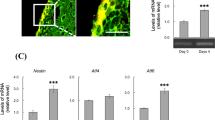
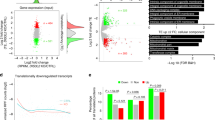
Abstract
The role of endoplasmic reticulum (ER) homeostasis and protein quality control in the regulation of WNT signaling is not understood. Here we provide evidence for a role of calreticulin in the regulation of WNT signaling. We show that a deficiency in calreticulin disrupted WNT signaling, and prevented cell cycle progression via the miR-302 microRNA family. These effects were dependent on the Ca2+ buffering capacity of calreticulin, as the protein is important in regulating ER Ca2+ release and activation of Ca2+-dependent kinase and phosphatase cascades (including c-Src, Akt, and PTP1B). We also show that calreticulin plays a role in the secretion and ER retention of WNT3a, thereby affecting downstream WNT signaling. In calreticulin-deficient ES cells, the WNT and miR-302 dependent maintenance of the naïve ES cell state and the transition to primed pluripotency transition were lost, preventing cells from undergoing accurate differentiation. Together, these findings demonstrate unexpected roles of calreticulin and ER Ca2+ homeostasis/signaling in the canonical WNT signaling pathway.










Similar content being viewed by others
Abbreviations
- ER:
-
Endoplasmic reticulum
- BMP:
-
Bone morphogenetic protein
- BAPTA-AM:
-
1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid
- CaMKII:
-
Ca2+/calmodulin kinase II
- c/EBP:
-
CCAAT/enhancer binding protein
- EpiS:
-
Epiblast like stem cell
- Fgf:
-
Fibroblast growth factor
- GSK3β:
-
Glycogen synthase kinase 3β
- JNK:
-
c-Jun kinase
- Klf4:
-
Krueppel-like factor 4
- MEF2c:
-
Myocyte enhancer factor 2
- αMHC:
-
α myosin heavy chain
- NLK:
-
Nemo-like kinase
- Oct-4:
-
Octamer-binding protein 4
- Otx2:
-
Orthodenticle homeobox 2
- PPAR:
-
Peroxisome proliferator-activated receptor
- PTP1B:
-
Protein tyrosine phosphatase 1B
- SERCA:
-
Sarco/endoplasmic reticulum Ca2+-ATPase
- Smad:
-
Mothers against decapentaplegic homolog
- Sox2:
-
Sex determining region Y-box 2
- TCF:
-
T-cell factor
- TGF:
-
Transforming growth factor
References
Michalak, M., Groenendyk, J., Szabo, E., Gold, L. I., & Opas, M. (2009). Calreticulin, a multi-process calcium-buffering chaperone of the endoplasmic reticulum. Biochemistry Journal, 417, 651–666.
Fadel, M. P., Dziak, E., Lo, C. M., et al. (1999). Calreticulin affects focal contact-dependent but not close contact-dependent cell-substratum adhesion. Journal of Biological Chemistry, 274, 15085–15094.
Faustino, R. S., Chiriac, A., Niederlander, N. J., et al. (2010). Decoded calreticulin-deficient embryonic stem cell transcriptome resolves latent cardiophenotype. Stem Cells, 28, 1281–1291.
Lynch, J., Guo, L., Gelebart, P., et al. (2005). Calreticulin signals upstream of calcineurin and MEF2C in a critical Ca2+-dependent signaling cascade. Journal of Cell Biology, 170, 37–47.
Li, J., Pucéat, M., Perez-Terzic, C., et al. (2002). Calreticulin reveals a critical Ca2+ checkpoint in cardiac myofibrillogenesis. Journal of Cell Biology, 158, 103–113.
Ueno, S., Weidinger, G., Osugi, T., et al. (2007). Biphasic role for Wnt/beta-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proceedings of the National Academy of Sciences of the United States of America, 104, 9685–9690.
Paige, S. L., Osugi, T., Afanasiev, O. K., Pabon, L., Reinecke, H., & Murry, C. E. (2010). Endogenous Wnt/beta-catenin signaling is required for cardiac differentiation in human embryonic stem cells. PLoS One, 5, e11134.
Cohen, E. D., Wang, Z., Lepore, J. J., et al. (2007). Wnt/beta-catenin signaling promotes expansion of Isl-1-positive cardiac progenitor cells through regulation of FGF signaling. Journal of Clinical Investigation, 117, 1794–1804.
Clevers, H., & Nusse, R. (2012). Wnt/beta-catenin signaling and disease. Cell, 149, 1192–1205.
Yang, F., Zeng, Q., Yu, G., Li, S., & Wang, C. Y. (2006). Wnt/beta-catenin signaling inhibits death receptor-mediated apoptosis and promotes invasive growth of HNSCC. Cellular Signalling, 18, 679–687.
Lai, S. L., Chien, A. J., & Moon, R. T. (2009). Wnt/Fz signaling and the cytoskeleton: potential roles in tumorigenesis. Cell Research, 19, 532–545.
Brannon, M., Gomperts, M., Sumoy, L., Moon, R. T., & Kimelman, D. (1997). A beta-catenin/XTcf-3 complex binds to the siamois promoter to regulate dorsal axis specification in Xenopus. Genes & Development, 11, 2359–2370.
Gottardi, C. J., & Gumbiner, B. M. (2004). Role for ICAT in beta-catenin-dependent nuclear signaling and cadherin functions. American Journal of Physiology. Cell Physiology, 286, C747–C756.
Luna-Ulloa, L. B., Hernandez-Maqueda, J. G., Castaneda-Patlan, M. C., & Robles-Flores, M. (2011). Protein kinase C in Wnt signaling: implications in cancer initiation and progression. IUBMB Life, 63, 915–921.
Szabo, E., Qiu, Y., Baksh, S., Michalak, M., & Opas, M. (2008). Calreticulin modulates commitment to adipocyte differentiation. Journal of Cell Biology, 182, 103–116.
Groenendyk, J., & Michalak, M. (2011). A genome-wide siRNA screen identifies novel Phospho-enzymes affecting Wnt/beta-catenin signaling in mouse embryonic stem cells. Stem Cell Reviews and Reports, 7, 910–926.
Coe, H., Jung, J., Groenendyk, J., Prins, D., & Michalak, M. (2010). ERp57 modulates STAT3 signaling from the lumen of the endoplasmic reticulum. Journal of Biological Chemistry, 285, 6725–6738.
Milner, R. E., Busaan, J., & Michalak, M. (1992). Isolation and characterization of different C-terminal fragments of dystrophin expressed in Escherichia coli. Biochemistry Journal, 288, 1037–1044.
Milner, R. E., Baksh, S., Shemanko, C., et al. (1991). Calreticulin, and not calsequestrin, is the major calcium binding protein of smooth muscle sarcoplasmic reticulum and liver endoplasmic reticulum. Journal of Biological Chemistry, 266, 7155–7165.
Nakamura, K., Bossy-Wetzel, E., Burns, K., et al. (2000). Changes in endoplasmic reticulum luminal environment affect cell sensitivity to apoptosis. Journal of Cell Biology, 150, 731–740.
Montalibet, J., & Kennedy, B. P. (2004). Using yeast to screen for inhibitors of protein tyrosine phosphatase 1B. Biochemical Pharmacology, 68, 1807–1814.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Veeman, M. T., Axelrod, J. D., & Moon, R. T. (2003). A second canon. Functions and mechanisms of beta-catenin-independent Wnt signaling. Developmental Cell, 5, 367–377.
Subauste, M. C., Nalbant, P., Adamson, E. D., & Hahn, K. M. (2005). Vinculin controls PTEN protein level by maintaining the interaction of the adherens junction protein beta-catenin with the scaffolding protein MAGI-2. Journal of Biological Chemistry, 280, 5676–5681.
Coghlan, M. P., Culbert, A. A., Cross, D. A., et al. (2000). Selective small molecule inhibitors of glycogen synthase kinase-3 modulate glycogen metabolism and gene transcription. Chemistry & Biology, 7, 793–803.
Fadel, M. P., Szewczenko-Pawlikowski, M., Leclerc, P., et al. (2001). Calreticulin affects ß-catenin associated pathways. Journal of Biological Chemistry, 276, 27083–27089.
Wu, Q., Huang, J. H., Sampson, E. R., et al. (2009). Smurf2 induces degradation of GSK-3beta and upregulates beta-catenin in chondrocytes: a potential mechanism for Smurf2-induced degeneration of articular cartilage. Experimental Cell Research, 315, 2386–2398.
Yum, S., Lee, S. J., Piao, S., et al. (2009). The role of the Ser/Thr cluster in the phosphorylation of PPPSP motifs in Wnt coreceptors. Biochemical and Biophysical Research Communications, 381, 345–349.
Bechard, M., & Dalton, S. (2009). Subcellular localization of glycogen synthase kinase 3beta controls embryonic stem cell self-renewal. Molecular Cell Biology, 29, 2092–2104.
van Noort, M., Meeldijk, J., van der Zee, R., Destree, O., & Clevers, H. (2002). Wnt signaling controls the phosphorylation status of beta-catenin. Journal of Biological Chemistry, 277, 17901–17905.
Fukumoto, S., Hsieh, C. M., Maemura, K., et al. (2001). Akt participation in the Wnt signaling pathway through Dishevelled. Journal of Biological Chemistry, 276, 17479–17483.
Datta, S. R., Brunet, A., & Greenberg, M. E. (1999). Cellular survival: a play in three Akts. Genes & Development, 13, 2905–2927.
Shaw, M., Cohen, P., & Alessi, D. R. (1997). Further evidence that the inhibition of glycogen synthase kinase-3beta by IGF-1 is mediated by PDK1/PKB-induced phosphorylation of Ser-9 and not by dephosphorylation of Tyr-216. FEBS Letters, 416, 307–311.
Amit, S., Hatzubai, A., Birman, Y., et al. (2002). Axin-mediated CKI phosphorylation of beta-catenin at Ser 45: a molecular switch for the Wnt pathway. Genes & Development, 16, 1066–1076.
Yanagawa, S., Lee, J. S., Matsuda, Y., & Ishimoto, A. (2000). Biochemical characterization of the Drosophila axin protein. FEBS Letters, 474, 189–194.
Liu, C., Li, Y., Semenov, M., et al. (2002). Control of beta-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell, 108, 837–847.
Yost, C., Farr, G. H., 3rd, Pierce, S. B., Ferkey, D. M., Chen, M. M., & Kimelman, D. (1998). GBP, an inhibitor of GSK-3, is implicated in Xenopus development and oncogenesis. Cell, 93, 1031–1041.
Piedra, J., Miravet, S., Castano, J., et al. (2003). p120 Catenin-associated Fer and Fyn tyrosine kinases regulate beta-catenin Tyr-142 phosphorylation and beta-catenin-alpha-catenin Interaction. Molecular Cell Biology, 23, 2287–2297.
Roura, S., Miravet, S., Piedra, J., Garciade Herreros, A., & Dunach, M. (1999). Regulation of E-cadherin/Catenin association by tyrosine phosphorylation. Journal of Biological Chemistry, 274, 36734–36740.
Okuda, M., Takahashi, M., Suero, J., et al. (1999). Shear stress stimulation of p130(cas) tyrosine phosphorylation requires calcium-dependent c-Src activation. Journal of Biological Chemistry, 274, 26803–26809.
Muller, T., Choidas, A., Reichmann, E., & Ullrich, A. (1999). Phosphorylation and free pool of beta-catenin are regulated by tyrosine kinases and tyrosine phosphatases during epithelial cell migration. Journal of Biological Chemistry, 274, 10173–10183.
Papp, S., Zhang, X., Szabo, E., Michalak, M., & Opas, M. (2008). Expression of endoplasmic reticulum chaperones in cardiac development. Open Cardiovascular Medicine Journal, 2, 31–35.
Szabo, E., Papp, S., & Opas, M. (2007). Differential calreticulin expression affects focal contacts via the calmodulin/CaMK II pathway. Journal of Cellular Physiology, 213, 269–277.
Papp, S., Fadel, M. P., Kim, H., McCulloch, C. A., & Opas, M. (2007). Calreticulin affects fibronectin-based cell-substratum adhesion via the regulation of c-Src activity. Journal of Biological Chemistry, 282, 16585–16598.
Franco, S. J., Rodgers, M. A., Perrin, B. J., et al. (2004). Calpain-mediated proteolysis of talin regulates adhesion dynamics. Nature Cell Biology, 6, 977–983.
Herr, P., & Basler, K. (2012). Porcupine-mediated lipidation is required for Wnt recognition by Wls. Developmental Biology, 361, 392–402.
Doubravska, L., Krausova, M., Gradl, D., et al. (2011). Fatty acid modification of Wnt1 and Wnt3a at serine is prerequisite for lipidation at cysteine and is essential for Wnt signalling. Cellular Signalling, 23, 837–848.
Arnold, S. J., Stappert, J., Bauer, A., Kispert, A., Herrmann, B. G., & Kemler, R. (2000). Brachyury is a target gene of the Wnt/beta-catenin signaling pathway. Mechanisms of Development, 91, 249–258.
Nichols, J., & Smith, A. (2009). Naive and primed pluripotent states. Cell Stem Cell, 4, 487–492.
Tang, Q. Q., & Lane, M. D. (2012). Adipogenesis: from stem cell to adipocyte. Annual Review of Biochemistry, 81, 715–736.
Nishanian, T. G., Kim, J. S., Foxworth, A., & Waldman, T. (2004). Suppression of tumorigenesis and activation of Wnt signaling by bone morphogenetic protein 4 in human cancer cells. Cancer Biology & Therapy, 3, 667–675.
Chen, G., Deng, C., & Li, Y. P. (2012). TGF-beta and BMP signaling in osteoblast differentiation and bone formation. International Journal of Biological Sciences, 8, 272–288.
Liao, B., Bao, X., Liu, L., et al. (2011). MicroRNA cluster 302–367 enhances somatic cell reprogramming by accelerating a mesenchymal-to-epithelial transition. Journal of Biological Chemistry, 286, 17359–17364.
ten Berge, D., Kurek, D., Blauwkamp, T., et al. (2011). Embryonic stem cells require Wnt proteins to prevent differentiation to epiblast stem cells. Nature Cell Biology, 13, 1070–1075.
Anton, R., Kestler, H. A., & Kuhl, M. (2007). Beta-catenin signaling contributes to stemness and regulates early differentiation in murine embryonic stem cells. FEBS Letters, 581, 5247–5254.
Lyashenko, N., Winter, M., Migliorini, D., Biechele, T., Moon, R. T., & Hartmann, C. (2011). Differential requirement for the dual functions of beta-catenin in embryonic stem cell self-renewal and germ layer formation. Nature Cell Biology, 13, 753–761.
Lynch, J. M., Chilibeck, K., Qui, Y., & Michalak, M. (2006). Assembling pieces of the cardiac puzzle; calreticulin and calcium-dependent pathways in cardiac development, health, and disease. Trends in Cardiovascular Medicine, 16, 65–69.
Mesaeli, N., Nakamura, K., Zvaritch, E., et al. (1999). Calreticulin is essential for cardiac development. Journal of Cell Biology, 144, 857–868.
Christophersen, N. S., & Helin, K. (2010). Epigenetic control of embryonic stem cell fate. Journal of Experimental Medicine, 207, 2287–2295.
Fernando, R. I., Litzinger, M., Trono, P., Hamilton, D. H., Schlom, J., & Palena, C. (2010). The T-box transcription factor Brachyury promotes epithelial-mesenchymal transition in human tumor cells. Journal of Clinical Investigation, 120, 533–544.
Czyz, J., & Wobus, A. (2001). Embryonic stem cell differentiation: the role of extracellular factors. Differentiation, 68, 167–174.
Mohammad, K. S., Javelaud, D., Fournier, P. G., et al. (2011). TGF-beta-RI kinase inhibitor SD-208 reduces the development and progression of melanoma bone metastases. Cancer Research, 71, 175–184.
Lin, J., Fernandez, I., & Roy, K. (2011). Development of feeder-free culture systems for generation of ckit + sca1+ progenitors from mouse iPS cells. Stem Cell Reviews, 7, 736–747.
Lin, S. L., Chang, D. C., Chang-Lin, S., et al. (2008). Mir-302 reprograms human skin cancer cells into a pluripotent ES-cell-like state. RNA, 14, 2115–2124.
Li, M. A., & He, L. (2012). microRNAs as novel regulators of stem cell pluripotency and somatic cell reprogramming. Bioessays, 34, 670–680.
Card, D. A., Hebbar, P. B., Li, L., et al. (2008). Oct4/Sox2-regulated miR-302 targets cyclin D1 in human embryonic stem cells. Molecular Cell Biology, 28, 6426–6438.
Kang, H., Louie, J., Weisman, A., et al. (2012). Inhibition of microRNA-302 (miR-302) by bone morphogenetic protein 4 (BMP4) facilitates the BMP signaling pathway. Journal of Biological Chemistry, 287, 38656–38664.
Chang, S. F., Chang, T. K., Peng, H. H., et al. (2009). BMP-4 induction of arrest and differentiation of osteoblast-like cells via p21 CIP1 and p27 KIP1 regulation. Molecular Endocrinology, 23, 1827–1838.
Brautigam, C., Raggioli, A., & Winter, J. (2013). The Wnt/beta-Catenin pathway regulates the expression of the miR-302 cluster in mouse ESCs and P19 cells. PLoS One, 8, e75315.
Evans, A. L., Faial, T., Gilchrist, M. J., et al. (2012). Genomic targets of Brachyury (T) in differentiating mouse embryonic stem cells. PLoS One, 7, e33346.
Acknowledgments
This work was supported by grants from the Canadian Institutes of Health Research (CIHR) (MOP-53050, MOP-15415, MOP-15291). We thank M. Opas, University of Toronto, for advice and help with immunocytochemistry (Fig. 4). We are grateful to Lori Cormack and Virginie Martin for invaluable help with initial experiments, advice, and support at the initial stages of this work. We thank Michel Puceat, Joanna Jung and Daniel Prins for critical reading of the manuscript and for very helpful comments.
Disclosures
The authors indicate no potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 22.6 kb)
Supplemental Fig. S1
(JPEG 639 kb)
Supplemental Fig. S2
(JPEG 691 kb)
Supplemental Fig. S3
(JPEG 390 kb)
Supplemental Fig. S4
(JPEG 551 kb)
Supplemental Fig. S5
(JPEG 515 kb)
Rights and permissions
About this article
Cite this article
Groenendyk, J., Michalak, M. Disrupted WNT Signaling in Mouse Embryonic Stem Cells in the Absence of Calreticulin. Stem Cell Rev and Rep 10, 191–206 (2014). https://doi.org/10.1007/s12015-013-9488-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-013-9488-6