Abstract
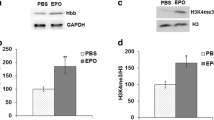
Multiple sclerosis (MS) is characterized by demyelination and progressive neurological disability. Previous studies have reported defects to mitochondria in MS including decreased expression of nuclear encoded electron transport chain subunit genes and inhibition of respiratory complexes. We previously reported increased levels of the hemoglobin β subunit (Hbb) in mitochondrial fractions isolated from postmortem MS cortex compared to controls. In the present study, we performed immunohistochemistry to determine the distribution of Hbb in postmortem MS cortex and identified proteins which interact with Hbb by liquid chromatography tandem mass spectrometry (LC-MS/MS). We found that Hbb was enriched in pyramidal neurons in internal layers of the cortex and interacts with subunits of ATP synthase, histones, and a histone lysine demethylase. We also found that Hbb is present in the nucleus and that expression of Hbb in SH-SY5Y neuroblastoma cells increased trimethylation of histone H3 on lysine 4 (H3K4me3), a histone mark that regulates cellular metabolism. These data suggest that Hbb may be a part of a mechanism linking neuronal energetics with epigenetic changes to histones in the nucleus and may provide neuroprotection in MS by supporting neuronal metabolism.





Similar content being viewed by others
References
Bellelli A, Brunori M, Miele AE, Panetta G, Vallone B (2006) The allosteric properties of hemoglobin: insights from natural and site directed mutants. Curr Protein Pept Sci 7:17–45
Benavides-Piccione R, DeFelipe J (2007) Distribution of neurons expressing tyrosine hydroxylase in the human cerebral cortex. J Anat 211(2):212–22
Biagioli M, Pinto M, Cesselli D, Zaninello M, Lazarevic D, Roncaglia P, Simone R, Vlachouli C, Plessy C, Bertin N, Beltrami A, Kobayashi K, Gallo V, Santoro C, Ferrer I, Rivella S, Beltrami CA, Carninci P, Raviola E, Gustincich S (2009) Unexpected expression of α- and β-globin in mesencephalic dopaminergic neurons and glial cells. Proc Natl Acad Sci U S A 106:15454–15459
Bjartmar C, Kidd G, Mörk S, Rudick R, Trapp BD (2000) Neurological disability correlates with spinal cord axonal loss and reduced N-acetyl-aspartate in chronic multiple sclerosis patients. Ann Neurol 48:893–901
Bö L, Geurts JJ, Mörk SJ, van der Valk P (2006) Grey matter pathology in multiple sclerosis. Acta Neurol Scand Suppl 183:48–50
Broadwater L, Pandit A, Azzam S, Clements R, Vadnal J, Yong VW, Freeman EJ, Gregory RB, McDonough J (2011) Analysis of the mitochondrial proteome in multiple sclerosis cortex. Biochim Biophys Acta 1812:630–641
Cader S, Johansen-Berg H, Wylezinska M, Palace J, Behrens TE, Smith S, Matthews PM (2007) Discordant white matter N-acetylasparate and diffusion MRI measures suggest that chronic metabolic dysfunction contributes to axonal pathology in multiple sclerosis. Neuroimage 36(1):19–27
DeFelipe J, Farinas I (1992) The pyramidal neuron of the cerebral cortex: morphological and chemical characteristics of the synaptic inputs. Prog Neurobiol 39(6):563–607
De Stefano N, Narayanan S, Francis GS, Arnaoutelis R, Tartaglia MC, Antel JP, Matthews PM, Arnold DL (2001) Evidence of axonal damage in the early stages of multiple sclerosis and its relevance to disability. Arch Neurol 58(1):65–70
Dutta R, McDonough J, Yin X, Peterson J, Chang A, Torres T, Gudz T, Macklin WB, Lewis DA, Fox RJ, Rudick R, Mirnics K, Trapp BD (2006) Mitochondrial dysfunction as a cause of axonal degeneration in multiple sclerosis patients. Ann Neurol 59:478–489
England JD, Gamboni F, Levinson SR (1991) Increased numbers of sodium channels form along demyelinated axons. Brain Res 548(1-2):334–7
Fisher E, Lee JC, Nakamura K, Rudick RA (2008) Gray matter atrophy in multiple sclerosis: a longitudinal study. Ann Neurol 64(3):255–65
Ge Y, Gonen O, Inglese M, Babb JS, Markowitz CE, Grossman RI (2004) Neuronal cell injury precedes brain atrophy in multiple sclerosis. Neurology 62(4):624–7
Gut P, Verdin E (2013) The nexus of chromatin regulation and intermediary metabolism. Nature 502(7472):489–98. doi:10.1038/nature12752, Review
Inglese M, Ge Y, Filippi M, Falini A, Grossman RI, Gonen O (2004) Indirect evidence for early widespread gray matter involvement in relapsing-remitting multiple sclerosis. Neuroimage 21:1825–1829
Li S, Clements R, Sulak M, Gregory R, Freeman E, McDonough J (2013) Decreased NAA in gray matter is correlated with decreased availability of acetate in white matter in postmortem multiple sclerosis cortex. Neurochem Res 38(11):2385–96
Liu L, Zeng M, Stamler JS (1999) Hemoglobin induction in mouse macrophages. Proc Natl Acad Sci U S A 96(12):6643–7
Nishi H, Inagi R, Kato H, Tanemoto M, Kojima I, Son D, Fujita T, Nangaku M (2008) Hemoglobin is expressed by mesangial cells and reduces oxidant stress. J Am Soc Nephrol 19(8):1500–8
Noseworthy JH, Lucchinetti C, Rodriguez M, Weinshenker BG (2000) Multiple sclerosis. N Engl J Med 343(13):938–52
Pallotti F, Lenaz G (2007) Isolation and subfractionation of mitochondria from animal cells and tissue culture lines. Methods Cell Biol 80:3–44
Pandit A, Vadnal J, Houston S, Freeman E, McDonough J (2009) Impaired regulation of electron transport chain subunit genes by nuclear respiratory factor 2 in multiple sclerosis. J Neurol Sci 279:14–20
Rahaman MM, Straub AC (2013) The emerging roles of somatic globins in cardiovascular redox biology and beyond. Redox Biol 1:405–10
Rasband WS (1997-2014) ImageJ, U. S. National institutes of health, Bethesda, Maryland, USA, http://imagej.nih.gov/ij/
Reeder BJ (2010) The redox activity of hemoglobins: from physiologic functions to pathologic mechanisms. Antioxid Redox Signal 13(7):1087–123. doi:10.1089/ars.2009.2974
Richter F, Meurers BH, Zhu C, Medvedeva VP, Chesselet M-F (2009) Neurons express hemoglobin α- and β-chains in rat and human brains. J Comp Neurol 515:538–547
Salminen A, Kauppinen A, Hiltunen M, Kaarniranta K (2014) Krebs cycle intermediates regulate DNA and histone methylation: epigenetic impact on the aging process. Ageing Res Rev 16:45–65
Schelshorn DW, Schneider K, Weber AW, Kruger D, Dittgen C, Burgers HF, Sabouri F, Gassler N, Bach A, Martin H, Maurer MH (2009) Expression of hemoglobin in rodent neurons. J Cerebr Blood Flow Metab 29:585–595
Shephard F, Greville-Heygate O, Marsh O, Anderson S, Chakrabarti L (2014) A mitochondrial location for haemoglobins—dynamic distribution in ageing and Parkinson’s disease. Mitochondrion 14(1):64–72. doi:10.1016/j.mito.2013.12.001
Singhal NK, Li S, Arning E, Alkhayer K, Clements R, Sarcyk Z, Dassanayake RS, Brasch NE, Freeman EJ, Bottiglieri T, McDonough J (2015) Changes in methionine metabolism and histone H3 Trimethylation are linked to mitochondrial defects in multiple sclerosis. J Neurosci 35(45):15170–15186
Straub AC, Lohman AW, Billaud M, Johnstone SR, Dwyer ST, Lee MY, Bortz PS, Best AK, Columbus L, Gaston B, Isakson BE (2012) Endothelial cell expression of haemoglobin α regulates nitric oxide signalling. Nature 491(7424):473–7
Vissers MC, Kuiper C, Dachs GU (2014) Regulation of the 2-oxoglutarate-dependent dioxygenases and implications for cancer. Biochem Soc Trans 42(4):945–51
Waxman SG (2006) Ions, energy and axonal injury: towards a molecular neurology of multiple sclerosis. Trends Mol Med 12(5):192–5
Witte ME, Nijland PG, Drexhage JA, Gerritsen W, Geerts D, van Het Hof B, Reijerkerk A, de Vries HE, van der Valk P, van Horssen J (2013) Reduced expression of PGC-1α partly underlies mitochondrial changes and correlates with neuronal loss in multiple sclerosis cortex. Acta Neuropathol 125(2):231–43
Acknowledgments
We would like to thank the Rocky Mountain MS Center which is funded by the National Multiple Sclerosis Society and the Brain and Spinal Cord Resource Center at UCLA for tissue. This research was partially funded by NIH Grant R21NS075645 (JM and EF) and from funds from the College of Arts and Sciences at Kent State University (JM).
Author information
Authors and Affiliations
Corresponding author
Additional information
Nolan Brown and Kholoud Alkhayer contributed equally to this work.
Rights and permissions
About this article
Cite this article
Brown, N., Alkhayer, K., Clements, R. et al. Neuronal Hemoglobin Expression and Its Relevance to Multiple Sclerosis Neuropathology. J Mol Neurosci 59, 1–17 (2016). https://doi.org/10.1007/s12031-015-0711-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-015-0711-6