Abstract
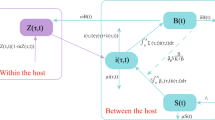
We propose two differential equation-based models to investigate the impact of awareness programs on cholera dynamics. The first model represents the disease transmission rates as decreasing functions of the number of awareness programs, whereas the second model divides the susceptible individuals into two distinct classes depending on their awareness/unawareness of the risk of infection. We study the essential dynamical properties of each model, using both analytical and numerical approaches. We find that the two models, though closely related, exhibit significantly different dynamical behaviors. Namely, the first model follows regular threshold dynamics while rich dynamical behaviors such as backward bifurcation may arise from the second one. Our results highlight the importance of validating key modeling assumptions in the development and selection of mathematical models toward practical application.








Similar content being viewed by others
References
Ahituv A, Hotz VJ, Philipson T (1996) The responsiveness of the demand for condoms to the local prevalence of AIDS. J Hum Resour 31:869–897
Anderson RM, May RM (1991) Infectious diseases of humans: dynamics and control. Oxford University Press, Oxford
Andrews JR, Basu S (2011) Transmission dynamics and control of cholera in Haiti: an epidemic model. Lancet 377(9773):1248–1255
Capasso V, Paveri-Fontana SL (1979) A mathematical model for the 1973 cholera epidemic in the European Mediterranean region. Rev Epidemiol Sante 27(2):121–132
Capasso V, Serio G (1978) A generalization of the Kermack-McKendrick deterministic epidemic model. Math Biosci 42:43–61
Collinson S, Heffernan JM (2014) Modelling the effects of media during an influenza epidemic. BMC Public Health 14:376
Colwell RR (2006) A global and historical perspective of the genus Vibrio. In: Thompson FL, Austin B, Swings J (eds) The biology of vibrios. ASM Press, Washington DC, pp 3–11
Cui J, Tao X, Zhu H (2008) An SIS infection model incorporating media coverage. Rocky Mt J Math 38:1323–1334
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J Math Biol 28:365–382
Einarsdóttir J, Passa A, Gunnlaugsson G (2001) Health education and cholera in rural Guinea-Bissau. Int J Infect Dis 5(3):133–138
Ellwein L, Tran H, Zapata C, Novak V, Olufsen M (2008) Sensitivity analysis and model assessment: mathematical models for arterial blood flow and blood pressure. J Cardiovasc Eng 8:94–108
Faruque SM, Nair GB (2008) Vibrio cholerae: genomics and molecular biology. Caister Academic Press, Poole
Funk S, Gilad E, Janse VAA (2010) Endemic disease, awareness, and local behavioural response. J Theor Biol 264:501–509
Funk S, Salathé M, Jansen VAA (2010) Modelling the influence of human behaviour on the spread of infectious diseases: a review. J R Soc Interface 7(50):1247–1256
Funk S, Bansal S, Bauch CT, Eames KTD, Ken WJ, Edmunds AP, Galvani PK (2015) Nine challenges in incorporating the dynamics of behaviour in infectious diseases models. Epidemics 10:21–25
Gao D, Ruan S (2011) An SIS patch model with variable transmission coefficients. Math Biosci 232:110–115
Global Task Force on Cholera Control (2004) Cholera outbreak: assessing the outbreak response and improving preparedness. World Health Organization, Geneva
Gumel AB (2012) Causes of backward bifurcations in some epidemiological models. J Math Anal Appl 395(1):355–365
Hartley DM, Morris JG, Smith DL (2006) Hyperinfectivity: a critical element in the ability of V. cholerae to cause epidemics? PLoS Med 3(1):e7
Kaur N, Ghosh M, Bhatia SS (2014) Modeling and analysis of an SIRS epidemic model with effect of awareness programs by media. Int J Math Comput Phys Quant Eng 8:233–239
Kiss IZ, Cassell J, Recker M, Simon PL (2010) The impact of information transmission on epidemic outbreaks. Math Biosci 225(1):1–10
Kitaoka M, Miyata ST, Unterweger D, Pukatzki S (2011) Antibiotic resistance mechanisms of Vibrio cholerae. J Med Microbiol 60(4):397–407
LaSalle JP (1976) The stability of dynamical systems. In: Regional conference series in applied mathematics, SIAM, Philadelphia
Leung GM, Lam TH, Ho LM, Ho SY, Chan BH, Wong IO, Hedley AJ (2003) The impact of community psychological responses on outbreak control for severe acute respiratory syndrome in Hong Kong. J Epidemiol Community Health 57(11):857–863
Liu R, Wu J, Zhu H (2007) Media/psychological impact on multiple outbreaks of emerging infectious diseases. Comput Math Methods Med 8:153–164
Longini IM, Nizam A, Ali M, Yunus M, Shenvi N, Clemens JD (2007) Controlling endemic cholera with oral vaccines. PLoS Med 4(11):e336
Lucas ME, Deen JL, von Seidlein L et al (2005) Effectiveness of mass oral cholera vaccination in Beira. Mozambique. N Engl J Med 352(8):757–767
Misra AK, Sharma A, Shukla JB (2011) Modeling and analysis of effects of awareness programs by media on the spread of infectious diseases. Math Comput Model 53:1221–1228
Mukandavire Z, Liao S, Wang J, Gaff H, Smith DL, Morris JG (2011) Estimating the reproductive numbers for the 2008–2009 cholera outbreaks in Zimbabwe. Proc Nat Acad Sci USA 108:8767–8772
Nelson EJ, Harris JB, Morris JG, Calderwood SB, Camilli A (2009) Cholera transmission: the host, pathogen and bacteriophage dynamics. Nat Rev Microbiol 7:693–702
Philipson T (1996) Private vaccination and public health: an empirical examination for US measles. J Hum Resour 31:611–630
Posny D, Wang J (2014) Modelling cholera in periodic environments. J Biol Dyn 8(1):1–19
Shuai Z, van den Driessche P (2013) Global stability of infectious disease models using Lyapunov functions. SIAM J Appl Math 73(4):1513–1532
Samanta S, Rana S, Sharma A, Misra AK, Chattopadhyay J (2013) Effect of awareness programs by media on the epidemic outbreaks: a mathematical model. Appl Math Comput 219(12):6965–6977
Thieme HR (1993) Persistence under relaxed point-dissipativity (with application to an endemic model). SIAM J Math Anal 24:407–435
Tian JP, Wang J (2011) Global stability for cholera epidemic models. Math Biosci 232(1):31–41
Tien JH, Earn DJD (2010) Multiple transmission pathways and disease dynamics in a waterborne pathogen model. Bull Math Biol 72(6):1506–1533
Tracht SM, Del Valle SY, Hyman JM (2010) Mathematical modeling of the effectiveness of facemasks in reducing the spread of novel influenza A (H1N1). PLoS ONE 5(2):e9018
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Wang J, Liao S (2012) A generalized cholera model and epidemic-endemic analysis. J Biol Dyn 6(2):568–589
Wang X, Wang J (2015) Analysis of cholera epidemics with bacterial growth and spatial movement. J Biol Dyn 9(1):233–261
Wang X, Gao D, Wang J (2015) Influence of human behavior on cholera dynamics. Math Biosci 267:41–52
World Health Organization (2009) Public health campaigns: getting the message across. World Health Organization, Geneva
Yorke JA, London WP (1973) Recurrent outbreaks of measles, chickenpox and mumps, II. Systematic differences in contact rates and stochastic effects. Am J Epidemiol 98:469–482
Zuo L, Liu M (2014) Effect of awareness programs on the epidemic outbreaks with time delay. Abstr Appl Anal 2014:940841
Acknowledgements
J.W. is supported in part by NSF (Nos. 1412826 and 1557739). X.W. is partially supported by a Grant from the Simons foundation (No. 317407). D.G. would like to thank the Models of Infectious Disease Agent Study (MIDAS) (NIH NIGMS U01GM087728), the National Natural Science Foundation of China (No. 11601336), the Program for Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning, and Shanghai Gaofeng Project for University Academic Program Development.
Author information
Authors and Affiliations
Corresponding author
Appendix: Global Stability of the Endemic Equilibrium of Model One
Appendix: Global Stability of the Endemic Equilibrium of Model One
Let \(E^*=(S^*, I^*, R^*, B^*, M^*)\) denote an endemic equilibrium of model (1). To establish the global stability of \(E^*\), we make the following assumptions:
for \(0\le I\le N\) and \(M^0\le M\le M_{\mathrm{max}}\), and
for \(M^0\le M\le M_{\mathrm{max}}\) and \(0\le B\le B_{\mathrm{max}}\).
Theorem 4.1
Suppose that (i) assumptions (20) and (21) are satisfied; (ii) \(\xi (M)\equiv \xi \) is constant; (iii) \(\sigma = 0\). If \(\mathcal {R}_0>1\), then system (1) has a unique endemic equilibrium \(E^*\) that is globally asymptotically stable in \(\mathring{\Omega }\).
Proof
For system (1), motivated by Tien and Earn (2010), we consider a Lyapunov function
where \(c_i>0\) (\(i=1,2,3\)) are constants to be determined. It is easy to verify that \(\mathcal {L}\ge 0 \) for all \(S, I, B, M>0\), and \(\mathcal {L}=0\) iff \((S, I, B, M)=(S^*,I^*,B^*,M^*)\). Differentiating \(\mathcal {L}\) along solutions of (1) and applying all equations of (4) except the third one, we obtain
Notice that \(x-1\ge \ln (x)\) for any \(x>0\), and the equality holds iff \(x=1.\) Together with (20), we find that
Likewise, using (21), we obtain
Meanwhile, one can verify that
Similarly, we have
It follows from (23)–(26) that the Eq. (22) yields
Take \(c_1= \xi \eta I^*\), \(c_2 = \eta \beta _2(M^*)S^*B^*/(B^*+K)\) and \(c_3=\xi \beta _1(M^*)S^*I^*\). One can verify by direct calculation that the right-hand side of the inequality (27) is zero. This shows \(\mathcal {L}'\le 0\) with the chosen positive constants \(c_1, c_2\), and \(c_3.\) Moreover, if \(\mathcal {L}'=0\), then there exists a constant \(\hat{k}\) such that
However, by the last equation of (4), \(0=\varLambda + \eta \hat{k} I^*-\nu \hat{k}M^*\). This implies that \(\hat{k}=1\). Meanwhile, \(R=R^*\) which follows from the third equation of (4). Thus, the largest invariant set for which \(\mathcal {L}'=0\) contains only the EE. Therefore, by LaSalle’s invariant principle (LaSalle 1976), the EE is globally asymptotically stable in \(\mathring{\Omega }\) when \(\mathcal {R}_0>1\).
\(\square \)
Rights and permissions
About this article
Cite this article
Yang, C., Wang, X., Gao, D. et al. Impact of Awareness Programs on Cholera Dynamics: Two Modeling Approaches. Bull Math Biol 79, 2109–2131 (2017). https://doi.org/10.1007/s11538-017-0322-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0322-1