Abstract
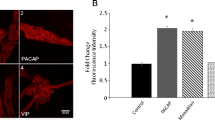
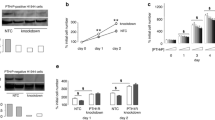
The signal transduction mechanisms of pituitary adenylate cyclase activating polypeptide (PACAP) were investigated in lung cancer cells. Previously, PACAP-27 addition to NCI-H838 cells increased phosphatidylinositol turnover and intracellular cAMP leading to proliferation of lung cancer cells. Also, PACAP receptors (PAC1) regulated the tyrosine phosphorylation of ERK, focal adhesion kinase, and paxillin. In this communication, the effects of PACAP on cytosolic Ca2+ and PYK-2 tyrosine phosphorylation were investigated. PACAP-27 increased cytosolic Ca2+ within seconds after addition to FURA-2 AM loaded NCI-H838 cells. The increase in cytosolic Ca2+ caused by PACAP was inhibited by PACAP(6–38) (PAC1 antagonist), U73122 (phospholipase C inhibitor), or BAPTA (calcium chelator), but not H89 (PKA inhibitor). PACAP-38, but not vasoactive intestinal peptide (VIP), addition to NCI-H838 or H1299 cells significantly increased the tyrosine phosphorylation of PYK-2 after 2 min. The increase in PYK-2 tyrosine phosphorylation caused by PACAP was inhibited by PACAP(6–38), U73122, or BAPTA, but not H89. The results suggest that PAC1 regulates PYK-2 tyrosine phosphorylation in a calcium-dependent manner.




Similar content being viewed by others
Abbreviations
- PACAP:
-
Pituitary adenylate cyclase activating polypeptide
- PYK-2:
-
Proline-rich tyrosine kinase 2
- FAK:
-
Focal adhesion kinase
- R:
-
Receptor
- SCLC:
-
Small cell lung cancer
- NSCLC:
-
Non-SCLC
- VIP:
-
Vasoactive intestinal peptide
- Ca2+ :
-
Calcium
- GPCR:
-
G-protein coupled receptor
- pY:
-
Phosphotyrosine
References
Bellis SL, Perotta JA, Curtis MS, Turner CE (1997) Adhesion of fibroblasts to fibronectin stimulates both serine and tyrosine phosphorylation of paxillin. Biochem J 325:375–381
Calaib MB, Polte TR, Hanks SK (1995) Tyrosine phosphorylation of focal adhesion kinase at sites in the catalytic domain regulates kinase activity. Mol Cell Biol 15:954–963
Dikic I, Tokiwa G, Lev S, Courtneidge SA, Schlessinger J (1996) A role for Pyk2 and Src in linking G-protein-coupled receptors with MAP kinase activation. Nature 383:547–550
Halder J, Lin YG, Merritt WM, Spannuth WA, Nick AM, Honda T, Kamat AA, Han LY, Kim TJ, Lu C, Tari AM, Bornmann W, Fernandez A, Lopez-Berestein G, Sood AK (2007) Therapeutic efficacy of a novel focal adhesion kinase inhibitor TAE226 in ovarian carcinoma. Cancer Res 67:10976–10983
Hall JE, Fu W, Schaller MD (2011) Focal adhesion kinase: exploring FAK structure to gain insight into function. Int Rev Cell Mol Biol 288:185–225
Ishihara T, Shigemoto R, Mori K, Takahashi K, Nagata S (1992) Functional expression and tissue distribution of a novel receptor for vasoactive intestinal polypeptide. Neuron 8:811–819
Kuwabara K, Nakaoka T, Sato K, Nishishita T, Sasaki T, Yamashita N (2004) Differential regulation of cell migration and proliferation through proline-rich tyrosine kinase 2 in endothelial cells. Endocrinology 145:3324–3330
Lee M, Jensen RT, Bepler G, Korman LY, Moody TW (1990) Vasoactive intestinal polypeptide binds with high affinity to non-small cell lung cancer cells and elevates cAMP” levels. Peptides 11:1205–1210
Lev S, Moreno H, Martinez R, Canoll P, Peles E, Musacchio JM, Plowman GD, Rudy B, Schlessinger JA (1995) Protein tyrosine kinase PYK2 involved in Ca(2+)-induced regulation of ion channel and MAP kinase functions. Nature 376:737–745
Lipinski CA, Loftus JC (2010) Targeting Pyk2 for therapeutic intervention. Expert Opin Ther Targets 14:95–108
Lipinski CA, Tran NL, Menashi E, Rohl C, Kloss J, Bay RC, Berens ME, Loftus JC (2005) The tyrosine kinase PYK2 promotes migration and invasion of glioma cells. Neoplasia 7:435–445
Liu TJ, LaFortune T, Honda T, Ohmori O, Hatakeyama S, Meyer T, Jackson D, deGroot J, Yung WK (2007) Inhibition of both focal adhesion kinase and insulin-like growth factor-I receptor kinase suppresses glioma proliferation in vitro and in vivo. Mol Cancer Ther 6:1357–1367
Lutz EM, Sheward WJ, West KM, Morrow JA, Harmar AJ (1993) The VIP2 receptor molecular characterization of a cDNA encoding a novel receptor for vasoactive intestinal peptide. FEBS Lett 334:3–8
May V, Lutz E, MacKenzie C, Schutz K, Dozark K, Braas K (2010) Pituitary adenylate cyclase-activating polypeptide (PACAP)/PAC1HOP1 receptor activation coordinates multiple neurotophic signaling pathways. J Biol Chem 285:9749–9761
Moody TW, Jensen RT (2006) VIP and PACAP as autocrine growth factors in breast and lung cancer. In: Kastin A (ed) Handbook of biologically active peptides. Elsevier, Amsterdam, pp 493–498
Moody TW, Leyton J, Chan D, Brenneman DC, Fridkin M, Gelber E, Levy A, Gozes I (2001) VIP receptor antagonists potentiate the action of chemotherapeutic drugs on breast cancer cells. Breast Cancer Res Treat 68:55–64
Moody TW, Leyton J, Casibang M, Pisegna J, Jensen RT (2002) PACAP-27 tyrosine phosphorylates mitogen activated protein kinase and increases VEGF mRNAs in human lung cancer cells. Regul Pept 109:135–140
Moody TW, Chan D, Fahrenkrug J, Jensen RT (2003a) Neuropeptides as autocrine growth factors in cancer cells. Curr Pharm Des 9:495–509
Moody TW, Hill JM, Jensen RT (2003b) VIP as a trophic factor in the CNS and cancer cells. Peptides 24:163–177
Moody TW, Leyton J, Jensen RT (2012a) Pituitary adenylate cyclase-activating polypeptide causes increased tyrosine phosphorylation of focal adhesion kinase and paxillin. J Mol Neurosci 46:68–74
Moody TW, Osefo N, Nuche-Berenguer B, Ridnour L, Wink D, Jensen RT (2012b) Pituitary adenylate cyclase activating polypeptide causes tyrosine phosphorylation of the EGF receptor in lung cancer cells. J Pharm Exp Ther [Epub ahead of print]
Pace A, Garcia-Marin LJ, Tapia JA, Bragado MJ, Jensen RT (2003) Phosphospecific site tyrosine phosphorylation of p125FAK and proline-rich kinase 2 is differentially regulated by cholecystokinin receptor type A activation in pancreatic acini. J Biol Chem 278:19008–19016
Picascia A, Stanzione R, Chieffi P, Kisslinger A, Dikic I, Tramontano D (2002) Proline-rich tyrosine kinase 2 regulates proliferation and differentiation of prostate cells. Mol Cell Endocrinol 186:81–87
Pisegna J, Wank SA (1993) Molecular cloning and functional expression of the pituitary adenylate cyclase activating polypeptide type I receptor. Proc Natl Acad Sci USA 90:6345–6349
Pisegna JR, Wank SA (1996) Cloning and characterization of the signal transduction of four splice variants of the human pituitary adenylate cyclase activating polypeptide receptor. Evidence for dual coupling to adenylate cyclase and phospholipase C. J Biol Chem 271:17267–17274
Reubi JC, Laderach U, Waser B, Gebbers JD, Robberecht P, Laissue JA (2000) Vasoactive intestinal peptide/pituitary adenylate cyclase activating polypeptide receptor subtypes in human tumors and their tissues of origin. Cancer Res 60:3105–3112
Roberts WG, Ung E, Whalen P, Cooper B, Hulford C, Autry C, Richter D, Emerson E, Lin J, Kath J, Coleman K, Yao L, Martinez-Alsina L, Lorenzen M, Berliner M, Luzzio M, Patel N, Schmitt E, LaGreca S, Jani J, Wessel M, Marr E, Griffor M, Vasdos F (2008) Antitumor activity and pharmacology of a selective focal adhesion kinase inhibitor, PF-562,271. Cancer Res 68:1935–1944
Rolle S, Grosse R, Buech T, Chubanov V, Gudermann T (2008) Essential role of Pyk2 and Src kinase activation in neuropeptide-induced proliferation of small cell lung cancer cells. Oncogene 27:1737–1748
Schaller MD, Borgman CA, Cobb BS, Vaines RR, Reynolds AB, Parsons JT (1992) P125FAK, a structurally distinctive protein tyrosine kinase associated with focal adhesions. Proc Natl Acad Sci USA 89:192–196
Shedden K, Taylor JM, Enkemann SA, Tsao MS, Yeatman TJ, Gerald WL, Eschrich S, Jurisica I, Giordano TJ, Misek DE, Chang AC, Zhu CQ, Strumpf D, Hanash S, Shepherd FA, Ding K, Seymour L, Naoki K, Pennell N, Weir B, Verhaak R, Ladd-Acosta C, Golub T, Gruidl M, Sharma A, Szoke J, Zakowski M, Rusch V, Kris M, Viale A, Motoi N, Travis W, Conley B, Seshan VE, Meyerson M, Kuick R, Dobbin KK, Lively T, Jacobson JW, Beer DG (2008) Gene expression-based survival prediction in lung adenocarcinoma: a multi-site blinded validation study. Nat Med 14:822–827
Spengler D, Waeber C, Pantaloni C, Holsboer F, Bockaert J, Seeburg PH, Journot L (1993) Differential signal transduction by five splice variants of the PACAP receptor. Nature 365:170–175
Tapia JA, Ferris HA, Jensen RT, Garcia LJ (1999) Cholecystokinin activates PYK2/CAKβ by a phospholipase C-dependent mechanism and its association with the mitogen-activated protein kinase signaling pathway in pancreatic acinar cells. J Biol Chem 274:31261–31271
Wittau N, Grosse R, Kalkbrenner F, Gohla A, Schultz G, Gudermann T (2000) The galanin receptor type 2 initiates multiple signaling pathways in small cell lung cancer cells by coupling to Gq, Gi and G12 proteins. Oncogene 19:1318–1328
Zhang S, Qiu X, Gu Y, Wang E (2008) Up-regulation of proline-rich tyrosine kinase 2 in non-small cell lung cancer. Lung Cancer 62:295–301
Zia F, Fagarasan M, Bitar K, Coy DH, Pisegna J, Wank S, Moody TW (1995) PACAP receptors regulate the growth of non-small cell lung cancer cells. Cancer Res 55:4886–4891
Zrihan-Licht S, Fu Y, Settleman J, Schinkmann K, Shaw L, Keydar I, Avraham S, Avraham H (2000) RAFTK/PYK2 tyrosine kinase mediates the association of P190 RhoGAP with RasGAP and is involved in breast cancer cell invasion. Oncogene 19:1318–28
Acknowledgment
This research is supported in part by intramural funds of the NCI and NIDDK of NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moody, T.W., Di Florio, A. & Jensen, R.T. PYK-2 is Tyrosine Phosphorylated after Activation of Pituitary Adenylate Cyclase Activating Polypeptide Receptors in Lung Cancer Cells. J Mol Neurosci 48, 660–666 (2012). https://doi.org/10.1007/s12031-012-9785-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9785-6