Abstract
Background
Although patients experience hair loss and dry skin which may be attributable to deficiency in essential fatty acids (EFAs), the impact of bariatric surgeries on EFA status is unknown.
Methods
This study aimed to assess plasma phospholipid fatty acid profiles following adjustable gastric banding (AGB), which restricts dietary fat intake, versus Roux-en-Y gastric bypass (RYGB), which also promotes fat malabsorption. Serial measures were obtained before and 1 and 6 months from women undergoing RYGB (N = 13) and AGB (N = 5). Measures included the composition of plasma fatty acids in phospholipids, dietary intake, and body fat mass. Friedman and Mann–Whitney tests were used to assess differences over time and between groups, respectively, p < 0.05.
Results
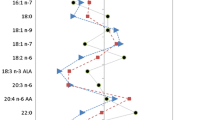
Dietary intake of fats decreased equally at 1 and 6 months following RYGB and AGB. By 6 months, the RYGB group lost more body fat. There were no remarkable changes in EFA in plasma phospholipids following AGB. However, following RYGB, a transient increase in 20:4N6 (+18 %) and a decrease in 20:3N6 at 1 (−47 %) and 6 months (−47 %) were observed. Similar changes were observed in N3 fatty acids following RYGB, including a transient increase in 22:6N3 (+11 %) and decreases in 20:5N3 (−79 and −67 % at 1 and 6 months, respectively). EFA status improved following surgery in the RYGB group.
Conclusions
We demonstrate alterations in plasma EFA following RYGB. The status of EFA improved, but the decrease in 20:5N3, the precursor for anti-inflammatory eicosanoids, may be a concern.
Similar content being viewed by others
Abbreviations
- EFA:
-
Essential fatty acids
- RYGB:
-
Roux-en-Y gastric bypass
- AGB:
-
Adjustable gastric banding
- AA:
-
Arachidonic acid
- EPA:
-
Eicosapentaenoic acid
- DHA:
-
Docosahexaenoic acid
- LA:
-
Linoleic acid
- ALA:
-
Alpha-linolenic acid
- A-CTSI:
-
Atlanta Clinical and Translational Science Institute
- BMI:
-
Body mass index
- CRP:
-
C-reactive protein
- FFA:
-
Free fatty acids
References
Holman RT, Johnson SB, Hatch TF. A case of human linolenic acid deficiency involving neurological abnormalities. Am J Clin Nutr. 1982;35:617–23.
Davis BC, Kris-Etherton PM. Achieving optimal essential fatty acid status in vegetarians: current knowledge and practical implications. Am J Clin Nutr. 2003;78:640–6.
Nakamura MT, Nara TY. Structure, function, and dietary regulation of delta 6, delta 5, and delta 9 desaturates. Annu Rev Nutr. 2004;24:345–76.
Araya J, Rodrigo R, Pettinelli P, et al. Decreased liver fatty acid delta-6 and delta-5 desaturase activity in obese patients. Obesity (Silver Spring). 2010;18:1460–3.
Brignardello J, Morales P, Diaz E, et al. Increase of plasma fatty acids without changes in n-6/n-3-PUFA ratio in asymptomatic obese subjects. Arch Latinoam Nutr. 2011;61:149–53.
Blond JP, Henchiri C, Bezard J. Delta 6 and delta 5 desaturase activities in liver from obese Zucker rats at different ages. Lipids. 1989;24:389–95.
Rodriguez Y, Giri M, Rottiers R, et al. Obese type 2 diabetics and obese patients have comparable plasma phospholipid fatty acid compositions deviating from that of healthy individuals. Prostaglandins Leukot Essent Fat Acids. 2004;71:303–8.
Steffen BT, Steffen LM, Tracy R, et al. Obesity modifies the association between plasma phospholipid polyunsaturated fatty acids and markers of inflammation: the Multi-Ethnic Study of Atherosclerosis. Int J Obes. 2012;36:797–804.
Mueller-Cunningham WM, Quintana R, Kasim-Karakas SE. An ad libitum, very low-fat diet results in weight loss and changes in nutrient intakes in postmenopausal women. J Am Diet Assoc. 2003;103:1600–6.
Noakes M, Clifton PM. Changes in plasma lipids and other cardiovascular risk factors during 3 energy-restricted diets differing in total fat and fatty acid composition. Am J Clin Nutr. 2000;71:706–12.
USDA/HHS. Dietary guidelines for Americans, 2010. U.S. Government Printing Office. Washington, DC: 2010.
Tang AB, Nishimura KY, Phinney SD. Preferential reduction in adipose tissue a-linolenic acid (18:3w3) during very low calorie dieting despite supplementation with 18:3w3. Lipids. 1993;28:987–93.
Cunnane SC, Ross R, Bannister JL, et al. β-oxidation of linoleate in obese men undergoing weight loss. Am J Clin Nutr. 2001;73:709–14.
Phinney SD, Davis PG, Johnson SB, et al. Obesity and weight loss alter serum phospholipids in humans. Am J Clin Nutr. 1991;53:831–8.
Holman RT, Adams CE, Nelson RA, et al. Patients with anorexia nervosa demonstrate deficiencies of selected fatty acid, compensatory changes in nonessential fatty acids and decreased fluidity of plasma lipids. J Nutr. 1995;125:901–7.
Sweene I, Rosling A, Tengblad S, et al. Essential fatty acid status in teenage girls with eating disorders and weight loss. Acta Paediatr. 2011;100:762–7.
Christophe A, Vermeulen A. Effects of weight loss on the fatty acid composition of serum lipids in obese women. Ann Nutr Metab. 1992;36:336–42.
Adams TD, Stroup AM, Gress RE, et al. Cancer incidence and mortality after gastric bypass surgery. Obesity (Silver Spring). 2009;17:796–802.
Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366:1567–76.
Sjostrom L, Peltonen M, Jacobson P, et al. Bariatric surgery and long-term cardiovascular events. JAMA. 2012;307:56–65.
Moize V, Andreu A, Rodriguez L, et al. Protein intake and lean tissue mass retention following bariatric surgery. Clin Nutr. 2013;32:550–5.
Carlin AM, Rao DS, Yager KM, et al. Effect of gastric bypass surgery on vitamin D nutritional status. Surg Obes Relat Dis. 2006;2:638–42.
Ruz M, Carrasco F, Rojas P, et al. Iron absorption and iron status are reduced after Roux-en-Y gastric bypass. Am J Nutr. 2009;90:527–32.
Gletsu-Miller N, Wright BN. Mineral malnutrition following bariatric surgery. Adv Nutr. 2013;4:506–17.
Halawi A, Abiad F, Abbas O. Bariatric surgery and its effects on the skin and skin diseases. Obes Surg. 2013;23:408–13.
Coupaye M, Puchaux K, Bogard C, et al. Nutritional consequences of adjustable gastric banding and gastric bypass: a 1-year prospective study. Obes Surg. 2009;19:56–65.
Elizondo A, Araya J, Rodrigo R, et al. Effects of weight loss on liver and erythrocyte polyunsaturated fatty acid pattern and oxidative stress status in obese patients with non-alcoholic fatty liver disease. Biol Res. 2008;41:59–68.
Kumar R, Lieske JC, Collazo-Clavell ML, et al. Fat malabsorption and increased oxalate absorption are common after Roux-en-Y gastric bypass surgery. Surgery. 2011;149:654–61.
Odstrcil EA, Martinez JG, Santa Ana CA, et al. The contribution of malabsorption to the reduction in net energy absorption after long-limb Roux-en-Y gastric bypass. Am J Clin Nutr. 2010;92:704–13.
Gletsu-Miller N, Hansen JM, Jones DP, et al. Loss of total and visceral adipose tissue mass predicts decreases in oxidative stress after weight-loss surgery. Obesity (Silver Spring). 2009;17:439–46.
Lin E, Phillips LS, Ziegler TR, et al. Increases in adiponectin predict improved liver, but not peripheral, insulin sensitivity in severely obese women during weight loss. Diabetes. 2007;56:735–42.
American-College-of-Surgeons. ACS BSCN accreditation program manual. https://www.facs.org/quality-programs/mbsaqip 26 May 2015.
Lin E, Davis SS, Srinivasan J, et al. Dual mechanism for type-2 diabetes resolution after Roux-en-Y gastric bypass. Am Surg. 2009;75:498–502. discussion -3.
Anonymous. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2012;35 Suppl 1:S64–71.
Gletsu-Miller N, Kahn HS, Gasevic D, Liang Z, Frediani JK, Torres WE, Ziegler TR, Phillips LS, Lin E. Sagittal abdominal diameter and visceral adiposity: correlates of beta-cell function and dysglycemia in severely obese women. Obes Surg. 2013.
Liang Y, Roede JR, Dikalov S, et al. Determination of ebselen-sensitive reactive oxygen metabolites (ebROM) in human serum based upon N, N'-diethyl-1,4-phenylenediamine oxidation. Clin Chim Acta. 2012;414:1–6.
Ohta A, Mayo MC, Kramer N, et al. Rapid analysis of fatty acids in plasma lipids. Lipids. 1990;25:742–7.
Antalis CJ, Stevens LJ, Campbell M, et al. Omega-3 fatty acid status in attention-deficit/hyperactivity disorder. Prostaglandins Leukot Essent Fat Acids. 2006;75:299–308.
Holman RT, Smythe L, Johnson S. Effect of sex and age on fatty acid composition of human serum lipids. Am J Clin Nutr. 1979;32:2390–9.
Mechanick JI, Kushner RF, Sugerman HJ, et al. American Association of Clinical Endocrinologists, The Obesity Society, and American Society for Metabolic & Bariatric Surgery medical guidelines for clinical practice for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient. Obesity (Silver Spring). 2009;17 Suppl 1:S1–70.
Garg ML, Thomson AB, Clandinin MT. Interactions of saturated, n-6 and n-3 polyunsaturated fatty acids to modulate arachidonic acid metabolism. J Lipid Res. 1990;31:271–7.
Olbers T, Bjorkman S, Lindroos A, et al. Body composition, dietary intake, and energy expenditure after laparoscopic Roux-en-Y gastric bypass and laparoscopic vertical banded gastroplasty: a randomized clinical trial. Ann Surg. 2006;244:715–22.
Carswell KA, Vincent RP, Belgaumkar AP, Sherwood RA, Amiel SA, Patel AG, le Roux CW. The effect of bariatric surgery on intestinal absorption and transit time. Obes Surg. 2013.
Rossner S, Walldius G, Bjorvell H. Fatty acid composition in serum lipids and adipose tissue in severe obesity before and after six weeks of weight loss. Int J Obes. 1989;13:603–12.
Kunesova M, Phinney S, Hainer V, et al. The responses of serum and adipose fatty acids to a one-year weight reduction regimen in female obese monozygotic twins. Ann N Y Acad Sci. 2002;967:311–23.
Tiikkainen M, Bergholm R, Rissanen A, et al. Effects of equal weight loss with orlistat and placebo on body fat and serum fatty acid composition and insulin resistance in obese women. Am J Clin Nutr. 2004;79:22–30.
Katz DP, Knittle JL. Effects of hypocaloric diet low in essential fatty acids on in vitro human adipose tissue prostaglandin production and essential fatty acid status. Nutrition. 1991;7:256–9.
Kunesova M, Braunerova R, Hlavaty P, et al. The influence of n-3 polyunsaturated fatty acids and very low calorie diet during a short-term weight reducing regimen on weight loss and serum fatty acid composition in severely obese women. Physiol Res. 2006;55:63–72.
Hlavaty P, Kunesova M, Gojova M, et al. Change in fatty acid composition of serum lipids in obese females after short-term weight-reducing regimen with the addition of n-3 long chain polyunsaturated fatty acids in comparison to controls. Physiol Res. 2008;57 Suppl 1:S57–65.
Haugaard SB, Vaag A, Hoy CE, et al. Desaturation of skeletal muscle structural and depot lipids in obese individuals during a very-low-calorie diet intervention. Obesity (Silver Spring). 2007;15:117–25.
Phinney SD, Tang AB, Johnson SB, et al. Reduced adipose 18:3w3 with weight loss by very low calorie dieting. Lipids. 1990;25:798–806.
Stamatikos AD, Paton CM. Role of stearoyl-CoA desaturase-1 in skeletal muscle function and metabolism. Am J Physiol Endocrinol Metab. 2013;305:E767–75.
Demaria EJ, Winegar DA, Pate VW, et al. Early postoperative outcomes of metabolic surgery to treat diabetes from sites participating in the ASMBS bariatric surgery center of excellence program as reported in the Bariatric Outcomes Longitudinal Database. Ann Surg. 2010;252:559–66. discussion 66-7.
Keidar A, Hershkop KJ, Marko L, et al. Roux-en-Y gastric bypass vs sleeve gastrectomy for obese patients with type 2 diabetes: a randomised trial. Diabetologia. 2013;56:1914–8.
Bradley D, Conte C, Mittendorfer B, et al. Gastric bypass and banding equally improve insulin sensitivity and beta cell function. J Clin Invest. 2012;122:4667–74.
Ballantyne GH, Farkas D, Laker S, et al. Short-term changes in insulin resistance following weight loss surgery for morbid obesity: laparoscopic adjustable gastric banding versus laparoscopic Roux-en-Y gastric bypass. Obes Surg. 2006;16:1189–97.
Poslusna K, Ruprich J, de Vries JH, et al. Misreporting of energy and micronutrient intake estimated by food records and 24 hour recalls, control and adjustment methods in practice. Br J Nutr. 2009;101 Suppl 2:S73–85.
Acknowledgments
We thank all study participants. We thank Marnie Van Meter and Mateusz Stochelski for their technical assistance for the fatty acid analysis. This work was supported by the National Institute of Health grants R03 DK067167 and R21 DK 075745 (to NGM), K24 RR023356 (to TRZ), and the Atlanta Clinical and Translational Science Institute grant UL1 RR025008.
Conflict of Interest
The authors declare that they have no competing interests.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Clinical Trial Registry: ClinicalTrials.gov #NCT00228579
Rights and permissions
About this article
Cite this article
Forbes, R., Gasevic, D., Watson, E.M. et al. Essential Fatty Acid Plasma Profiles Following Gastric Bypass and Adjusted Gastric Banding Bariatric Surgeries. OBES SURG 26, 1237–1246 (2016). https://doi.org/10.1007/s11695-015-1876-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1876-3