in the urine of interstitial cystitis and bladder cancer patients. J Urol 161(2):438–441 discussion 441–432
Otto C, Martin M, Wolfer DP, Lipp HP, Maldonado R, Schutz G (2001) Altered emotional behavior in PACAP-type-I-receptor-deficient mice. Brain Res Mol Brain Res 92(1–2):78–84
Parsons CL (2007) The role of the urinary epithelium in the pathogenesis of interstitial cystitis/prostatitis/urethritis. Urology 69(4 Suppl):9–16. https://doi.org/10.1016/j.urology.2006.03.084
Paterson S, Schmelz M, McGlone F, Turner G, Rukwied R (2009) Facilitated neurotrophin release in sensitized human skin. Eur J Pain 13(4):399–405. https://doi.org/10.1016/j.ejpain.2008.05.005
Patnaik SS, Lagana AS, Vitale SG, Buttice S, Noventa M, Gizzo S et al (2017) Etiology, pathophysiology and biomarkers of interstitial cystitis/painful bladder syndrome. Arch Gynecol Obstet 295(6):1341–1359. https://doi.org/10.1007/s00404-017-4364-2
Persu C, Cauni V, Gutue S, Blaj I, Jinga V, Geavlete P (2010) From interstitial cystitis to chronic pelvic pain. J Med Life 3(2):167–174
Pezet S, McMahon SB (2006) Neurotrophins: mediators and modulators of pain. Annu Rev Neurosci 29:507–538. https://doi.org/10.1146/annurev.neuro.29.051605.112929
Pohlack ST, Nees F, Ruttorf M, Cacciaglia R, Winkelmann T, Schad LR, Witt SH, Rietschel M, Flor H (2015) Neural mechanism of a sex-specific risk variant for posttraumatic stress disorder in the type I receptor of the pituitary adenylate cyclase activating polypeptide. Biol Psychiatry 78(12):840–847. https://doi.org/10.1016/j.biopsych.2014.12.018
Qiao LY, Vizzard MA (2002) Cystitis-induced upregulation of tyrosine kinase (TrkA, TrkB) receptor expression and phosphorylation in rat micturition pathways. J Comp Neurol 454(2):200–211. https://doi.org/10.1002/cne.10447
Qiao LY, Vizzard MA (2004) Up-regulation of phosphorylated CREB but not c-Jun in bladder afferent neurons in dorsal root ganglia after cystitis. J Comp Neurol 469(2):262–274
Ressler KJ, Mercer KB, Bradley B, Jovanovic T, Mahan A, Kerley K, Norrholm SD, Kilaru V, Smith AK, Myers AJ, Ramirez M, Engel A, Hammack SE, Toufexis D, Braas KM, Binder EB, May V (2011) Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature 470(7335):492–497. https://doi.org/10.1038/nature09856
Ritter AM, Lewin GR, Kremer NE, Mendell LM (1991) Requirement for nerve growth factor in the development of myelinated nociceptors in vivo. Nature 350(6318):500–502. https://doi.org/10.1038/350500a0
Sant GR, Hanno PM (2001) Interstitial cystitis: current issues and controversies in diagnosis. Urology 57(6 Suppl 1):82–88
Sant GR, Kempuraj D, Marchand JE, Theoharides TC (2007) The mast cell in interstitial cystitis: role in pathophysiology and pathogenesis. Urology 69(4 Suppl):34–40. https://doi.org/10.1016/j.urology.2006.08.1109
Sarchielli P, Mancini ML, Floridi A, Coppola F, Rossi C, Nardi K, Acciarresi M, Pini LA, Calabresi P (2007) Increased levels of neurotrophins are not specific for chronic migraine: evidence from primary fibromyalgia syndrome. J Pain 8(9):737–745. https://doi.org/10.1016/j.jpain.2007.05.002
Schnegelsberg B, Sun TT, Cain G, Bhattacharya A, Nunn PA, Ford AP et al (2010) Overexpression of NGF in mouse urothelium leads to neuronal hyperinnervation, pelvic sensitivity, and changes in urinary bladder function. Am J Physiol Regul Integr Comp Physiol 298(3):R534–R547. https://doi.org/10.1152/ajpregu.00367.2009
Spengler D, Waeber C, Pantaloni C, Holsboer F, Bockaert J, Seeburg PH et al (1993) Differential signal transduction by five splice variants of the PACAP receptor. Nature 365(6442):170–175. https://doi.org/10.1038/365170a0
Stroth N, Eiden LE (2010) Stress hormone synthesis in mouse hypothalamus and adrenal gland triggered by restraint is dependent on pituitary adenylate cyclase-activating polypeptide signaling. Neuroscience 165(4):1025–1030. https://doi.org/10.1016/j.neuroscience.2009.11.023
Studeny S, Cheppudira BP, Meyers S, Balestreire EM, Apodaca G, Birder LA, Braas KM, Waschek JA, May V, Vizzard MA (2008) Urinary bladder function and somatic sensitivity in vasoactive intestinal polypeptide (VIP)−/− mice. J Mol Neurosci 36(1–3):175–187. https://doi.org/10.1007/s12031-008-9100-8
Theoharides TC, Sant GR, el-Mansoury M, Letourneau R, Ucci AA Jr, Meares EM Jr (1995) Activation of bladder mast cells in interstitial cystitis: a light and electron microscopic study. J Urol 153(3 Pt 1):629–636
Tsukiyama N, Saida Y, Kakuda M, Shintani N, Hayata A, Morita Y, Tanida M, Tajiri M, Hazama K, Ogata K, Hashimoto H, Baba A (2011) PACAP centrally mediates emotional stress-induced corticosterone responses in mice. Stress 14(4):368–375. https://doi.org/10.3109/10253890.2010.544345
Vaudry D, Falluel-Morel A, Bourgault S, Basille M, Burel D, Wurtz O, Fournier A, Chow BKC, Hashimoto H, Galas L, Vaudry H (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61(3):283–357. https://doi.org/10.1124/pr.109.001370
Vizzard MA (2000a) Changes in urinary bladder neurotrophic factor mRNA and NGF protein following urinary bladder dysfunction. Exp Neurol 161(1):273–284. https://doi.org/10.1006/exnr.1999.7254
Vizzard MA (2000b) Up-regulation of pituitary adenylate cyclase-activating polypeptide in urinary bladder pathways after chronic cystitis. J Comp Neurol 420(3):335–348
Vizzard MA (2001a) Alterations in neuropeptide expression in lumbosacral bladder pathways following chronic cystitis. J Chem Neuroanat 21(2):125–138
Vizzard MA, Erdman SL, Forstermann U, de Groat WC (1994) Ontogeny of nitric oxide synthase in the lumbosacral spinal cord of the neonatal rat. Brain Res Dev Brain Res 81(2):201–217
Woolf CJ, Allchorne A, Safieh-Garabedian B, Poole S (1997) Cytokines, nerve growth factor and inflammatory hyperalgesia: the contribution of tumour necrosis factor alpha. Br J Pharmacol 121(3):417–424. https://doi.org/10.1038/sj.bjp.0701148
Yoshimura N, de Groat WC (1999) Increased excitability of afferent neurons innervating rat urinary bladder after chronic bladder inflammation. J Neurosci 19(11):4644–4653
Yoshimura N, Bennett NE, Hayashi Y, Ogawa T, Nishizawa O, Chancellor MB, de Groat WC, Seki S (2006) Bladder overactivity and hyperexcitability of bladder afferent neurons after intrathecal delivery of nerve growth factor in rats. J Neurosci 26(42):10847–10855. https://doi.org/10.1523/JNEUROSCI.3023-06.2006
Yu Y, de Groat WC (2008) Sensitization of pelvic afferent nerves in the in vitro rat urinary bladder-pelvic nerve preparation by purinergic agonists and cyclophosphamide pretreatment. Am J Physiol Renal Physiol 294(5):F1146–F1156. https://doi.org/10.1152/ajprenal.00592.2007
Zhang Q, Shi TJ, Ji RR, Zhang YZ, Sundler F, Hannibal J et al (1995) Expression of pituitary adenylate cyclase-activating polypeptide in dorsal root ganglia following axotomy: time course and coexistence. Brain Res 705(1–2):149–158
Zhang YZ, Hannibal J, Zhao Q, Moller K, Danielsen N, Fahrenkrug J, Sundler F (1996) Pituitary adenylate cyclase activating peptide expression in the rat dorsal root ganglia: up-regulation after peripheral nerve injury. Neuroscience 74(4):1099–1110
Zvara P, Vizzard MA (2007) Exogenous overexpression of nerve growth factor in the urinary bladder produces bladder overactivity and altered micturition circuitry in the lumbosacral spinal cord. BMC Physiol 7:9. https://doi.org/10.1186/1472-6793-7-9
Funding
Research described herein was funded by the National Institutes of Health (NIH) Grants RO1-DK051369 (MAV) and RO1-DK060481 (MAV). This publication was also made possible by NIH Grants: 5 P30 RR032135 from the COBRE Program of the National Center for Research Resources and 8 P30 GM103498 from the National Institute of General Medical Sciences.
Author information
Authors and Affiliations
Contributions
K.T., J.O., B.G., H.H., V.M., and M.V. conceived, discussed, and outlined the review. K.T., J.O., B.G., H.H., V.M., and M.V. drafted and revised the paper.
Corresponding author
Ethics declarations
Studies Involving Animal Research
The studies described from the Vizzard laboratory were performed in accordance with institutional and national guidelines and regulations. The University of Vermont Institutional Animal Care and Use Committee approved all experimental protocols involving animal use. Animal care was under the supervision of the University of Vermont’s Office of Animal Care Management in accordance with the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) and National Institutes of Health guidelines. All efforts were made to minimize the potential for animal pain, stress, or distress.
Conflicts of Interest
The authors declare that the research described from the Vizzard laboratory were conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The funding entity, NIH, had no role in the studies described including: design, data collection, and analysis of studies performed in the Vizzard laboratory, decision to publish, or preparation of the review. The contents are solely the responsibility of the authors and do not necessarily represent the official views of NIH.
Rights and permissions
About this article
Cite this article
Ojala, J., Tooke, K., Hsiang, H. et al. PACAP/PAC1 Expression and Function in Micturition Pathways. J Mol Neurosci 68, 357–367 (2019). https://doi.org/10.1007/s12031-018-1170-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1170-7