Opinion statement
-
Herpes simplex encephalitis (HSE) is a neurologically devastating illness associated with substantial morbidity and mortality in adults and older children.
-
The correct recognition of HSE, and distinguishing it from other forms of encephalitis, brain abscess or mimicking disorders early in the course of evaluation facilitates treatment decisions when neurologic complications occur.
-
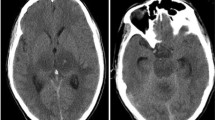
The combination of clinical characteristics, magnetic resonance imaging of the head, and polymerase chain reaction (PCR) analysis of cerebrospinal fluid for herpes simplex type 1 DNA is a sensitive and specific way of diagnosing HSE for proper treatment.
-
Early treatment is essential. HSE is treated with acyclovir 10 mg/kg every 8 hours intravenously for 21 days, unless the illness is mild, or if nephrotoxicity occurs.
-
Whether prolonged therapy with oral antiviral treatment after standard intravenous acyclovir offers any advantage from the standpoint of neurologic morbidity or mortality is unproven.
-
PCR-negative typical cases and PCR-positive atypical cases of HSE can occur and require treatment with acyclovir.
Similar content being viewed by others
References and Recommended Reading
Whitley RJ, Alford CA, Hirsh MS, et al.: Vidarabine versus acyclovir therapy in herpes simplex encephalitis. N Engl J Med 1986, 314:144–149. This paper is proof of efficacy of acyclovir in HSE over other agents.
Skoldenberg B, Forsgren M, Alestig K, et al.: Acyclovir versus vidarabine in herpes simplex encephalitis: randomized multi-center study in consecutive Swedish patients. Lancet 1984, 2:707–711. This paper also provides proof of efficacy of acyclovir in HSE over other agents.
Whitley R, Lakeman AD, Nahmias A, Roizman B: DNA restriction enzyme analysis of herpes simplex virus isolates obtained from patients with encephalitis. N Engl J Med 1982, 307:1060–1062. This is an old but elegant study not likely to be repeated which examines whether viral isolates of the trigeminal ganglion and brain in the same patient are identical or not. It was a draw, 50% were, and 50% were not, keeping the mystery of pathogenesis alive.
Esiri MM: Herpes simplex encephalitis in immunohistological study of the distribution of viral antigen within the brain. J Neurol Sci 1982, 54:209–226. This study is old, but very detailed, about the virologic distribution of HSV in the brain of HSE patients.
Domingues RB, Lakeman FD, Mayo MS, Whitley RJ: Application of competitive PCR to cerebrospinal fluid samples from patients with herpes simplex encephalitis. J Clin Microbiol 1998, 36:2229–2234. This study shows that PCR HSV viral load in the cerebrospinal fluid correlates with the outcome of disease, and emphasizes the need for early treatment.
Domingues RB, Fink MCD, Tsanaclis AMC, et al.: Diagnosis of herpes simplex encephalitis by a magnetic resonance imaging and polymerase chain reaction assay of cerebrospinal fluid. J of Neuro Sci 1998, 157:148–153. This study provides nice correlation between MRI and spinal fluid studies of HSE.
Schroth G, Gawehn J, Thron A, et al.: Early diagnosis of herpes simplex encephalitis by MRI. Neurology 1987, 37:179–183. This is one of the early studies pointing to the superiority of MRI for evaluating HSE.
Cinque P, Cleator GM, Weber T, et al.: The role of laboratory investigation in the diagnosis and management of the patients with suspected herpes simplex encephalitis: a consensus report. J Neurol Neurosurg Psych 1996, 61:339–345. This is a consensus report on the efficacy of PCR as a diagnostic tool for HSE.
Rowley AH, Whitley RJ, Lakeman FD, Wolinsky SM: Rapid detection of herpes simplex virus DNA in cerebrospinal fluid of patients with herpes simplex encephalitis. Lancet 1990, 1:440–441.
Espy MJ, Ross TK, Teo R, et al.: Evaluation of Light Cycler PCR for implementation of laboratory diagnosis of herpes simplex virus infections. J Clin Microbiol 2000, 38:3116–3118.
McDermott SS, McDermott PF, Skare J, et al.: Positive CSF HSV PCR in patients with GBM: a note of caution. Neurology 2000, 54:746–749. One example, among others in the literature, and many clinicians’ personal experience that false positive PCR can occur, and clinical and MRI data need to be integrated in every case.
Puchhammer-Stockl E, Presterl E, Croy C, et al.: Screening for possible failure of herpes simplex virus PCR in spinal fluid for the diagnosis of herpes simplex encephalitis. J Med Virol 2001, 64:531–536.
Lakeman FD, Whitley RJ: Diagnosis of herpes simplex encephalitis: application of polymerase chain reaction to cerebrospinal fluid from brain-biopsied patients and correlation with disease. J Infect Dis 1995, 171:857–863.
Aurelius E, Johansson B, Skoldenberg B, et al.: Rapid diagnosis of herpes simplex encephalitis by nested polymerase chain reaction assay of cerebrospinal fluid. Lancet 1991, 337:189–192.
O’Sullivan CE, Aksamit AJ, Harrington JR, et al.: Clinical spectrum and laboratory characteristics associated with detection of herpes simplex virus DNA in cerebrospinal fluid. Mayo Clin Proc 2003, 78:1347–1352.
Gordon B, Selnes OA, Hart J, et al.: Long term cognitive sequelae of acyclovir treated herpes simplex encephalitis. Arch Neurol 1990, 47:646–647. This study is a detailed example of the serious long-term neurologic sequelae of HSE.
Crumpacker CS, Gonzalez RG, Makar RS: Case 26-2003: A fifty-year-old Columbian man with fever and seizures. N Engl J Med 2003, 349:789–796.
Keating MR: Antiviral agents for non-human immunodeficiency virus infections. Mayo Clin Proc 1999, 74:1266–1283.
Collaborative Antiviral Study Group: study #204, Phase-III double-blind placebo controlled trial of long term therapy of herpes simplex encephalitis: An evaluation of valacyclovir. http://www.casg.uab.edu/. Accessed October 14, 2004. This is the CASG website for information on the ongoing treatment trial of of valacyclovir in HSE.
Baringer JR, Klasssen T, Grumm F: Experimental herpes simplex virus encephalitis: effect of corticosteroids and pyrimidine nucleoside. Arch Neurol 1976, 33:442–446.
Kimberlin DW, Lin C-Y, Jacobs RF, et al.: Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections. Pediatrics 2001, 108:230–238.
Forghani B, Dupuis KW, Schmidt NJ: Rapid detection of herpes simplex virus DNA in human brain tissue by in situ hybridization. J Clin Microbiol 1985, 22:656–658.
Spuler A, Blaszyk H, Parisi JE, Davis DH: Herpes simplex encephalitis after brain surgery: case report in review of the literature. J Neurol Neurosurg Psych 1999, 67:239–242.
Aldea S, Joly L-M, Roujeau T, et al.: Post-operative herpes simplex virus encephalitis after neurosurgery: case report and review of the literature. Clin Inf Dis 2003, 36:96–99.
Thieben MJ, Lennon VA, Boeve BF, et al.: Potentially reversible autoimmune limbic encephalitis with neuronal potassium channel antibody. Neurology 2004, 62:1177–1182.
Vincent A, Buckley C, Schott JM, et al.: Potassium channel antibody-associated encephalopathy: a potentially immunotherapy-responsive form of limbic encephalitis. Brain 2004, 127:701–712. These are two studies about the new entity of autoimmune limbic encephalitis that mimics HSE in many ways, and can be treated.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aksamit, A.J. Herpes simplex encephalitis in adults and older children. Curr Treat Options Neurol 7, 145–150 (2005). https://doi.org/10.1007/s11940-005-0023-1
Issue Date:
DOI: https://doi.org/10.1007/s11940-005-0023-1